This evidence summary presents the best available evidence regarding pregabalin add-on for drug-resistant focal epilepsy.1
Introduction
Approximately 3–3.5% of Australians will experience epilepsy at some point in their lives. This adds up to over 250,000 Australians being diagnosed with epilepsy. Epilepsy is associated with an increased risk of death, up to 2–3 times the general population. Epilepsy is considered as resolved when patients remain seizure-free for 10 years and with no seizure medication for the last five years.2
A recent systematic review found that the prevalence of epilepsy did not differ by age group, sex, or study quality. Furthermore, the lifetime prevalence and incidence rate of epilepsy were higher in low to middle-income countries. Epilepsies of unknown etiology and those with generalised seizures had the highest prevalence compared to other aetiologies.2,3
Antiepileptic drugs (AEDs) are the mainstay of treatment for managing epilepsy. They include first and second-generation anti-epileptics. However, other nonpharmacological treatments have been trialled and these include; the ketogenic diet and neuro-stimulation, which were shown to improve seizure control.3
Pregabalin is one of the second-generation antiepileptics. It has antiepileptic, analgesic, and anxiolytic properties. Pregabalin is not protein bound, is 90% bioavailable, and reaches peak plasma concentrations within 1.5 hours of oral administration. Its steady state is achieved within 24–48 hours. The current evidence summary will present the effectiveness of pregablin as an add-on therapy for drug-resistant focal epilepsy.
Characteristics of the study
The review included randomised controlled trials, double-blind or single-blinded trials, placebo controlled or active controlled and parallel-group or cross-over studies. Participants were included if they were of any age and diagnosed with drug-resistant focal epilepsy (i.e. experiencing simple focal, complex focal, or secondary generalised tonic clonic seizures). Studies where pregabalin was used as an add-on therapy for drug-resistant epilepsy were included.
Quality of the studies
All the included studies had either a low or unclear risk of bias. Selection bias was the main source of bias reported in most of the studies due to incomplete explanation of the randomisation process.
Results
- The following databases were searched; Cochrane Register of Studies (CRS Web, 5 July 2018), MEDLINE (Ovid, 1946 to 5 July 2018), ClinicalTrials.gov using the search strategy and World Health Organization International Clinical Trials Registry Platform (ICTRP, 5 July 2018).
- The primary outcome measure was a 50% or greater reduction in seizure frequency and secondary outcomes were seizure freedom, treatment withdrawal and adverse events.
- The review included nine studies with a total of 3,327 participants. The study participants were randomised to either pregabalin, placebo, or another AED in addition to their usual AEDs.
- Seven out of nine studies compared pregabalin to placebo. Participants randomised to pregabalin were significantly more likely to achieve a 50% or greater reduction in seizure frequency compared to placebo (RR 2.28, 95% CI 1.52 to 3.42).
- Pregabalin was significantly associated with seizure freedom when the dose was increased from 300 mg/day to 600 mg (RR 3.94, 95% CI 1.50 to 10.37).
- Participants were significantly more likely to withdraw from pregabalin treatment group than placebo for any reason (RR 1.35, 95% CI 1.11 to 1.65) and for adverse effects (RR 2.65, 95% CI 1.88 to 3.74, 7 trials).
- Three studies compared pregabalin to three active-control drugs: lamotrigine, levetiracetam and gabapentin. Participants allocated to pregabalin were significantly more likely to achieve a 50% or greater reduction in seizure frequency than those allocated to lamotrigine (RR 1.47, 95% CI 1.03 to 2.12) but not those allocated to levetiracetam (RR 0.94, 95% CI 0.80 to 1.11) or gabapentin (RR 0.96, 95% CI 0.82 to 1.12).
- There was no significant difference between pregabalin and lamotrigine (RR 1.39, 95% CI 0.40 to 4.83) for seizure freedom, however, significantly fewer participants achieved seizure freedom with add-on pregabalin compared to levetiracetam (RR 0.50, 95% CI 0.30 to 0.85).
- There was no significant difference between pregabalin and lamotrigine (RR 1.07, 95% CI 0.75 to 1.52), levetiracetam (RR 1.03, 95% CI 0.71 to 1.49), or gabapentin (RR 0.78, 95% CI 0.57 to 1.07) for treatment withdrawal due to any reason or due to adverse effects.
- The main adverse events experienced by the participants in the included studies were: ataxia, dizziness, somnolence, weight gain and fatigue.
Conclusion
Based on these studies, it was found that pregabalin when used as an add-on therapy was effective at reducing 50% or greater seizure frequency and improve seizure freedom. This was demonstrated in the range of 150 mg to 600 mg/day. Higher doses were associated with increased adverse events frequency.
Implications for research and practice
Pregabalin could be used to manage drug- resistant epilepsy as an add-on therapy for doses up to 600 mg for up to 14 weeks. However, longer-term studies are needed to determine the safety and efficacy profile of pregabalin. Studies addressing health quality of life and economic benefits are needed.
