

td_module_mega_menu Object
(
[post] => WP_Post Object
(
[ID] => 31597
[post_author] => 12189
[post_date] => 2026-04-09 16:34:44
[post_date_gmt] => 2026-04-09 06:34:44
[post_content] => Case scenario
Kurt, a 52-year-old slightly overweight electrician, comes into your pharmacy complaining of a burning feeling in his chest after meals, which has persisted for a few weeks.
[caption id="attachment_28205" align="alignright" width="388"] This CPD activity is sponsored by Reckitt. All content is the true, accurate and independent opinion of the speakers and the views expressed are entirely their own.[/caption]
You ask about other symptoms and he describes a burning sensation behind his sternum, generally mild but worse when he lies down after dinner, and the occasional sour taste in the back of the throat 1–2 times a week. He shares that he eats irregularly, is a heavy coffee drinker due to shift work and often has large late-evening meals. He finds late-night snacking, alcohol and spicy takeaways aggravate his symptoms. Kurt takes no other medicines, has no allergies and is a non-smoker.
This CPD activity is sponsored by Reckitt. All content is the true, accurate and independent opinion of the speakers and the views expressed are entirely their own.[/caption]
You ask about other symptoms and he describes a burning sensation behind his sternum, generally mild but worse when he lies down after dinner, and the occasional sour taste in the back of the throat 1–2 times a week. He shares that he eats irregularly, is a heavy coffee drinker due to shift work and often has large late-evening meals. He finds late-night snacking, alcohol and spicy takeaways aggravate his symptoms. Kurt takes no other medicines, has no allergies and is a non-smoker.
Learning objectivesAfter reading this article, pharmacists should be able to:
|
As accessible primary healthcare providers, community pharmacists are often the first healthcare provider consulted when a patient presents with symptoms of heartburn, regurgitation or reflux.
Globally, the prevalence of heartburn (retrosternal burning or pain), dyspepsia (indigestion, upper abdomen discomfort) and regurgitation (perception of flow of refluxed stomach content into the mouth or hypopharynx) in adults varies widely. Depending on the definition used and geographical location, prevalence ranges from 2.5% to 52.1%.1
While often asymptomatic, it may result in symptoms caused by refluxate flowing backwards into the oesophagus. This condition, termed gastro-oesophageal reflux (GOR, or simply reflux), is a common physiological event that can occur at any age and is generally considered non-erosive.2
Reflux aetiology involves dysfunction of the lower oesophageal sphincter (LOS) and, in some patients, distal oesophageal motility issues.2,3 A pronounced gastric acid pocket – a highly acidic, unbuffered layer of stomach acid forming on top of food in the upper stomach after a meal, acting as a reservoir that can easily backflow into the oesophagus – significantly contributes to reflux symptoms.4
Other components of gastric juice, such as bile, digestive enzymes and microbial pathogens, can also cause oesophageal symptoms.5-7 Pepsin is an endopeptidase activated at low pH to break down proteins, and can play a pivotal role in damaging the oesophageal surface.5-7 Bile is a digestive fluid designed to break down fats. When bile contacts sensitive mucosa, it can cause painful inflammation and the burning sensation associated with heartburn.5-7 The combination of acid, pepsin and bile can collectively act to assault the oesophageal mucosa.5-7
Gastro-oesophageal reflux disease (GORD) is usually associated with defective LOS function.3 It can cause reflux sufficiently severe to damage mucosa and result in oesophagitis or stricture. A meta-analysis of GORD epidemiology suggested global pooled prevalence of 13.9%.8
Laryngopharyngeal reflux (LPR) occurs when refluxate travels to the throat or higher, and may be the underlying cause of atypical reflux symptoms such as hoarse throat, changes in voice quality, excessive throat clearing and coughing.9 Suspicion of atypical reflux or ‘alarm’ symptoms requires medical referral e.g. cardiac-like chest pain, difficulty swallowing, unintended weight loss, severe abdominal pain, sudden onset reflux symptoms in a person ≥65 years, haematemesis or melaena.3
Symptoms across these conditions overlap and do not distinguish non-erosive from erosive pathology, making it difficult to assign a diagnosis without medical investigation such as endoscopy.
As there is no simple, widely accepted screening tool to differentiate between reflux and GORD, and no correlation between the symptom severity and visible changes in the oesophageal mucosa,10 pharmacists must rely on structured history-taking to identify the most appropriate management.
The use of a mnemonic e.g. Chief Complaint History Taking (LOQQSAM),11 together with targeted age or life-event-related questions, are useful to guide symptom description and triage:
The aim of empiric reflux therapy is to control symptoms and improve quality of life. This review focuses on non-pregnant adults and young people, with the management approach dependent on symptom frequency and severity. Options include risk reduction and prevention through lifestyle modification and/or pharmacological treatment.
Lifestyle modifications
The pharmacist’s role includes assessing for reflux risk factors, particularly those that are modifiable, and advising on strategies to prevent or reduce risk. Non-pharmacological strategies to reduce reflux symptom frequency or severity in this cohort include12,13:
A systematic literature review of clinical trial evidence and clinical practice guidelines14 identified that the strongest levels of evidence for non-pharmacological strategies to improve reflux symptoms were for:
Evidence was categorised with a Grade, based on the National Health and Medical Research Council Levels of Evidence for each study type.15,16
Pharmacological treatment
Most national and international guidelines recommend that a formal diagnostic workup beyond patient history is not required to diagnose the majority of patients who present with upper gastrointestinal symptoms.12,14,17-22
Patient safety is not compromised by typical symptoms of acute reflux (or GORD) being initially managed by pharmacists empirically with a short over-the-counter (OTC) trial of acid suppressant and/or alginate medicines. In Australia, the four OTC therapeutic class options available are: antacids, alginates, histamine-2 receptor antagonists (H2RAs) and proton pump inhibitors (PPIs).
Antacids
Antacid formulations contain salts of magnesium, aluminium, calcium or sodium. Antacids neutralise hydrochloric acid secreted by gastric parietal cells, leading to increased pH of gastric contents – with potency generally expressed in terms of acid neutralising capacity (ANC).23
However, onset and duration of action depend on the dose forms (powder and liquid have a higher ANC than tablets) and extent of chewing (for tablets). Onset of action is rapid, often less than a minute, with duration of action ranging from 20–60 minutes to up to 2–3 hours, based on whether taken fasting or 1 hour after a meal, respectively.18 So, antacids need to be taken frequently during the day to provide efficient relief of persistent symptoms.
Antacids provide rapid but temporary symptom relief. In 2013, the World Gastroenterology Organisation developed guidelines for the community-based management of common gastrointestinal symptoms – recommending antacids as an appropriate OTC option for infrequent, mild or moderate symptoms of heartburn.12
While updated with a global focus in 2017, antacid +/- alginate therapy remained an empirical recommendation for self-care in ‘countries with limited resources and low Helicobacter pylori prevalence’.13 They generally have a good safety profile when used below the maximum daily dose.19
Most impact on bowel function is dose-related, with calcium and magnesium salts causing diarrhoea and aluminium salts causing constipation.18 However, due to the potential for systemic adverse effects with sodium bicarbonate, regular use should be avoided in older people, those with renal or liver impairment, or those on a sodium-restricted diet.24,25
Alginates/antacid-alginate combinations
Alginates are seaweed-derived, natural polysaccharides.26 Most formulations consist of three chemical components: sodium alginate, sodium bicarbonate and calcium carbonate. The latter two constituents react with stomach acid to yield carbon dioxide and free calcium ions, respectively.27
The combination produces a low-density viscous gel (‘raft’) that floats on top of the stomach contents, forming a physical barrier to reduce or prevent the postprandial acid pocket from inducing reflux symptoms.27 Alginates can bind bile salts in refluxate, which contributes to their mucoprotective effect.28
Onset of action, symptom relief and duration of action are brand or formulation specific; alginates typically act quickly after administration, and may provide symptom relief for several hours.27–29 Onset of action has been demonstrated to be slower than antacids but faster than either a PPI or H2RA.29 This attribute is particularly useful for nocturnal symptoms and post-meal reflux.
Antacid-alginate combinations have shown to provide rapid and effective relief of mild to moderate reflux symptoms.24 In 2017, Leiman and colleagues30 published a meta-analysis of randomised-controlled trials (RCTs) for treatment of symptomatic GORD (2,095 adult patients – nine studies [N=900] comparing alginate-based products to placebo or antacid, and five studies [N=1195] to PPIs or H2RAs).
Alginate-based products increased the odds of GORD symptom resolution when compared to placebo or antacids (OR:4.42; 95% CI 2.45–7.97), with a moderate degree of heterogeneity between studies (I2=71%, P=0.001). Alginates appeared less effective than PPIs or H2RAs, but the pooled estimate was not statistically significant (OR:0.58; 95% CI 0.27–1.22).30 While, in clinical trials, alginate caused adverse effects with a comparable rate to placebo,30 they are often combined with antacids or contain excipients that may cause adverse effects.
Histamine-2 receptor antagonists (H2RAs)
By blocking the histamine receptor and histamine stimulation of parietal cell acid secretion, H2RAs competitively suppress both stimulated and basal gastric acid secretion induced by histamine.32 H2RAs have good bioavailability.33
Onset of gastric relief is about 60 minutes (decreasing stomach acid production within 1 to 3 hours), with a dose-dependent duration of action of 4 to 12 hours.34 Although less effective than PPIs, H2RAs are useful for mild and/or intermittent symptoms or when PPIs are unsuitable.17,19 Ranitidine is the only H2RA available in short-course Schedule 2 packs, while others are Schedule 4, restricting OTC H2RA options.
Proton pump inhibitors (PPIs)
PPIs are the most potent OTC acid suppression therapy available.21 They are acid-labile pro-drugs. As food can decrease bioavailability, PPIs are best taken on an empty stomach, once daily 30–60 minutes before breakfast.19 Proton pumps become activated during meals, and administration prior to food intake will enhance efficacy.18,19
PPIs are selectively taken up by gastric parietal cells and concentrated within the acidic canalicular space where they become protonated and convert to an active sulfenamide form. The activated compound then covalently binds to cysteine residues on the luminal surface of the hydrogen/potassium ATPase.
By irreversibly inactivating this proton pump, PPIs suppress both basal and stimulated gastric acid secretion. Acid production remains reduced until new pumps are synthesised, a process that may take up to 36 hours.35
Once absorbed, onset of action is about 90 minutes.36 While PPIs have a short elimination half-life (1–2 hours), their pharmacodynamic effect to reliably maintain intragastric pH >4 can last 15 to 21 hours, due to their irreversible covalent bonding to the proton pump. This provides symptom relief for 24 hours or longer, after 1 to 3 days of treatment.37,38
The delay between PPI initiation and inhibition of acid secretion reaching steady state means that patients with frequent reflux symptoms may benefit from concomitant therapy with an agent that provides short-term relief, such as an antacid-alginate, while waiting for the PPI to take effect. Additionally, by co-localising to the postprandial acid pocket to create a mechanical barrier that suppresses gastric reflux at the LOS, and binding bile salts to contribute to the mucoprotective effect, antacid-alginates may be considered as adjunctive therapy in some patients with breakthrough symptoms, and in PPI deprescribing to help manage symptom rebound.39,40
OTC PPIs are indicated for the initial relief of reflux and GORD as:
In contrast, aligned with the Therapeutic Guidelines (TG), pharmacists approved for expanded scope can prescribe 4 weeks of ‘standard dose’ PPI (esomeprazole 20 mg, lansoprazole 30 mg, omeprazole 20 mg, pantoprazole 40 mg or rabeprazole 20 mg), 0.5–1 hour before a meal.17
In RCTs, 2–4-weeks of PPIs at doses approved for OTC use are usually effective in the management of those with frequent reflux symptoms.37,42,43 A non-response to a short course of acid-suppressive therapy warrants prompt medical consultation, as it can suggest other diagnoses.
Globally, guideline differences in pharmacist empirical adult management of reflux or GORD largely stem from regulatory limitations, differences in definition of symptom severity and frequency, and whether used for initial or maintenance therapy.
TG recommendations include OTC and prescription options from all four reflux medicines classes. For mild, intermittent symptoms (specified as 1–2 episodes weekly), options are an antacid-alginate, combined magnesium-aluminium antacid or H2RA (all when necessary), or standard dose PPI once daily.17
For frequent or severe typical GORD symptoms, TG recommend a 4–8 week initial course of standard dose PPI; then when symptom control is adequate, step down to maintenance therapy (second daily or half the daily dose to therapy only on days when symptoms arise).17
Unfortunately, TG does not provide guidance when reflux (not GORD) symptoms are more frequent or severe. The Australasian College of Pharmacy guidelines attempt a more nuanced, pharmacist-focused approach to initial management (Figure 1).14
[caption id="attachment_31817" align="alignnone" width="2453"]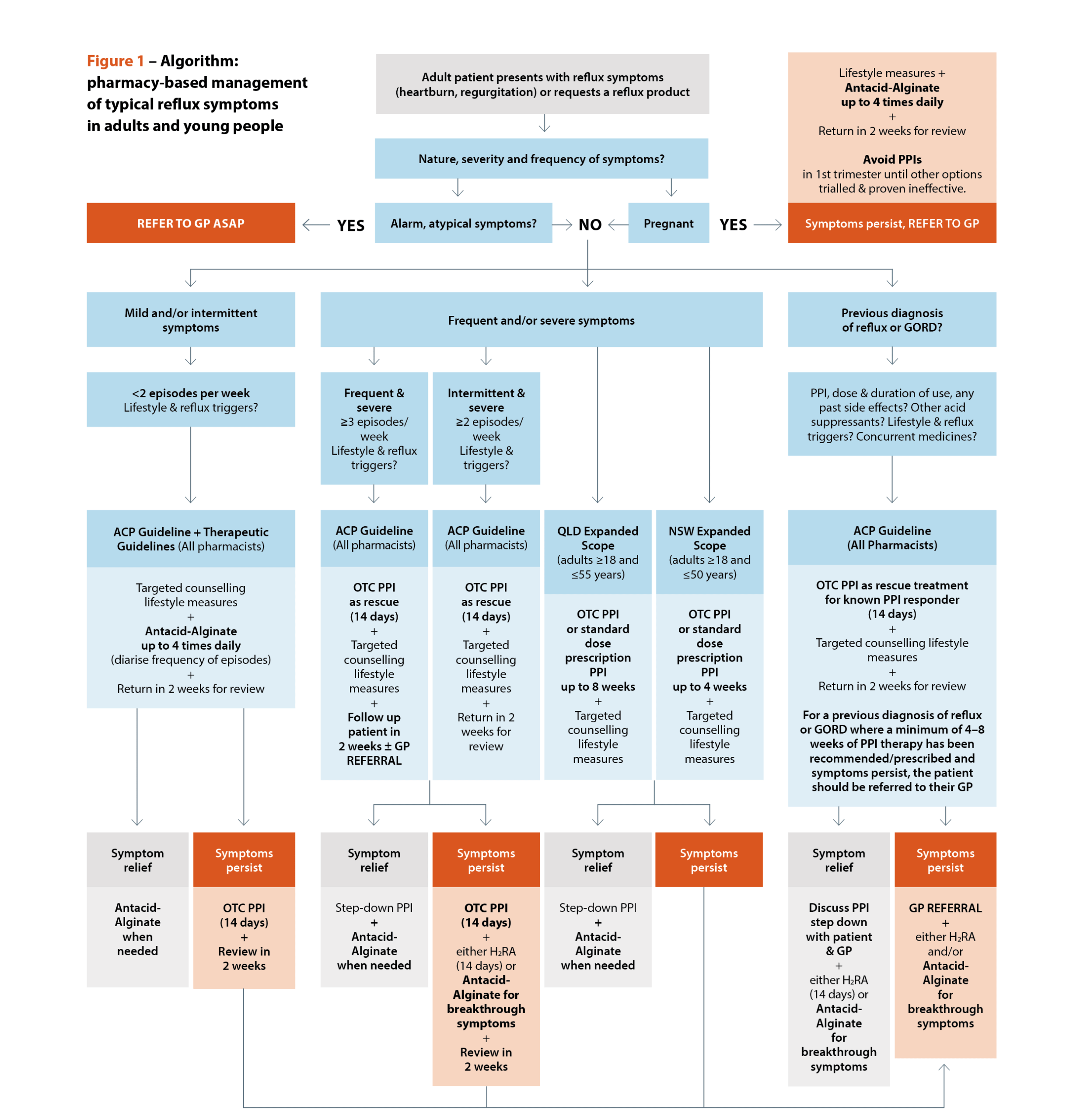 Sources: Australasian College of Pharmacy. Management of reflux: a guideline for pharmacists. Queensland Health. Queensland Community Pharmacy Gastro-oesophageal Reflux and Gastro-oesophageal Reflux Disease – Clinical Practice Guideline. NSW Health. NSW Pharmacist Practice Standards for gastro-oesophageal reflux and gastro-oesophageal reflux disease.[/caption]
Sources: Australasian College of Pharmacy. Management of reflux: a guideline for pharmacists. Queensland Health. Queensland Community Pharmacy Gastro-oesophageal Reflux and Gastro-oesophageal Reflux Disease – Clinical Practice Guideline. NSW Health. NSW Pharmacist Practice Standards for gastro-oesophageal reflux and gastro-oesophageal reflux disease.[/caption]
Irrespective of approach, there is general consensus that13,14,17-19:
Australian pharmacists are expanding their scope of practice, with full scope of practice gaining momentum. In 3 years, the vision has evolved from a 2022 Queensland Government election commitment to work with pharmacy stakeholders to design and implement a pilot of pharmacists practicing to their full scope (the Queensland Community Pharmacy Scope of Practice Pilot) to business-as-usual community pharmacy prescribing services.
Since 1 July 2025, pharmacists completing additional training and meeting the requirements of the Extended Practice Authority have been able to provide clinical advice and treatment, which may include prescribing medicines to adults (18–55 years) for a range of specified acute common conditions, in accordance with a tailored clinical practice guideline.44
NSW Health has also introduced Practice Standards (adults ≥18 to ≤50 years) for expanded pharmacist scope, informed by the Queensland Health clinical practice guidelines and NSW Health emergency care assessment and treatment protocols. It is hoped that a national approach will follow, as other states roll out extended scope services.45 Both jurisdictions list gastro-oesophageal reflux and GORD as an included condition and service, with a companion clinical practice guideline and Practice Standard, respectively.46,47
All pharmacists should take advantage of these reflux resources as they provide an evidence-informed framework for patient assessment and management, primarily aligned to the TG.16 Additional expanded scope training is beneficial to improve pharmacists’ clinical skills and confidence. However, a quality and consistent approach to clinical assessment, triage, and referral is embedded in the competency standards for all pharmacists.48
While extended scope imposes formal responsibilities for patient consultation, documentation and follow-up – pharmacists should undertake these quality care strategies. The main prescribing advantages of expanded scope is extending the initial standard dose PPI treatment period from 2–4 weeks, and, in Queensland, access to Schedule 4 H2RAs. Importantly, the Clinical Practice Guideline and Practice Standard both focus heavily on the use of non-pharmacological management options in reflux symptom management.
Pharmacists play a key role in assessing reflux symptoms through structured history-taking, identifying red flags, and selecting appropriate non-pharmacological and pharmacological options based on symptom frequency and severity. Judicious use of antacid–alginate therapy for rapid relief and adjunctive support during PPI initiation or step-down can optimise symptom control while promoting safe, evidence-based self-management.
Effective management of reflux and GORD in pharmacy practice requires a patient-centred approach that integrates lifestyle modification with tailored use of OTC and, where appropriate, expanded-scope prescribing options. By applying current guidelines and supporting timely review and referral, pharmacists can improve symptom outcomes and quality of life.
Case scenario continuedYou explain to Kurt that his lifestyle is likely contributing to his symptoms and offer targeted counselling to reduce modifiable risks. You provide an OTC PPI once daily, taken 30–60 minutes before his first daily meal for 14 days, and an antacid-alginate, to use for 1–3 days before the PPI starts to work and for breakthrough symptoms, especially at night, when needed. You make an appointment with him to return in 2 weeks to check his progress, or earlier to you or his GP if symptoms worsen. At review, Kurt says he is much improved by his medicines and is avoiding his reflux triggers, especially raising the bedhead. He is happy to step-down to use of antacid-alginate when needed, and return if his symptoms worsen in the future. |
td_module_mega_menu Object
(
[post] => WP_Post Object
(
[ID] => 31434
[post_author] => 12254
[post_date] => 2026-04-09 14:39:38
[post_date_gmt] => 2026-04-09 04:39:38
[post_content] =>
Case scenarioStavros, a 35-year-old male with focal epilepsy, presents to the pharmacy to collect his medicines. He appears tired and mentions he has been having more frequent breakthrough seizures lately. As you chat, he reveals he recently started lorazepam 1 mg twice daily for generalised anxiety disorder (GAD), prescribed by his new GP. Stavros has been missing doses of his anti-seizure medicine, carbamazepine, as he feels it worsens his drowsiness and confusion. |
 Dr Ming S Soh PhD, BPharm (Hons)[/caption]
Dr Ming S Soh PhD, BPharm (Hons)[/caption]
Epilepsy is a chronic neurological condition characterised by a persistent tendency to produce seizures.1,2 According to the International League Against Epilepsy (ILAE), epilepsy is diagnosed when an individual experiences at least two unprovoked seizures more than 24 hours apart, one unprovoked seizure with a high probability (≥60%) of recurrence, or when an epilepsy syndrome is identified.3 Seizures result from abnormal, excessive electrical discharges in the brain, and vary in type, severity and frequency.4 While most seizures are brief, lasting seconds to minutes, they can become prolonged and continuous (i.e. status epilepticus), which requires urgent, intensive medical intervention.4 Globally, epilepsy affects around 50 million people.5,6 It can affect individuals across all age groups, races and ethnicities, and can significantly disrupt quality of life.7,8 Epilepsy is often associated with neurobiological, cognitive, psychological and social sequelae.1,2
Management of epilepsy primarily involves antiseizure medicines (ASM), administered as monotherapy or polytherapy depending on the patient’s response.8,9 In some cases, surgery, dietary therapy or neurostimulation may be considered.9 Treatment complexity, adverse drug reactions and non-adherence to medicines can hinder seizure control and prevent optimal outcomes.8
Learning OutcomesAfter reading this article, pharmacists should be able to:
|
In practice, pharmacists provide therapeutic drug monitoring (hospital setting), Home Medicines Reviews (HMR), MedsChecks and patient counselling, and they support the management of common comorbidities such as depression, anxiety and cognitive impairments, which can complicate treatment.6,8,9

Globally, the pooled incidence rate of epilepsy is estimated at 50–61.4 cases per 100,000 person-years.2,10,11 However, incidence varies by region and income level. In high-income countries, rates typically range from 40–70 per 100,000 person-years, while in low- and middle-income countries, incidence can reach 100–190 per 100,000 person-years, reflecting disparities in healthcare access, diagnostic capacity and exposure to risk factors.2,10 The highest incidence of epilepsy occurs in childhood, reflecting the higher seizure susceptibility of the developing brain.12
In Australia, the lifetime risk of developing epilepsy is estimated to be up to 4%, while approximately 0.6–1% of the population is living with epilepsy at any given time.13,14 Modelled data estimates that prevalence peaks in males aged 60–64 (8.54 per 1,000), and in females aged 50–54 (9.57 per 1,000).15 National surveillance data indicates that prevalence generally remains high into older age groups, largely due to increased rates of stroke, neurodegenerative diseases and brain tumours.2,14,15
Disparities are pronounced among Aboriginal and Torres Strait Islander people, who experience twice the prevalence of epilepsy compared to non-Aboriginal and Torres Strait Islander people.14,16 Aboriginal and Torres Strait Islander people also face higher rates of hospitalisation, reduced access to specialist care and ASM, and significantly elevated epilepsy-related mortality.16 The disability-adjusted life years for Aboriginal and Torres Strait Islander people with epilepsy are double those of non-Aboriginal and Torres Strait Islander people.16
Epilepsy has a multifactorial aetiology. The ILAE classifies epilepsy aetiologies into six main categories: genetic, structural, infectious, metabolic, immune and unknown (see Figure 1).17,18 Genetic mutations generally affect neuronal excitability, especially ion channels, and are linked to specific epilepsy syndromes.18–20 Structural causes include brain malformations, trauma, stroke and tumours. Infections like meningitis or encephalitis can provoke seizures through inflammation. Metabolic causes arise from systemic or inherited metabolic disorders that disrupt brain chemistry or energy production. Autoimmune responses, where the body’s immune system attacks brain tissue, can also lead to seizures. Unknown causes refer to cases where no clear aetiology can be identified despite thorough investigation. Neurodegenerative causes are increasingly recognised as important contributors to epilepsy.18 Despite structured classifications, many overlap. For example, an infection such as encephalitis may lead to structural brain damage.18 Risk factors include family history, perinatal insults, head trauma, brain infections, febrile seizures and socioeconomic disparities, which can influence both the likelihood of developing epilepsy as well as its clinical progression.21
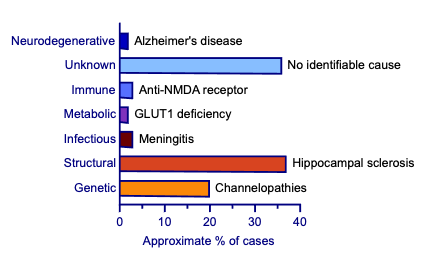
Epilepsy involves several interconnected mechanisms that predominate in different seizure types and epilepsy syndromes and are targeted by specific ASM.