References
- Panebianco M, Bresnahan R, Hemming K, et al. Pregabalin add-on for drug-resistant focal epilepsy. Cochrane Database of Systematic Reviews 2019, Issue 7. At: https://www.ncbi.nlm.nih.gov/pubmed/31287157
- Perucca P, Sche_ er IE, Kiley M. The management of epilepsy in children and adults. Med J Aust 2018;208(5):226–33. At: https://www.mja.com.au/system/files/issues/208_05/10.5694mja17.00951.pdf
- Fiest KM, Sauro KM, Wiebe S, et al. Prevalence and incidence of epilepsy: a systematic review and meta-analysis of international studies. Neurol 2017;88(3)296–303. At: https://www.ncbi.nlm.nih.gov/pubmed/27986877




 Adjunct Professor Manya Angley FPS (CredPharm MMR) Credentialed and Disability Pharmacist
Adjunct Professor Manya Angley FPS (CredPharm MMR) Credentialed and Disability Pharmacist Penny Beirne MPS
Penny Beirne MPS



 This CPD activity is sponsored by Reckitt. All content is the true, accurate and independent opinion of the speakers and the views expressed are entirely their own.[/caption]
This CPD activity is sponsored by Reckitt. All content is the true, accurate and independent opinion of the speakers and the views expressed are entirely their own.[/caption]
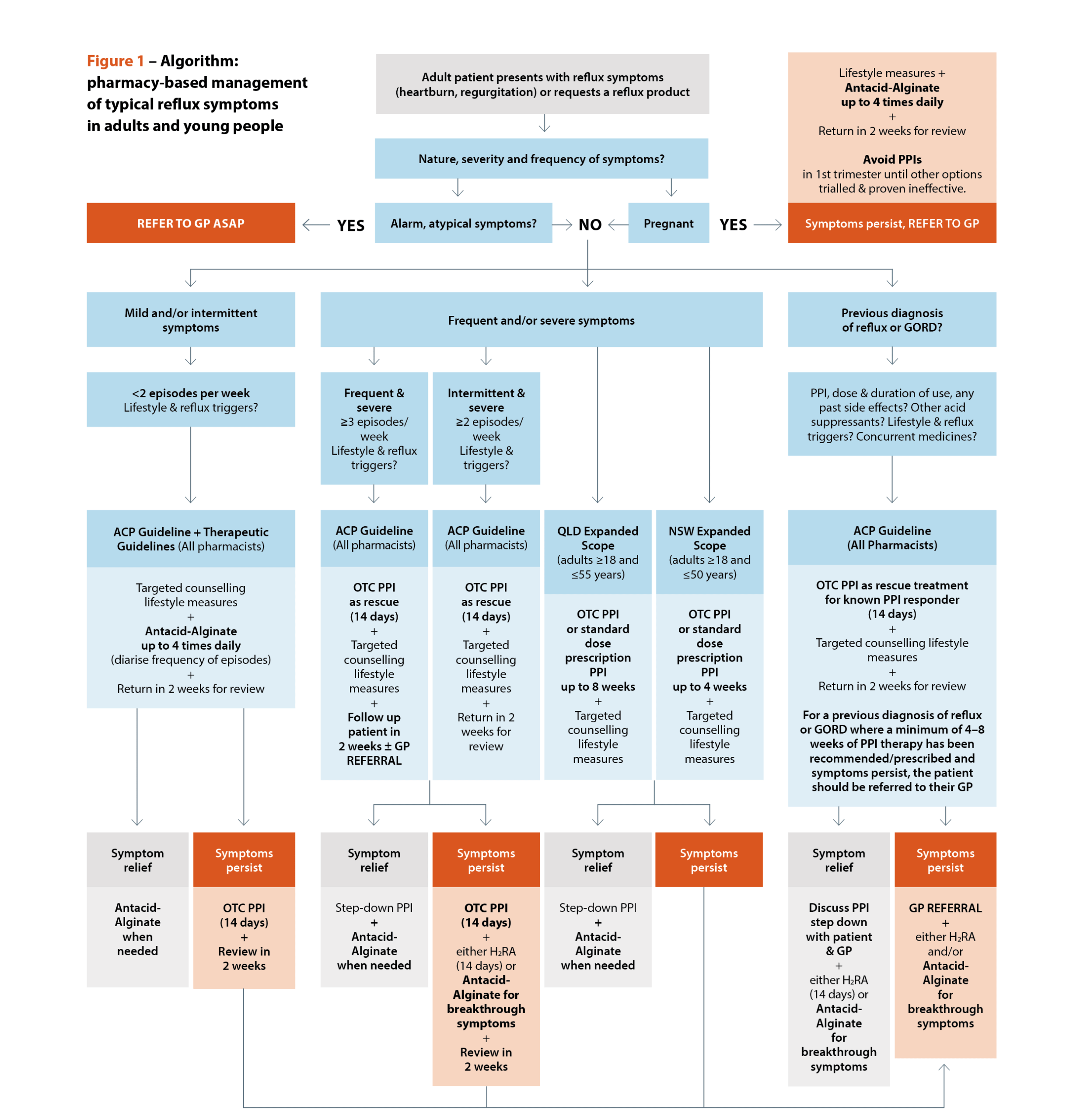 Sources: Australasian College of Pharmacy. Management of reflux: a guideline for pharmacists. Queensland Health. Queensland Community Pharmacy Gastro-oesophageal Reflux and Gastro-oesophageal Reflux Disease – Clinical Practice Guideline. NSW Health. NSW Pharmacist Practice Standards for gastro-oesophageal reflux and gastro-oesophageal reflux disease.[/caption]
Sources: Australasian College of Pharmacy. Management of reflux: a guideline for pharmacists. Queensland Health. Queensland Community Pharmacy Gastro-oesophageal Reflux and Gastro-oesophageal Reflux Disease – Clinical Practice Guideline. NSW Health. NSW Pharmacist Practice Standards for gastro-oesophageal reflux and gastro-oesophageal reflux disease.[/caption]