Neuronal hyperexcitability is increased neuronal responsiveness due to ion-channel dysfunction, resulting in spontaneous, excessive neuronal firing. This is a key mechanism underlying all seizure types, but is not the sole pathophysiological mechanism.4,22,23
Hypersynchrony is the abnormal synchronisation of neuronal activity which facilitates seizure propagation across brain regions, particularly in generalised epilepsies such as absence and tonic-clonic seizures.1,4,12,18
Excitatory/inhibitory neurotransmitter imbalance refers to the disruption of glutamate and gamma-aminobutyric acid (GABA) neurotransmission, shifting networks toward hyperexcitability and is central to both focal and generalised epilepsy.4,17
Neuroinflammation refers to the immune activation and cytokine release (e.g. IL-1 beta, TNF-alpha) that can enhance excitability, and are implicated in structural focal epilepsies, post-stroke epilepsy and autoimmune epilepsies. Anti-inflammatory and immunomodulatory therapies may be used in selected cases.4,24
mTOR pathway dysregulation is the abnormal mTOR signalling that alters synaptic plasticity, contributing to epileptogenesis, most notably in tuberous sclerosis complex–associated epilepsy, where mTOR inhibitors such as everolimus are used as targeted therapy.18
Oxidative stress is the excess reactive oxygen species that can damage neurons and lower seizure thresholds, contributing to seizures in metabolic and neurodegenerative epilepsies.24
Mitochondrial dysfunction refers to impaired energy metabolism, which reduces neuronal resilience and is characteristic of mitochondrial epilepsies and some childhood epileptic encephalopathies.24
Synaptic reorganisation refers to structural changes, including aberrant axon sprouting and new excitatory circuit formation, and are commonly seen in temporal lobe epilepsy following brain injury or prolonged seizures.4
Recognising underlying epileptogenic mechanisms aids interpretation of seizure type/classification and guides appropriate ASM selection.
Focal seizures (formerly known as partial)
Focal seizures originate in a specific area of one cerebral hemisphere. Depending on the brain area affected, they may present with localised motor activity like rhythmic jerking or muscle stiffening, sensory changes such as tingling or visual distortions, or autonomic signs, including flushing and gastrointestinal discomfort.12 Awareness may or may not be impaired during these seizures.25 Some focal seizures begin with an aura, such as déjà vu or intense emotions, during which a person remains alert. First-line treatment includes carbamazepine, lamotrigine or levetiracetam, where selection is guided by contraindications and other patient-specific factors.26
Generalised seizures
Generalised seizures involve widespread electrical activity across both cerebral hemispheres and typically include tonic-clonic, absence, myoclonic and atonic seizures.12,25
Tonic-clonic seizures (formerly known as grand mal)
Tonic-clonic seizures are characterised by a sudden loss of consciousness, muscle stiffening followed by rhythmic jerking, and a post-seizure phase with confusion, agitation or fatigue. They can sometimes be fatal due to associated complications. Some tonic-clonic seizures begin as focal seizures with preserved awareness, but progress to loss of consciousness as they generalise across both hemispheres of the brain.12 First-line treatment is sodium valproate, unless contraindicated or otherwise inappropriate for the patient (e.g. individuals of childbearing potential).26
Absence seizures (formerly known as petit mal)
Absence seizures are typically characterised by brief episodes of staring or unresponsiveness, and are often unnoticed or mistaken for daydreaming.12,25 First-line treatment includes ethosuximide or sodium valproate where selection is guided by contraindications and other patient-specific factors.26
Myoclonic seizures
Myoclonic seizures present as sudden and brief muscle jerks.12,25 First-line treatment is sodium valproate unless contraindicated or otherwise inappropriate (e.g. individuals of childbearing potential).26
Atonic seizures
Atonic seizures result in a sudden loss of muscle tone, leading to falls.12,25
For further information, see Australian Medicines Handbook: Epilepsy.26
Diagnosis of epilepsy involves detailed clinical history, neurological examination and supportive investigations.12,17 Electroencephalogram (EEG) is central for detecting epileptiform discharges, while neuroimaging techniques such as computed tomography (CT) and magnetic resonance imaging (MRI) can help identify structural causes.12 In complex cases, additional tools including positron emission tomography (PET)/single photon emission computed tomography (SPECT), magnetoencephalography (MEG) and genetic testing can support diagnosis and guide treatment. The advanced neuroimaging modalities are especially useful for presurgical evaluations in specialised centres.12 Neuropsychological assessments can also provide insight into cognitive impacts.1 ILAE classification recommends a structured, three-level diagnostic approach, identifying seizure type, epilepsy type and specific epilepsy syndrome, while integrating aetiology at each stage to inform treatment decisions.3 Syncope, psychogenic non-epileptic seizures, transient ischaemic attacks, migraine auras and sleep disorders can mimic epileptic seizures but differ in aetiology and treatment.12,27
Prognosis varies widely, depending on seizure type, aetiology, age of onset and treatment response.12 Many patients can achieve seizure control with ASM, and some enter long-term remission. However, around one-third of patients develop drug-resistant epilepsy, unresponsive to two or more ASM.12 Early age of onset, especially in infancy, and epilepsy with structural lesions such as hippocampal sclerosis, are often linked to poorer outcomes.28,29 Sudden unexpected death in epilepsy remains a serious risk,30 particularly in patients with uncontrolled generalised tonic-clonic seizures and early-onset syndromes such as Dravet syndrome (severe infantile-onset developmental epileptic encephalopathy), which has a 3–5-fold higher mortality risk than the general epilepsy population.31 Early diagnosis and individualised treatment strategies improve outcomes.
The primary aim is to achieve seizure freedom with minimal side effects, improving quality of life. Treatment recommendations follow a stepwise approach, guided by seizure classification, epilepsy type, underlying aetiology, side effect profile, cost, potential for measuring serum drug concentration, pharmacokinetics and patient-specific factors such as age, pregnancy, medicine interactions and comorbidities.17,26,32,33
Initial management involves monotherapy with an appropriate ASM. Dose is generally started low and titrated slowly to achieve seizure control. If control is not achieved with maximally tolerated doses of a single agent, a second agent may be added. The second agent may be considered for monotherapy if the patient and prescriber are willing to gradually withdraw the first agent. If two agents do not promote seizure control, the second agent may be withdrawn gradually and replaced with a third.34
Treatment with an ASM should never be ceased abruptly.34
Key treatment considerations
For further information, see Therapeutic Guidelines: Neurology.34
Evidence is limited for cannabis-derived products. Cannabidiol is approved for specific syndromes but requires specialist oversight.33,34
Non-pharmacological management includes surgery following EEG/MRI evaluation, vagus nerve and deep brain stimulation, ketogenic diet and gene therapy. Yoga, meditation and acupuncture have limited evidence for use.17,31–35
One of the most common challenges during epilepsy treatment is adherence. Missed doses or abrupt discontinuation can trigger recurrent seizures which significantly increase mortality risk.33 Medicine interactions are another significant concern, particularly with older ASM such as phenytoin and carbamazepine, which are enzyme inducers and can alter the efficacy of other medicines.33 Patients should be informed that some medicines can lower their seizure threshold. These include theophylline, pethidine, certain antidepressants (uncommon), and some antipsychotics. Some important medicine interactions are shown in Table 1.

Treatment complications range from mild to severe. Common side effects include fatigue, dizziness, cognitive issues such as memory or attention difficulties, and abdominal upset including pain, nausea and vomiting.33
More serious but less frequent reactions include liver and renal toxicity, bone marrow suppression and skin reactions.26 Long-term use of certain ASM including carbamazepine and phenytoin may also reduce bone density, increasing fracture risk.12,33 Additionally, some ASM including levetiracetam and topiramate can worsen mood disorders, contributing to anxiety and depression.26,33
Age also influences management.32,33 Children often require weight-based dosing and closer monitoring due to variable metabolism, while older adults are more susceptible to adverse effects and medicine interactions due to comorbidities and declining organ function.34
To manage these risks, routine monitoring, including liver and renal function tests, therapeutic drug monitoring, bone health assessments and mental health screening, is essential.
Epilepsy treatment requires regular follow-up to assess seizure control, treatment efficacy and adverse effects. Monitoring for drugs with narrow therapeutic index should also be considered. Treatment plans should be reviewed annually, or sooner, to discuss any changes in seizure occurrence.
Patients in Australia can access further advice and support through multiple channels, including:
Epilepsy Action Australia – education, telehealth and resources
Epilepsy Foundation – seizure management and support services
National Epilepsy Support Service/Epilepsy Smart Australia – 1300 761 487 Monday to Friday, from 9 am to 5 pm (AEST).
Pharmacists support epilepsy management across both community and hospital settings. Hospital pharmacists contribute by implementing therapeutic drug monitoring, especially for ASM with narrow therapeutic windows, collaborating with multidisciplinary teams to optimise treatment plans, monitoring for adverse drug reactions, and managing complex cases involving polytherapy or comorbidities. Community pharmacists can provide ongoing support and medicines counselling, and conduct MedsChecks, detect side effects early, and address mental health concerns. Together, pharmacists provide personalised care that can help improve seizure control, enhance treatment safety and reduce mortality.
With appropriate treatment, many people with epilepsy can achieve good seizure control and lead full, active lives. However, outcomes depend on early intervention, consistent follow-up, monitoring and personalised care. Pharmacist involvement helps reduce complications, improves medicine adherence and supports overall wellbeing.
Case scenario continuedYou recognise that emotional stress, non-adherence and adverse effects are likely precipitating Stavros’ seizures. Concomitant use of lorazepam and carbamazepine contributes to additive CNS depression, causing sedation. As Stavros prefers pharmacological treatment for GAD, you call his GP and suggest sertraline 50 mg daily, noting that carbamazepine may reduce sertraline concentrations via enzyme induction, and clinical response should be monitored. You recommend ceasing lorazepam and encourage cognitive behavioural therapy. The GP reviews Stavros and he returns with a cessation plan for lorazepam and a prescription for sertraline. You counsel on treatment expectations and the importance of adherence. A month later, Stavros returns, feeling alert with no recent seizures. |

DR Ming S Soh PhD, BPharm (Hons) is a Research Fellow at the Florey Institute of Neuroscience and Mental Health, University of Melbourne, with expertise in preclinical models of genetic epilepsy and therapeutic development.
Fisher RS, van Emde Boas W, Blume W, et al. Epileptic seizures and epilepsy: definitions proposed by the International League Against Epilepsy (ILAE) and the International Bureau for Epilepsy (IBE). Epilepsia. 2005;46(4):470–472.
Beghi E. The epidemiology of epilepsy. Neuroepidemiology. 2020;54(2):185–191.
Scheffer IE, Berkovic S, Capovilla G, et al. ILAE classification of the epilepsies: position paper of the ILAE commission for classification and terminology. Epilepsia. 2017;58(4):512–521.
Scharfman HE. The neurobiology of epilepsy. Curr Neurol Neurosci Rep. 2007;7(4):348–354.
World Health Organization. Epilepsy. 2022. Available from: www.who.int/news-room/fact-sheets/detail/epilepsy
Petrides M, Peletidi A, Nena E, et al. The role of pharmacists in enhancing epilepsy care: a systematic review of community and outpatient interventions. J Pharm Policy Pract. 2025;18(1):2487046.
Zack MM. National and state estimates of the numbers of adults and children with active epilepsy—United States, 2015. MMWR Morb Mortal Wkly Rep. 2017;66.
Bacci JL, Zaraa S, Stergachis A, et al. Community pharmacists’ role in caring for people living with epilepsy: a scoping review. Epilepsy Behav. 2021;117:107850.
Reis TM, Campos M, Nagai MM, et al. Contributions of pharmacists in the treatment of epilepsy: a systematic review. Am J Pharm Benefits. 2016;8(3):e55–e60.
Bell GS, Sander JW. The epidemiology of epilepsy: the size of the problem. Seizure. 2001;10(4):306–316.
Fiest KM, Sauro KM, Wiebe S, et al. Prevalence and incidence of epilepsy: a systematic review and meta-analysis of international studies. Neurology. 2017;88(3):296–303.
Stafstrom CE, Carmant L. Seizures and epilepsy: an overview for neuroscientists. Cold Spring Harb Perspect Med. 2015;5(6):a022426.
Mohanannair Geethadevi G, Chen Z, et al. Impact of epilepsy on productivity and quality of life: the Australian epilepsy project. Neurology. 2025;105(5):e214011.
Australian Institute of Health and Welfare. Epilepsy in Australia. Canberra: AIHW; 2022. Available from: www.aihw.gov.au/reports/chronic-disease/epilepsy-in-australia/contents/about
Foster E, Chen Z, Zomer E, et al. The costs of epilepsy in Australia: a productivity-based analysis. Neurology. 2020;95(24):e3221–e3231.
Keenan NF, Aitchison SG, Jetté N, et al. Epilepsy in the Indigenous peoples in Canada, Australia, New Zealand, and the USA: a systematic scoping review. Lancet Glob Health. 2025;13(4):e656–e668.
Vera-González A. Pathophysiological mechanisms underlying the etiologies of seizures and epilepsy. In: Czuczwar SJ, editor. Epilepsy. Brisbane: Exon Publications; 2022. p. 1–13.
Balestrini S, Arzimanoglou A, Blümcke I, et al. The aetiologies of epilepsy. Epileptic Disord. 2021;23(1):1–16.
Perucca P, Bahlo M, Berkovic SF. The genetics of epilepsy. Annu Rev Genomics Hum Genet. 2020;21(1):205–230.
Scheffer IE, Berkovic SF. The genetics of human epilepsy. Trends Pharmacol Sci. 2003;24(8):428–433.
Bhalla D, Godet B, Druet-Cabanac M, Preux P-M. Etiologies of epilepsy: a comprehensive review. Expert Rev Neurother. 2011;11(6):861–876.
Reid CA. Preface: ion channels and genetic epilepsy. J Neurochem. 2024;168(12):3829–3830.
Oyrer J, Maljevic S, Scheffer IE, et al. Ion channels in genetic epilepsy: from genes and mechanisms to disease-targeted therapies. Pharmacol Rev. 2018;70(1):142–173.
Fabisiak T, Patel M. Crosstalk between neuroinflammation and oxidative stress in epilepsy. Front Cell Dev Biol. 2022;10:976953.
Fisher RS, Cross JH, French JA, et al. Operational classification of seizure types by the International League Against Epilepsy: position paper of the ILAE commission for classification and terminology. Epilepsia. 2017;58(4):522–530.
Buckley N, editor. Australian medicines handbook. Adelaide: Australian Medicines Handbook Pty Ltd; 2025. Available from: https://amhonline.amh.net.au/
Cook M. Differential diagnosis of epilepsy. In: Shorvon S, Perucca E, Engel J Jr, editors. The treatment of epilepsy. 4th ed. Hoboken (NJ): John Wiley & Sons; 2015. p. 24–37.
Semah F, Picot M-C, Adam C, et al. Is the underlying cause of epilepsy a major prognostic factor for recurrence? Neurology. 1998;51(5):1256–1262.
Nickels K. Earlier is not always better: outcomes when epilepsy occurs in early life versus adolescence. Epilepsy Curr. 2020;20(1):27–29.
Devinsky O, Hesdorffer DC, Thurman DJ, et al. Sudden unexpected death in epilepsy: epidemiology, mechanisms, and prevention. Lancet Neurol. 2016;15(10):1075–1088.
Cooper MS, McIntosh A, Crompton DE, et al. Mortality in Dravet syndrome. Epilepsy Res. 2016;128:43–47.
Perucca E. General principles of medical management. In: Shorvon S, Perucca E, Engel J Jr, editors. The treatment of epilepsy. 4th ed. Hoboken (NJ): John Wiley & Sons; 2015. p. 110–123.
Perucca P, Scheffer IE, Kiley M. The management of epilepsy in children and adults. Med J Aust. 2018;208(5):226–233.
Therapeutic Guidelines Limited. eTG complete. Melbourne: Therapeutic Guidelines; 2025. Available from: www.tg.org.au
Baxendale S. Complementary and alternative treatments for epilepsy. In: Shorvon S, Perucca E, Engel J Jr, editors. The treatment of epilepsy. Hoboken (NJ): John Wiley & Sons; 2015. p. 298–310.
Johannessen SI, Johannessen Landmark C. Antiepileptic drug interactions: principles and clinical implications. Curr Neuropharmacol. 2010;8(3):254–267.
Buchanan N. Medications which may lower seizure threshold. Aust Prescr. 2001;24(1):8–9.
td_module_mega_menu Object
(
[post] => WP_Post Object
(
[ID] => 31751
[post_author] => 4862
[post_date] => 2026-04-08 11:44:33
[post_date_gmt] => 2026-04-08 01:44:33
[post_content] => New avenues for nurse prescribing have the potential to reshape frontline care. For pharmacists, it’s a live example of how scope expansion translates into real impact.
Nurse prescribing is entering a new phase in Australia, and pharmacists should be paying close attention.
While nurse practitioners and endorsed midwives have been able to prescribe Pharmaceutical Benefits Schedule (PBS) subsidised medicines since 2010, a new pathway will soon expand prescribing to a broader group of experienced nurses.
‘A lot of people wouldn't even know that nurses are already prescribing medications that are funded on the PBS,’ said Denise Lyons, President of the Australian Primary Health Care Nurses Association. ‘But there are more than 3,000 nurse practitioners and endorsed midwives in Australia now who are already prescribing.’
Under a new registration standard which came into effect in September last year, suitably qualified registered nurses will be able to prescribe medicines in Schedules 2, 3, 4 and 8 in partnership with an authorised prescriber.
‘It took until this year for universities to start the course that enables registered nurses to meet [the new standard],’ Ms Lyons said. ‘Most are 6-month postgraduate courses … So it will probably be around July 2026 that we will see the first [wave] of nurses completing the educational requirement.’
For pharmacists, this shift offers a live view of how prescribing reform is built – through legislation, governance, defined scope and, crucially, access to the PBS.
What’s changing?
The new framework is deliberately cautious. To become a designated RN prescriber, a nurse must:
‘Pharmacists and nurses are often people's first point of contact with the healthcare system, and it's really good if we can work to the top of our scope and do what we know we can do, with the right guardrails in place.' denise lyons
td_module_mega_menu Object
(
[post] => WP_Post Object
(
[ID] => 31438
[post_author] => 10832
[post_date] => 2026-04-07 14:50:18
[post_date_gmt] => 2026-04-07 04:50:18
[post_content] => Case scenario
Sarah, a 42-year-old woman, presents to the pharmacy reporting a small lump near the anus, occasional bright red bleeding on toilet paper after bowel movements, anal itching and discomfort. She mentions feeling embarrassed to seek advice and has been self-managing with sitz baths. She reports a history of chronic constipation with frequent training during defecation. She is not taking any regular medicines and requests an over-the-counter product to relieve her symptoms. You take the time to explore Sarah’s symptoms in more detail, to help identify any red flags that would require immediate referral. You ask about the duration, severity and other symptoms such as fatigue, unintentional weight loss and changes in bowel habits, all of which she denies.
Learning objectivesAfter reading this article, pharmacists should be able to:
|
Anal fissures and haemorrhoids are commonly encountered, but their epidemiology is not well documented. Haemorrhoids represent the most frequent reason for proctology consultations, followed by anal fissures.1 Haemorrhoids are estimated to affect up to 40% of adults in the general population, although many individuals are asymptomatic.2 Anal fissures occur in approximately 10–15% of the population.3

An anal fissure is a longitudinal tear in the anal canal, most often occurring in the posterior midline, just below the dentate line.4 Anal fissures are typically classified as primary or secondary, depending on the cause. Primary fissures are usually benign and caused by local trauma, such as the passage of hard stools, prolonged diarrhoea, vaginal delivery, repeated injury or anal penetration.4 Secondary fissures, which are often multiple and located off the midline, are uncommon, accounting for less than 1% of cases.5 They result from underlying conditions like previous anal surgery, inflammatory bowel disease (e.g. Crohn’s disease), colorectal malignancy, infections (e.g. HIV/AIDS), or dermatological conditions (e.g. psoriasis).6 Anal fissures are further described as acute or chronic, with chronic fissures lasting more than 6 weeks.7
[caption id="attachment_31774" align="alignright" width="200"] DR Amy Page (she/her) PhD, MClinPharm, GradDipBiostat, GCertHProfEd, GAICD, GStat, FSHPA, FPS is a consultant pharmacist, biostatistician, and the director of the Centre for Optimisation of Medicines at UWA’s School of Allied Health.[/caption]
DR Amy Page (she/her) PhD, MClinPharm, GradDipBiostat, GCertHProfEd, GAICD, GStat, FSHPA, FPS is a consultant pharmacist, biostatistician, and the director of the Centre for Optimisation of Medicines at UWA’s School of Allied Health.[/caption]
Anal fissures are often mistaken for haemorrhoids because of their overlapping symptoms. Haemorrhoids differ in that they are normal vascular cushions within the anal canal that maintain continence. Although part of normal anatomy, the term ‘haemorrhoid’ is commonly used when these vascular anal cushions, containing a rich arteriovenous network, become enlarged or displaced.8 Haemorrhoids may be classified as internal (located above the dentate line) or external (located below the dentate line). Factors such as constipation, straining during bowel movements and a low-fibre diet are commonly considered to increase the risk of haemorrhoids.9,10 However, the causal relationship between these factors and the development of haemorrhoids remains unclear. A possible genetic component that predisposes to smooth muscle, epithelial and connective tissue dysfunction, may contribute to haemorrhoids.11
It is important to note that certain medicines can contribute to or worsen constipation as an adverse effect, thereby aggravating haemorrhoids and anal fissures.9,10 Common examples include opioids, anticholinergics, and iron and calcium supplements.12 Regular medication review should be incorporated into comprehensive care to identify opportunities for deprescribing and prevent inappropriate prescribing cascades, with further guidance available in the new deprescribing guidelines.13
The symptoms of haemorrhoids may include rectal bleeding, anal pruritus, prolapse, faecal seepage and mucus discharge.7
Internal haemorrhoids are usually painless because the columnar epithelium is insensitive to touch and temperature.8 In contrast, external haemorrhoids are pain sensitive and may become thrombosed, often following straining during defecation.8
Anal fissures, by comparison, typically cause severe tearing pain that is provoked by defecation.4 A key distinguishing feature is that the pain of an anal fissure typically occurs during or immediately after defecation.

For diagnostic purposes, medical practitioners perform a physical examination with the individual lying on their side, gently parting the buttocks to inspect the posterior midline.4 Several other anorectal disorders may present with similar symptoms to anal fissures and haemorrhoids. Differential diagnoses include anal fistula and solitary rectal ulcer syndrome, which can often be distinguished based on their clinical appearance through careful physical examination. An anal fistula typically presents as a draining skin punctum.14 It is an abnormal hollow tract, most often arising from infection of an anal gland.15 Solitary rectal ulcer syndrome may present with anal pain, rectal bleeding, constipation, mucus discharge, excessive straining and a sensation of incomplete evacuation. Despite its name, the condition does not always involve an ulcer. Diagnosis requires a combination of endoscopic and histopathological findings, together with the individual’s reported symptoms.16
Haemorrhoids
[caption id="attachment_31775" align="alignright" width="200"] Hui Wen Quek (she/her) BPharm(Hons), GradCertAppPharmPrac is a pharmacist and PhD candidate at the University of Western Australia (UWA).[/caption]
Hui Wen Quek (she/her) BPharm(Hons), GradCertAppPharmPrac is a pharmacist and PhD candidate at the University of Western Australia (UWA).[/caption]
Non-pharmacological treatment options
First-line management for haemorrhoids usually involves lifestyle measures, which include ensuring adequate fluid and fibre intake, reducing prolonged time on the toilet and avoiding excessive straining, the use of sitz baths, and maintaining anal hygiene.10 In practice, dietary and lifestyle modifications are usually recommended as a conservative and preventive measure for haemorrhoids.17
Pharmacological treatment options
Over-the-counter therapies for internal haemorrhoids include ointments and suppositories that contain emollients, mild astringents, local anaesthetics and/or corticosteroids.8 These preparations temporarily relieve itching and discomfort. Prolonged use of topical corticosteroids is not recommended, as it increases the risk of local infections such as candidiasis.8 Similarly, prolonged use of anaesthetic-containing products should be avoided due to potential adverse effects, including skin sensitisation and dermatitis.8
Outpatient procedures and surgery
For individuals who do not respond to conservative measures, other treatment options for haemorrhoids include outpatient procedures and surgery.10 Outpatient procedures include rubber band ligation, infrared coagulation, bipolar probe, heater probe, sclerotherapy and cryotherapy.10 These are generally reserved for people with grade I or grade II haemorrhoids.10 Surgery can be considered for people who do not respond to or cannot tolerate outpatient procedures, as well as those with large external haemorrhoids, or combined internal and external haemorrhoids with prolapse.10
A recent meta-analysis found that surgical treatments, compared with conservative treatments, offer greater symptom relief, quicker recovery and lower recurrence rates.18 However, they do carry risks of procedure-related complications. In contrast, conservative treatments are safer and less invasive, but tend to provide slower symptom improvement and have higher recurrence rates.18 Therefore, treatment decisions should be individualised, taking into account symptom severity, individual preferences and risk tolerance.
Anal fissures
The primary aim of treatment for anal fissures is to reduce anal sphincter spasm, improve blood flow and promote healing.6 First-line management is usually non-operative and includes increasing dietary fibre, use of sitz baths and applying glyceryl trinitrate 0.2% ointment, three times daily for up to 4 weeks to relieve pain.19 Headache is a common adverse effect of glyceryl trinitrate due to systemic absorption, which could limit its use.20 Other topical agents used in clinical practice include lidocaine and hydrocortisone, although these are less effective than a combination of fibre intake and warm sitz baths.4
Botulinum toxin injections may also be considered and can achieve good healing rates.6,19 However, botulinum toxin may cause temporary reduced control of wind or bowel motions, which usually resolves as the effect of the injection wears off.4 Due to these adverse effects and the invasive nature of the procedure, botulinum toxin is generally reserved as a second-line therapy.4 If these treatment options are unsuccessful, surgical treatment may be required.6
Secondary or chronic anal fissures warrant additional investigation and multidisciplinary management is advised, especially when malignancy is suspected or confirmed.4
Pharmacists play a key role in recognising the symptoms of haemorrhoids and anal fissures, providing timely advice and suggesting a suitable course of action. Pharmacists can also educate individuals on first-line management strategies and provide lifestyle advice where appropriate to prevent recurrence. In addition, pharmacists can advocate for the safe use of over-the-counter treatments, advise on appropriate duration of use and counsel individuals on potential adverse effects.
Referral to a medical practitioner is advisable for individuals who21:
Anal fissures and haemorrhoids are common conditions encountered in primary care and can greatly impact quality of life. Pharmacists play a critical role in early management and referral, helping prevent complications and improve outcomes.
Case scenario continuedYou identify that Sarah has haemorrhoids and provide lifestyle advice, including increasing fluid and dietary fibre intake, avoiding prolonged straining during bowel movements and practising good anal hygiene. For symptomatic relief, you suggest a topical ointment containing cinchocaine 0.5% and zinc oxide 20%, to be applied as needed. You also advise her to consult her general practitioner if she experiences persistent bleeding, ongoing pain or worsening prolapse. With your support and advice, Sarah feels more confident in managing her condition effectively while reducing the risk of recurrence. |
[cpd_submit_answer_button]
Hui Wen Quek (she/her) BPharm(Hons), GradCertAppPharmPrac is a pharmacist and PhD candidate at the University of Western Australia (UWA).
DR Amy Page (she/her) PhD, MClinPharm, GradDipBiostat, GCertHProfEd, GAICD, GStat, FSHPA, FPS is a consultant pharmacist, biostatistician, and the director of the Centre for Optimisation of Medicines at UWA’s School of Allied Health.
Manisha Pillai (she/her) BPharm
Higuero T. Update on the management of anal fissure. J Visc Surg. 2015;152(2 Suppl):S37–S43.
Riss S, Weiser FA, Schwameis K, Riss T, Mittlböck M, Steiner G, et al. The prevalence of hemorrhoids in adults. Int J Colorectal Dis. 2012;27(2):215–220.
Vitton V, Bouchard D, Guingand M, Higuero T. Treatment of anal fissures: results from a national survey on French practice. Clin Res Hepatol Gastroenterol. 2022;46(4):101821.
Schlichtemeier S, Engel A. Anal fissure. Aust Prescr. 2016;39(1):14–17.
Zaghiyan KN, Fleshner P. Anal fissure. Clin Colon Rectal Surg. 2011;24(1):22–30.
Newman M, Collie M. Anal fissure: diagnosis, management, and referral in primary care. Br J Gen Pract. 2019;69(685):409–410.
Lyle V, Young C. Anal fissures: an update on treatment options. Aust J Gen Pract. 2024;53:33–35.
Therapeutic Guidelines Limited. Haemorrhoids. Melbourne: Therapeutic Guidelines Limited; 2022. Available from: https://www.tg.org.au
Lohsiriwat V. Hemorrhoids: from basic pathophysiology to clinical management. World J Gastroenterol. 2012;18(17):2009–2017.
Sandler RS, Peery AF. Rethinking what we know about hemorrhoids. Clin Gastroenterol Hepatol. 2019;17(1):8–15.
Zheng T, Ellinghaus D, Juzenas S, Cossais F, Burmeister G, Mayr G, et al. Genome-wide analysis of 944 133 individuals provides insights into the etiology of haemorrhoidal disease. Gut. 2021;70(8):1538–1549.
Therapeutic Guidelines Limited. Functional constipation in adults. Melbourne: Therapeutic Guidelines Limited; 2022. Available from: https://www.tg.org.au
Quek HW, Reus X, Lee K, Etherton-Beer C, Page A; Guideline Development Group. Deprescribing in older people: a clinical practice guideline. Perth (WA): The University of Western Australia; 2025.
Stewart D. Anal fissure: clinical manifestations, diagnosis, prevention. UpToDate. 2025. Available from: https://www.uptodate.com/contents/anal-fissure-clinical-manifestations-diagnosis-prevention
Therapeutic Guidelines Limited. Anorectal abscess and fistula. Melbourne: Therapeutic Guidelines Limited; 2022. Available from: https://www.tg.org.au
Sadeghi A, Biglari M, Forootan M, Adibi P. Solitary rectal ulcer syndrome: a narrative review. Middle East J Dig Dis. 2019;11(3):129–134.
Lohsiriwat V. Treatment of hemorrhoids: a coloproctologist's view. World J Gastroenterol. 2015;21(31):9245–9252.
Quan L, Bai X, Cheng F, Chen J, Ma H, Wang P, et al. Comparison of efficacy and safety between surgical and conservative treatments for hemorrhoids: a meta-analysis. BMC Gastroenterol. 2025;25(1):492.
Therapeutic Guidelines Limited. Anal fissure. Melbourne: Therapeutic Guidelines Limited; 2022. Available from: https://www.tg.org.au
Australian Medicines Handbook Pty Ltd. Glyceryl trinitrate (rectal). Adelaide: Australian Medicines Handbook Pty Ltd; 2025. Available from: https://amhonline.amh.net.au/
Allen S. Haemorrhoids and anal fissures. S Afr Pharm J. 2008;75(5):36.
td_module_mega_menu Object
(
[post] => WP_Post Object
(
[ID] => 31741
[post_author] => 3148
[post_date] => 2026-04-02 13:54:04
[post_date_gmt] => 2026-04-02 02:54:04
[post_content] => Community pharmacies are often the first place people turn when something feels ‘off’. But as pharmacists know, many requests for over-the-counter medicines are more complex than they may initially seem.
AP took a look at some presentations which might signal hidden red flags.
‘I just want a multivitamin’
When people present to community pharmacies, they often arrive with a solution already in mind. For example, ‘I need a strong painkiller,’ ‘I just want a multivitamin,’ or ‘What do you have for nerve pain?’
Nearly always, consistent with professional standards, these invite an open ended question to uncover the rationale for the request
 Gauri Godbole FPS[/caption]
Gauri Godbole FPS[/caption]
td_module_mega_menu Object
(
[post] => WP_Post Object
(
[ID] => 31597
[post_author] => 12189
[post_date] => 2026-04-09 16:34:44
[post_date_gmt] => 2026-04-09 06:34:44
[post_content] => Case scenario
Kurt, a 52-year-old slightly overweight electrician, comes into your pharmacy complaining of a burning feeling in his chest after meals, which has persisted for a few weeks.
[caption id="attachment_28205" align="alignright" width="388"] This CPD activity is sponsored by Reckitt. All content is the true, accurate and independent opinion of the speakers and the views expressed are entirely their own.[/caption]
You ask about other symptoms and he describes a burning sensation behind his sternum, generally mild but worse when he lies down after dinner, and the occasional sour taste in the back of the throat 1–2 times a week. He shares that he eats irregularly, is a heavy coffee drinker due to shift work and often has large late-evening meals. He finds late-night snacking, alcohol and spicy takeaways aggravate his symptoms. Kurt takes no other medicines, has no allergies and is a non-smoker.
This CPD activity is sponsored by Reckitt. All content is the true, accurate and independent opinion of the speakers and the views expressed are entirely their own.[/caption]
You ask about other symptoms and he describes a burning sensation behind his sternum, generally mild but worse when he lies down after dinner, and the occasional sour taste in the back of the throat 1–2 times a week. He shares that he eats irregularly, is a heavy coffee drinker due to shift work and often has large late-evening meals. He finds late-night snacking, alcohol and spicy takeaways aggravate his symptoms. Kurt takes no other medicines, has no allergies and is a non-smoker.
Learning objectivesAfter reading this article, pharmacists should be able to:
|
As accessible primary healthcare providers, community pharmacists are often the first healthcare provider consulted when a patient presents with symptoms of heartburn, regurgitation or reflux.
Globally, the prevalence of heartburn (retrosternal burning or pain), dyspepsia (indigestion, upper abdomen discomfort) and regurgitation (perception of flow of refluxed stomach content into the mouth or hypopharynx) in adults varies widely. Depending on the definition used and geographical location, prevalence ranges from 2.5% to 52.1%.1
While often asymptomatic, it may result in symptoms caused by refluxate flowing backwards into the oesophagus. This condition, termed gastro-oesophageal reflux (GOR, or simply reflux), is a common physiological event that can occur at any age and is generally considered non-erosive.2
Reflux aetiology involves dysfunction of the lower oesophageal sphincter (LOS) and, in some patients, distal oesophageal motility issues.2,3 A pronounced gastric acid pocket – a highly acidic, unbuffered layer of stomach acid forming on top of food in the upper stomach after a meal, acting as a reservoir that can easily backflow into the oesophagus – significantly contributes to reflux symptoms.4
Other components of gastric juice, such as bile, digestive enzymes and microbial pathogens, can also cause oesophageal symptoms.5-7 Pepsin is an endopeptidase activated at low pH to break down proteins, and can play a pivotal role in damaging the oesophageal surface.5-7 Bile is a digestive fluid designed to break down fats. When bile contacts sensitive mucosa, it can cause painful inflammation and the burning sensation associated with heartburn.5-7 The combination of acid, pepsin and bile can collectively act to assault the oesophageal mucosa.5-7
Gastro-oesophageal reflux disease (GORD) is usually associated with defective LOS function.3 It can cause reflux sufficiently severe to damage mucosa and result in oesophagitis or stricture. A meta-analysis of GORD epidemiology suggested global pooled prevalence of 13.9%.8
Laryngopharyngeal reflux (LPR) occurs when refluxate travels to the throat or higher, and may be the underlying cause of atypical reflux symptoms such as hoarse throat, changes in voice quality, excessive throat clearing and coughing.9 Suspicion of atypical reflux or ‘alarm’ symptoms requires medical referral e.g. cardiac-like chest pain, difficulty swallowing, unintended weight loss, severe abdominal pain, sudden onset reflux symptoms in a person ≥65 years, haematemesis or melaena.3
Symptoms across these conditions overlap and do not distinguish non-erosive from erosive pathology, making it difficult to assign a diagnosis without medical investigation such as endoscopy.
As there is no simple, widely accepted screening tool to differentiate between reflux and GORD, and no correlation between the symptom severity and visible changes in the oesophageal mucosa,10 pharmacists must rely on structured history-taking to identify the most appropriate management.
The use of a mnemonic e.g. Chief Complaint History Taking (LOQQSAM),11 together with targeted age or life-event-related questions, are useful to guide symptom description and triage:
The aim of empiric reflux therapy is to control symptoms and improve quality of life. This review focuses on non-pregnant adults and young people, with the management approach dependent on symptom frequency and severity. Options include risk reduction and prevention through lifestyle modification and/or pharmacological treatment.
Lifestyle modifications
The pharmacist’s role includes assessing for reflux risk factors, particularly those that are modifiable, and advising on strategies to prevent or reduce risk. Non-pharmacological strategies to reduce reflux symptom frequency or severity in this cohort include12,13:
A systematic literature review of clinical trial evidence and clinical practice guidelines14 identified that the strongest levels of evidence for non-pharmacological strategies to improve reflux symptoms were for:
Evidence was categorised with a Grade, based on the National Health and Medical Research Council Levels of Evidence for each study type.15,16
Pharmacological treatment
Most national and international guidelines recommend that a formal diagnostic workup beyond patient history is not required to diagnose the majority of patients who present with upper gastrointestinal symptoms.12,14,17-22
Patient safety is not compromised by typical symptoms of acute reflux (or GORD) being initially managed by pharmacists empirically with a short over-the-counter (OTC) trial of acid suppressant and/or alginate medicines. In Australia, the four OTC therapeutic class options available are: antacids, alginates, histamine-2 receptor antagonists (H2RAs) and proton pump inhibitors (PPIs).
Antacids
Antacid formulations contain salts of magnesium, aluminium, calcium or sodium. Antacids neutralise hydrochloric acid secreted by gastric parietal cells, leading to increased pH of gastric contents – with potency generally expressed in terms of acid neutralising capacity (ANC).23
However, onset and duration of action depend on the dose forms (powder and liquid have a higher ANC than tablets) and extent of chewing (for tablets). Onset of action is rapid, often less than a minute, with duration of action ranging from 20–60 minutes to up to 2–3 hours, based on whether taken fasting or 1 hour after a meal, respectively.18 So, antacids need to be taken frequently during the day to provide efficient relief of persistent symptoms.
Antacids provide rapid but temporary symptom relief. In 2013, the World Gastroenterology Organisation developed guidelines for the community-based management of common gastrointestinal symptoms – recommending antacids as an appropriate OTC option for infrequent, mild or moderate symptoms of heartburn.12
While updated with a global focus in 2017, antacid +/- alginate therapy remained an empirical recommendation for self-care in ‘countries with limited resources and low Helicobacter pylori prevalence’.13 They generally have a good safety profile when used below the maximum daily dose.19
Most impact on bowel function is dose-related, with calcium and magnesium salts causing diarrhoea and aluminium salts causing constipation.18 However, due to the potential for systemic adverse effects with sodium bicarbonate, regular use should be avoided in older people, those with renal or liver impairment, or those on a sodium-restricted diet.24,25
Alginates/antacid-alginate combinations
Alginates are seaweed-derived, natural polysaccharides.26 Most formulations consist of three chemical components: sodium alginate, sodium bicarbonate and calcium carbonate. The latter two constituents react with stomach acid to yield carbon dioxide and free calcium ions, respectively.27
The combination produces a low-density viscous gel (‘raft’) that floats on top of the stomach contents, forming a physical barrier to reduce or prevent the postprandial acid pocket from inducing reflux symptoms.27 Alginates can bind bile salts in refluxate, which contributes to their mucoprotective effect.28
Onset of action, symptom relief and duration of action are brand or formulation specific; alginates typically act quickly after administration, and may provide symptom relief for several hours.27–29 Onset of action has been demonstrated to be slower than antacids but faster than either a PPI or H2RA.29 This attribute is particularly useful for nocturnal symptoms and post-meal reflux.
Antacid-alginate combinations have shown to provide rapid and effective relief of mild to moderate reflux symptoms.24 In 2017, Leiman and colleagues30 published a meta-analysis of randomised-controlled trials (RCTs) for treatment of symptomatic GORD (2,095 adult patients – nine studies [N=900] comparing alginate-based products to placebo or antacid, and five studies [N=1195] to PPIs or H2RAs).
Alginate-based products increased the odds of GORD symptom resolution when compared to placebo or antacids (OR:4.42; 95% CI 2.45–7.97), with a moderate degree of heterogeneity between studies (I2=71%, P=0.001). Alginates appeared less effective than PPIs or H2RAs, but the pooled estimate was not statistically significant (OR:0.58; 95% CI 0.27–1.22).30 While, in clinical trials, alginate caused adverse effects with a comparable rate to placebo,30 they are often combined with antacids or contain excipients that may cause adverse effects.
Histamine-2 receptor antagonists (H2RAs)
By blocking the histamine receptor and histamine stimulation of parietal cell acid secretion, H2RAs competitively suppress both stimulated and basal gastric acid secretion induced by histamine.32 H2RAs have good bioavailability.33
Onset of gastric relief is about 60 minutes (decreasing stomach acid production within 1 to 3 hours), with a dose-dependent duration of action of 4 to 12 hours.34 Although less effective than PPIs, H2RAs are useful for mild and/or intermittent symptoms or when PPIs are unsuitable.17,19 Ranitidine is the only H2RA available in short-course Schedule 2 packs, while others are Schedule 4, restricting OTC H2RA options.
Proton pump inhibitors (PPIs)
PPIs are the most potent OTC acid suppression therapy available.21 They are acid-labile pro-drugs. As food can decrease bioavailability, PPIs are best taken on an empty stomach, once daily 30–60 minutes before breakfast.19 Proton pumps become activated during meals, and administration prior to food intake will enhance efficacy.18,19
PPIs are selectively taken up by gastric parietal cells and concentrated within the acidic canalicular space where they become protonated and convert to an active sulfenamide form. The activated compound then covalently binds to cysteine residues on the luminal surface of the hydrogen/potassium ATPase.
By irreversibly inactivating this proton pump, PPIs suppress both basal and stimulated gastric acid secretion. Acid production remains reduced until new pumps are synthesised, a process that may take up to 36 hours.35
Once absorbed, onset of action is about 90 minutes.36 While PPIs have a short elimination half-life (1–2 hours), their pharmacodynamic effect to reliably maintain intragastric pH >4 can last 15 to 21 hours, due to their irreversible covalent bonding to the proton pump. This provides symptom relief for 24 hours or longer, after 1 to 3 days of treatment.37,38
The delay between PPI initiation and inhibition of acid secretion reaching steady state means that patients with frequent reflux symptoms may benefit from concomitant therapy with an agent that provides short-term relief, such as an antacid-alginate, while waiting for the PPI to take effect. Additionally, by co-localising to the postprandial acid pocket to create a mechanical barrier that suppresses gastric reflux at the LOS, and binding bile salts to contribute to the mucoprotective effect, antacid-alginates may be considered as adjunctive therapy in some patients with breakthrough symptoms, and in PPI deprescribing to help manage symptom rebound.39,40
OTC PPIs are indicated for the initial relief of reflux and GORD as:
In contrast, aligned with the Therapeutic Guidelines (TG), pharmacists approved for expanded scope can prescribe 4 weeks of ‘standard dose’ PPI (esomeprazole 20 mg, lansoprazole 30 mg, omeprazole 20 mg, pantoprazole 40 mg or rabeprazole 20 mg), 0.5–1 hour before a meal.17
In RCTs, 2–4-weeks of PPIs at doses approved for OTC use are usually effective in the management of those with frequent reflux symptoms.37,42,43 A non-response to a short course of acid-suppressive therapy warrants prompt medical consultation, as it can suggest other diagnoses.
Globally, guideline differences in pharmacist empirical adult management of reflux or GORD largely stem from regulatory limitations, differences in definition of symptom severity and frequency, and whether used for initial or maintenance therapy.
TG recommendations include OTC and prescription options from all four reflux medicines classes. For mild, intermittent symptoms (specified as 1–2 episodes weekly), options are an antacid-alginate, combined magnesium-aluminium antacid or H2RA (all when necessary), or standard dose PPI once daily.17
For frequent or severe typical GORD symptoms, TG recommend a 4–8 week initial course of standard dose PPI; then when symptom control is adequate, step down to maintenance therapy (second daily or half the daily dose to therapy only on days when symptoms arise).17
Unfortunately, TG does not provide guidance when reflux (not GORD) symptoms are more frequent or severe. The Australasian College of Pharmacy guidelines attempt a more nuanced, pharmacist-focused approach to initial management (Figure 1).14
[caption id="attachment_31817" align="alignnone" width="2453"] Sources: Australasian College of Pharmacy. Management of reflux: a guideline for pharmacists. Queensland Health. Queensland Community Pharmacy Gastro-oesophageal Reflux and Gastro-oesophageal Reflux Disease – Clinical Practice Guideline. NSW Health. NSW Pharmacist Practice Standards for gastro-oesophageal reflux and gastro-oesophageal reflux disease.[/caption]
Sources: Australasian College of Pharmacy. Management of reflux: a guideline for pharmacists. Queensland Health. Queensland Community Pharmacy Gastro-oesophageal Reflux and Gastro-oesophageal Reflux Disease – Clinical Practice Guideline. NSW Health. NSW Pharmacist Practice Standards for gastro-oesophageal reflux and gastro-oesophageal reflux disease.[/caption]
Irrespective of approach, there is general consensus that13,14,17-19:
Australian pharmacists are expanding their scope of practice, with full scope of practice gaining momentum. In 3 years, the vision has evolved from a 2022 Queensland Government election commitment to work with pharmacy stakeholders to design and implement a pilot of pharmacists practicing to their full scope (the Queensland Community Pharmacy Scope of Practice Pilot) to business-as-usual community pharmacy prescribing services.
Since 1 July 2025, pharmacists completing additional training and meeting the requirements of the Extended Practice Authority have been able to provide clinical advice and treatment, which may include prescribing medicines to adults (18–55 years) for a range of specified acute common conditions, in accordance with a tailored clinical practice guideline.44
NSW Health has also introduced Practice Standards (adults ≥18 to ≤50 years) for expanded pharmacist scope, informed by the Queensland Health clinical practice guidelines and NSW Health emergency care assessment and treatment protocols. It is hoped that a national approach will follow, as other states roll out extended scope services.45 Both jurisdictions list gastro-oesophageal reflux and GORD as an included condition and service, with a companion clinical practice guideline and Practice Standard, respectively.46,47
All pharmacists should take advantage of these reflux resources as they provide an evidence-informed framework for patient assessment and management, primarily aligned to the TG.16 Additional expanded scope training is beneficial to improve pharmacists’ clinical skills and confidence. However, a quality and consistent approach to clinical assessment, triage, and referral is embedded in the competency standards for all pharmacists.48
While extended scope imposes formal responsibilities for patient consultation, documentation and follow-up – pharmacists should undertake these quality care strategies. The main prescribing advantages of expanded scope is extending the initial standard dose PPI treatment period from 2–4 weeks, and, in Queensland, access to Schedule 4 H2RAs. Importantly, the Clinical Practice Guideline and Practice Standard both focus heavily on the use of non-pharmacological management options in reflux symptom management.
Pharmacists play a key role in assessing reflux symptoms through structured history-taking, identifying red flags, and selecting appropriate non-pharmacological and pharmacological options based on symptom frequency and severity. Judicious use of antacid–alginate therapy for rapid relief and adjunctive support during PPI initiation or step-down can optimise symptom control while promoting safe, evidence-based self-management.
Effective management of reflux and GORD in pharmacy practice requires a patient-centred approach that integrates lifestyle modification with tailored use of OTC and, where appropriate, expanded-scope prescribing options. By applying current guidelines and supporting timely review and referral, pharmacists can improve symptom outcomes and quality of life.
Case scenario continuedYou explain to Kurt that his lifestyle is likely contributing to his symptoms and offer targeted counselling to reduce modifiable risks. You provide an OTC PPI once daily, taken 30–60 minutes before his first daily meal for 14 days, and an antacid-alginate, to use for 1–3 days before the PPI starts to work and for breakthrough symptoms, especially at night, when needed. You make an appointment with him to return in 2 weeks to check his progress, or earlier to you or his GP if symptoms worsen. At review, Kurt says he is much improved by his medicines and is avoiding his reflux triggers, especially raising the bedhead. He is happy to step-down to use of antacid-alginate when needed, and return if his symptoms worsen in the future. |
td_module_mega_menu Object
(
[post] => WP_Post Object
(
[ID] => 31434
[post_author] => 12254
[post_date] => 2026-04-09 14:39:38
[post_date_gmt] => 2026-04-09 04:39:38
[post_content] =>
Case scenarioStavros, a 35-year-old male with focal epilepsy, presents to the pharmacy to collect his medicines. He appears tired and mentions he has been having more frequent breakthrough seizures lately. As you chat, he reveals he recently started lorazepam 1 mg twice daily for generalised anxiety disorder (GAD), prescribed by his new GP. Stavros has been missing doses of his anti-seizure medicine, carbamazepine, as he feels it worsens his drowsiness and confusion. |
 Dr Ming S Soh PhD, BPharm (Hons)[/caption]
Dr Ming S Soh PhD, BPharm (Hons)[/caption]
Epilepsy is a chronic neurological condition characterised by a persistent tendency to produce seizures.1,2 According to the International League Against Epilepsy (ILAE), epilepsy is diagnosed when an individual experiences at least two unprovoked seizures more than 24 hours apart, one unprovoked seizure with a high probability (≥60%) of recurrence, or when an epilepsy syndrome is identified.3 Seizures result from abnormal, excessive electrical discharges in the brain, and vary in type, severity and frequency.4 While most seizures are brief, lasting seconds to minutes, they can become prolonged and continuous (i.e. status epilepticus), which requires urgent, intensive medical intervention.4 Globally, epilepsy affects around 50 million people.5,6 It can affect individuals across all age groups, races and ethnicities, and can significantly disrupt quality of life.7,8 Epilepsy is often associated with neurobiological, cognitive, psychological and social sequelae.1,2
Management of epilepsy primarily involves antiseizure medicines (ASM), administered as monotherapy or polytherapy depending on the patient’s response.8,9 In some cases, surgery, dietary therapy or neurostimulation may be considered.9 Treatment complexity, adverse drug reactions and non-adherence to medicines can hinder seizure control and prevent optimal outcomes.8
Learning OutcomesAfter reading this article, pharmacists should be able to:
|
In practice, pharmacists provide therapeutic drug monitoring (hospital setting), Home Medicines Reviews (HMR), MedsChecks and patient counselling, and they support the management of common comorbidities such as depression, anxiety and cognitive impairments, which can complicate treatment.6,8,9

Globally, the pooled incidence rate of epilepsy is estimated at 50–61.4 cases per 100,000 person-years.2,10,11 However, incidence varies by region and income level. In high-income countries, rates typically range from 40–70 per 100,000 person-years, while in low- and middle-income countries, incidence can reach 100–190 per 100,000 person-years, reflecting disparities in healthcare access, diagnostic capacity and exposure to risk factors.2,10 The highest incidence of epilepsy occurs in childhood, reflecting the higher seizure susceptibility of the developing brain.12
In Australia, the lifetime risk of developing epilepsy is estimated to be up to 4%, while approximately 0.6–1% of the population is living with epilepsy at any given time.13,14 Modelled data estimates that prevalence peaks in males aged 60–64 (8.54 per 1,000), and in females aged 50–54 (9.57 per 1,000).15 National surveillance data indicates that prevalence generally remains high into older age groups, largely due to increased rates of stroke, neurodegenerative diseases and brain tumours.2,14,15
Disparities are pronounced among Aboriginal and Torres Strait Islander people, who experience twice the prevalence of epilepsy compared to non-Aboriginal and Torres Strait Islander people.14,16 Aboriginal and Torres Strait Islander people also face higher rates of hospitalisation, reduced access to specialist care and ASM, and significantly elevated epilepsy-related mortality.16 The disability-adjusted life years for Aboriginal and Torres Strait Islander people with epilepsy are double those of non-Aboriginal and Torres Strait Islander people.16
Epilepsy has a multifactorial aetiology. The ILAE classifies epilepsy aetiologies into six main categories: genetic, structural, infectious, metabolic, immune and unknown (see Figure 1).17,18 Genetic mutations generally affect neuronal excitability, especially ion channels, and are linked to specific epilepsy syndromes.18–20 Structural causes include brain malformations, trauma, stroke and tumours. Infections like meningitis or encephalitis can provoke seizures through inflammation. Metabolic causes arise from systemic or inherited metabolic disorders that disrupt brain chemistry or energy production. Autoimmune responses, where the body’s immune system attacks brain tissue, can also lead to seizures. Unknown causes refer to cases where no clear aetiology can be identified despite thorough investigation. Neurodegenerative causes are increasingly recognised as important contributors to epilepsy.18 Despite structured classifications, many overlap. For example, an infection such as encephalitis may lead to structural brain damage.18 Risk factors include family history, perinatal insults, head trauma, brain infections, febrile seizures and socioeconomic disparities, which can influence both the likelihood of developing epilepsy as well as its clinical progression.21

Epilepsy involves several interconnected mechanisms that predominate in different seizure types and epilepsy syndromes and are targeted by specific ASM.
Neuronal hyperexcitability is increased neuronal responsiveness due to ion-channel dysfunction, resulting in spontaneous, excessive neuronal firing. This is a key mechanism underlying all seizure types, but is not the sole pathophysiological mechanism.4,22,23
Hypersynchrony is the abnormal synchronisation of neuronal activity which facilitates seizure propagation across brain regions, particularly in generalised epilepsies such as absence and tonic-clonic seizures.1,4,12,18
Excitatory/inhibitory neurotransmitter imbalance refers to the disruption of glutamate and gamma-aminobutyric acid (GABA) neurotransmission, shifting networks toward hyperexcitability and is central to both focal and generalised epilepsy.4,17
Neuroinflammation refers to the immune activation and cytokine release (e.g. IL-1 beta, TNF-alpha) that can enhance excitability, and are implicated in structural focal epilepsies, post-stroke epilepsy and autoimmune epilepsies. Anti-inflammatory and immunomodulatory therapies may be used in selected cases.4,24
mTOR pathway dysregulation is the abnormal mTOR signalling that alters synaptic plasticity, contributing to epileptogenesis, most notably in tuberous sclerosis complex–associated epilepsy, where mTOR inhibitors such as everolimus are used as targeted therapy.18
Oxidative stress is the excess reactive oxygen species that can damage neurons and lower seizure thresholds, contributing to seizures in metabolic and neurodegenerative epilepsies.24
Mitochondrial dysfunction refers to impaired energy metabolism, which reduces neuronal resilience and is characteristic of mitochondrial epilepsies and some childhood epileptic encephalopathies.24
Synaptic reorganisation refers to structural changes, including aberrant axon sprouting and new excitatory circuit formation, and are commonly seen in temporal lobe epilepsy following brain injury or prolonged seizures.4
Recognising underlying epileptogenic mechanisms aids interpretation of seizure type/classification and guides appropriate ASM selection.
Focal seizures (formerly known as partial)
Focal seizures originate in a specific area of one cerebral hemisphere. Depending on the brain area affected, they may present with localised motor activity like rhythmic jerking or muscle stiffening, sensory changes such as tingling or visual distortions, or autonomic signs, including flushing and gastrointestinal discomfort.12 Awareness may or may not be impaired during these seizures.25 Some focal seizures begin with an aura, such as déjà vu or intense emotions, during which a person remains alert. First-line treatment includes carbamazepine, lamotrigine or levetiracetam, where selection is guided by contraindications and other patient-specific factors.26
Generalised seizures
Generalised seizures involve widespread electrical activity across both cerebral hemispheres and typically include tonic-clonic, absence, myoclonic and atonic seizures.12,25
Tonic-clonic seizures (formerly known as grand mal)
Tonic-clonic seizures are characterised by a sudden loss of consciousness, muscle stiffening followed by rhythmic jerking, and a post-seizure phase with confusion, agitation or fatigue. They can sometimes be fatal due to associated complications. Some tonic-clonic seizures begin as focal seizures with preserved awareness, but progress to loss of consciousness as they generalise across both hemispheres of the brain.12 First-line treatment is sodium valproate, unless contraindicated or otherwise inappropriate for the patient (e.g. individuals of childbearing potential).26
Absence seizures (formerly known as petit mal)
Absence seizures are typically characterised by brief episodes of staring or unresponsiveness, and are often unnoticed or mistaken for daydreaming.12,25 First-line treatment includes ethosuximide or sodium valproate where selection is guided by contraindications and other patient-specific factors.26
Myoclonic seizures
Myoclonic seizures present as sudden and brief muscle jerks.12,25 First-line treatment is sodium valproate unless contraindicated or otherwise inappropriate (e.g. individuals of childbearing potential).26
Atonic seizures
Atonic seizures result in a sudden loss of muscle tone, leading to falls.12,25
For further information, see Australian Medicines Handbook: Epilepsy.26
Diagnosis of epilepsy involves detailed clinical history, neurological examination and supportive investigations.12,17 Electroencephalogram (EEG) is central for detecting epileptiform discharges, while neuroimaging techniques such as computed tomography (CT) and magnetic resonance imaging (MRI) can help identify structural causes.12 In complex cases, additional tools including positron emission tomography (PET)/single photon emission computed tomography (SPECT), magnetoencephalography (MEG) and genetic testing can support diagnosis and guide treatment. The advanced neuroimaging modalities are especially useful for presurgical evaluations in specialised centres.12 Neuropsychological assessments can also provide insight into cognitive impacts.1 ILAE classification recommends a structured, three-level diagnostic approach, identifying seizure type, epilepsy type and specific epilepsy syndrome, while integrating aetiology at each stage to inform treatment decisions.3 Syncope, psychogenic non-epileptic seizures, transient ischaemic attacks, migraine auras and sleep disorders can mimic epileptic seizures but differ in aetiology and treatment.12,27
Prognosis varies widely, depending on seizure type, aetiology, age of onset and treatment response.12 Many patients can achieve seizure control with ASM, and some enter long-term remission. However, around one-third of patients develop drug-resistant epilepsy, unresponsive to two or more ASM.12 Early age of onset, especially in infancy, and epilepsy with structural lesions such as hippocampal sclerosis, are often linked to poorer outcomes.28,29 Sudden unexpected death in epilepsy remains a serious risk,30 particularly in patients with uncontrolled generalised tonic-clonic seizures and early-onset syndromes such as Dravet syndrome (severe infantile-onset developmental epileptic encephalopathy), which has a 3–5-fold higher mortality risk than the general epilepsy population.31 Early diagnosis and individualised treatment strategies improve outcomes.
The primary aim is to achieve seizure freedom with minimal side effects, improving quality of life. Treatment recommendations follow a stepwise approach, guided by seizure classification, epilepsy type, underlying aetiology, side effect profile, cost, potential for measuring serum drug concentration, pharmacokinetics and patient-specific factors such as age, pregnancy, medicine interactions and comorbidities.17,26,32,33
Initial management involves monotherapy with an appropriate ASM. Dose is generally started low and titrated slowly to achieve seizure control. If control is not achieved with maximally tolerated doses of a single agent, a second agent may be added. The second agent may be considered for monotherapy if the patient and prescriber are willing to gradually withdraw the first agent. If two agents do not promote seizure control, the second agent may be withdrawn gradually and replaced with a third.34
Treatment with an ASM should never be ceased abruptly.34
Key treatment considerations
For further information, see Therapeutic Guidelines: Neurology.34
Evidence is limited for cannabis-derived products. Cannabidiol is approved for specific syndromes but requires specialist oversight.33,34
Non-pharmacological management includes surgery following EEG/MRI evaluation, vagus nerve and deep brain stimulation, ketogenic diet and gene therapy. Yoga, meditation and acupuncture have limited evidence for use.17,31–35
One of the most common challenges during epilepsy treatment is adherence. Missed doses or abrupt discontinuation can trigger recurrent seizures which significantly increase mortality risk.33 Medicine interactions are another significant concern, particularly with older ASM such as phenytoin and carbamazepine, which are enzyme inducers and can alter the efficacy of other medicines.33 Patients should be informed that some medicines can lower their seizure threshold. These include theophylline, pethidine, certain antidepressants (uncommon), and some antipsychotics. Some important medicine interactions are shown in Table 1.

Treatment complications range from mild to severe. Common side effects include fatigue, dizziness, cognitive issues such as memory or attention difficulties, and abdominal upset including pain, nausea and vomiting.33
More serious but less frequent reactions include liver and renal toxicity, bone marrow suppression and skin reactions.26 Long-term use of certain ASM including carbamazepine and phenytoin may also reduce bone density, increasing fracture risk.12,33 Additionally, some ASM including levetiracetam and topiramate can worsen mood disorders, contributing to anxiety and depression.26,33
Age also influences management.32,33 Children often require weight-based dosing and closer monitoring due to variable metabolism, while older adults are more susceptible to adverse effects and medicine interactions due to comorbidities and declining organ function.34
To manage these risks, routine monitoring, including liver and renal function tests, therapeutic drug monitoring, bone health assessments and mental health screening, is essential.
Epilepsy treatment requires regular follow-up to assess seizure control, treatment efficacy and adverse effects. Monitoring for drugs with narrow therapeutic index should also be considered. Treatment plans should be reviewed annually, or sooner, to discuss any changes in seizure occurrence.
Patients in Australia can access further advice and support through multiple channels, including:
Epilepsy Action Australia – education, telehealth and resources
Epilepsy Foundation – seizure management and support services
National Epilepsy Support Service/Epilepsy Smart Australia – 1300 761 487 Monday to Friday, from 9 am to 5 pm (AEST).
Pharmacists support epilepsy management across both community and hospital settings. Hospital pharmacists contribute by implementing therapeutic drug monitoring, especially for ASM with narrow therapeutic windows, collaborating with multidisciplinary teams to optimise treatment plans, monitoring for adverse drug reactions, and managing complex cases involving polytherapy or comorbidities. Community pharmacists can provide ongoing support and medicines counselling, and conduct MedsChecks, detect side effects early, and address mental health concerns. Together, pharmacists provide personalised care that can help improve seizure control, enhance treatment safety and reduce mortality.
With appropriate treatment, many people with epilepsy can achieve good seizure control and lead full, active lives. However, outcomes depend on early intervention, consistent follow-up, monitoring and personalised care. Pharmacist involvement helps reduce complications, improves medicine adherence and supports overall wellbeing.
Case scenario continuedYou recognise that emotional stress, non-adherence and adverse effects are likely precipitating Stavros’ seizures. Concomitant use of lorazepam and carbamazepine contributes to additive CNS depression, causing sedation. As Stavros prefers pharmacological treatment for GAD, you call his GP and suggest sertraline 50 mg daily, noting that carbamazepine may reduce sertraline concentrations via enzyme induction, and clinical response should be monitored. You recommend ceasing lorazepam and encourage cognitive behavioural therapy. The GP reviews Stavros and he returns with a cessation plan for lorazepam and a prescription for sertraline. You counsel on treatment expectations and the importance of adherence. A month later, Stavros returns, feeling alert with no recent seizures. |

DR Ming S Soh PhD, BPharm (Hons) is a Research Fellow at the Florey Institute of Neuroscience and Mental Health, University of Melbourne, with expertise in preclinical models of genetic epilepsy and therapeutic development.
Fisher RS, van Emde Boas W, Blume W, et al. Epileptic seizures and epilepsy: definitions proposed by the International League Against Epilepsy (ILAE) and the International Bureau for Epilepsy (IBE). Epilepsia. 2005;46(4):470–472.
Beghi E. The epidemiology of epilepsy. Neuroepidemiology. 2020;54(2):185–191.
Scheffer IE, Berkovic S, Capovilla G, et al. ILAE classification of the epilepsies: position paper of the ILAE commission for classification and terminology. Epilepsia. 2017;58(4):512–521.
Scharfman HE. The neurobiology of epilepsy. Curr Neurol Neurosci Rep. 2007;7(4):348–354.
World Health Organization. Epilepsy. 2022. Available from: www.who.int/news-room/fact-sheets/detail/epilepsy
Petrides M, Peletidi A, Nena E, et al. The role of pharmacists in enhancing epilepsy care: a systematic review of community and outpatient interventions. J Pharm Policy Pract. 2025;18(1):2487046.
Zack MM. National and state estimates of the numbers of adults and children with active epilepsy—United States, 2015. MMWR Morb Mortal Wkly Rep. 2017;66.
Bacci JL, Zaraa S, Stergachis A, et al. Community pharmacists’ role in caring for people living with epilepsy: a scoping review. Epilepsy Behav. 2021;117:107850.
Reis TM, Campos M, Nagai MM, et al. Contributions of pharmacists in the treatment of epilepsy: a systematic review. Am J Pharm Benefits. 2016;8(3):e55–e60.
Bell GS, Sander JW. The epidemiology of epilepsy: the size of the problem. Seizure. 2001;10(4):306–316.
Fiest KM, Sauro KM, Wiebe S, et al. Prevalence and incidence of epilepsy: a systematic review and meta-analysis of international studies. Neurology. 2017;88(3):296–303.
Stafstrom CE, Carmant L. Seizures and epilepsy: an overview for neuroscientists. Cold Spring Harb Perspect Med. 2015;5(6):a022426.
Mohanannair Geethadevi G, Chen Z, et al. Impact of epilepsy on productivity and quality of life: the Australian epilepsy project. Neurology. 2025;105(5):e214011.
Australian Institute of Health and Welfare. Epilepsy in Australia. Canberra: AIHW; 2022. Available from: www.aihw.gov.au/reports/chronic-disease/epilepsy-in-australia/contents/about
Foster E, Chen Z, Zomer E, et al. The costs of epilepsy in Australia: a productivity-based analysis. Neurology. 2020;95(24):e3221–e3231.
Keenan NF, Aitchison SG, Jetté N, et al. Epilepsy in the Indigenous peoples in Canada, Australia, New Zealand, and the USA: a systematic scoping review. Lancet Glob Health. 2025;13(4):e656–e668.
Vera-González A. Pathophysiological mechanisms underlying the etiologies of seizures and epilepsy. In: Czuczwar SJ, editor. Epilepsy. Brisbane: Exon Publications; 2022. p. 1–13.
Balestrini S, Arzimanoglou A, Blümcke I, et al. The aetiologies of epilepsy. Epileptic Disord. 2021;23(1):1–16.
Perucca P, Bahlo M, Berkovic SF. The genetics of epilepsy. Annu Rev Genomics Hum Genet. 2020;21(1):205–230.
Scheffer IE, Berkovic SF. The genetics of human epilepsy. Trends Pharmacol Sci. 2003;24(8):428–433.
Bhalla D, Godet B, Druet-Cabanac M, Preux P-M. Etiologies of epilepsy: a comprehensive review. Expert Rev Neurother. 2011;11(6):861–876.
Reid CA. Preface: ion channels and genetic epilepsy. J Neurochem. 2024;168(12):3829–3830.
Oyrer J, Maljevic S, Scheffer IE, et al. Ion channels in genetic epilepsy: from genes and mechanisms to disease-targeted therapies. Pharmacol Rev. 2018;70(1):142–173.
Fabisiak T, Patel M. Crosstalk between neuroinflammation and oxidative stress in epilepsy. Front Cell Dev Biol. 2022;10:976953.
Fisher RS, Cross JH, French JA, et al. Operational classification of seizure types by the International League Against Epilepsy: position paper of the ILAE commission for classification and terminology. Epilepsia. 2017;58(4):522–530.
Buckley N, editor. Australian medicines handbook. Adelaide: Australian Medicines Handbook Pty Ltd; 2025. Available from: https://amhonline.amh.net.au/
Cook M. Differential diagnosis of epilepsy. In: Shorvon S, Perucca E, Engel J Jr, editors. The treatment of epilepsy. 4th ed. Hoboken (NJ): John Wiley & Sons; 2015. p. 24–37.
Semah F, Picot M-C, Adam C, et al. Is the underlying cause of epilepsy a major prognostic factor for recurrence? Neurology. 1998;51(5):1256–1262.
Nickels K. Earlier is not always better: outcomes when epilepsy occurs in early life versus adolescence. Epilepsy Curr. 2020;20(1):27–29.
Devinsky O, Hesdorffer DC, Thurman DJ, et al. Sudden unexpected death in epilepsy: epidemiology, mechanisms, and prevention. Lancet Neurol. 2016;15(10):1075–1088.
Cooper MS, McIntosh A, Crompton DE, et al. Mortality in Dravet syndrome. Epilepsy Res. 2016;128:43–47.
Perucca E. General principles of medical management. In: Shorvon S, Perucca E, Engel J Jr, editors. The treatment of epilepsy. 4th ed. Hoboken (NJ): John Wiley & Sons; 2015. p. 110–123.
Perucca P, Scheffer IE, Kiley M. The management of epilepsy in children and adults. Med J Aust. 2018;208(5):226–233.
Therapeutic Guidelines Limited. eTG complete. Melbourne: Therapeutic Guidelines; 2025. Available from: www.tg.org.au
Baxendale S. Complementary and alternative treatments for epilepsy. In: Shorvon S, Perucca E, Engel J Jr, editors. The treatment of epilepsy. Hoboken (NJ): John Wiley & Sons; 2015. p. 298–310.
Johannessen SI, Johannessen Landmark C. Antiepileptic drug interactions: principles and clinical implications. Curr Neuropharmacol. 2010;8(3):254–267.
Buchanan N. Medications which may lower seizure threshold. Aust Prescr. 2001;24(1):8–9.
td_module_mega_menu Object
(
[post] => WP_Post Object
(
[ID] => 31751
[post_author] => 4862
[post_date] => 2026-04-08 11:44:33
[post_date_gmt] => 2026-04-08 01:44:33
[post_content] => New avenues for nurse prescribing have the potential to reshape frontline care. For pharmacists, it’s a live example of how scope expansion translates into real impact.
Nurse prescribing is entering a new phase in Australia, and pharmacists should be paying close attention.
While nurse practitioners and endorsed midwives have been able to prescribe Pharmaceutical Benefits Schedule (PBS) subsidised medicines since 2010, a new pathway will soon expand prescribing to a broader group of experienced nurses.
‘A lot of people wouldn't even know that nurses are already prescribing medications that are funded on the PBS,’ said Denise Lyons, President of the Australian Primary Health Care Nurses Association. ‘But there are more than 3,000 nurse practitioners and endorsed midwives in Australia now who are already prescribing.’
Under a new registration standard which came into effect in September last year, suitably qualified registered nurses will be able to prescribe medicines in Schedules 2, 3, 4 and 8 in partnership with an authorised prescriber.
‘It took until this year for universities to start the course that enables registered nurses to meet [the new standard],’ Ms Lyons said. ‘Most are 6-month postgraduate courses … So it will probably be around July 2026 that we will see the first [wave] of nurses completing the educational requirement.’
For pharmacists, this shift offers a live view of how prescribing reform is built – through legislation, governance, defined scope and, crucially, access to the PBS.
What’s changing?
The new framework is deliberately cautious. To become a designated RN prescriber, a nurse must:
‘Pharmacists and nurses are often people's first point of contact with the healthcare system, and it's really good if we can work to the top of our scope and do what we know we can do, with the right guardrails in place.' denise lyons
td_module_mega_menu Object
(
[post] => WP_Post Object
(
[ID] => 31438
[post_author] => 10832
[post_date] => 2026-04-07 14:50:18
[post_date_gmt] => 2026-04-07 04:50:18
[post_content] => Case scenario
Sarah, a 42-year-old woman, presents to the pharmacy reporting a small lump near the anus, occasional bright red bleeding on toilet paper after bowel movements, anal itching and discomfort. She mentions feeling embarrassed to seek advice and has been self-managing with sitz baths. She reports a history of chronic constipation with frequent training during defecation. She is not taking any regular medicines and requests an over-the-counter product to relieve her symptoms. You take the time to explore Sarah’s symptoms in more detail, to help identify any red flags that would require immediate referral. You ask about the duration, severity and other symptoms such as fatigue, unintentional weight loss and changes in bowel habits, all of which she denies.
Learning objectivesAfter reading this article, pharmacists should be able to:
|
Anal fissures and haemorrhoids are commonly encountered, but their epidemiology is not well documented. Haemorrhoids represent the most frequent reason for proctology consultations, followed by anal fissures.1 Haemorrhoids are estimated to affect up to 40% of adults in the general population, although many individuals are asymptomatic.2 Anal fissures occur in approximately 10–15% of the population.3

An anal fissure is a longitudinal tear in the anal canal, most often occurring in the posterior midline, just below the dentate line.4 Anal fissures are typically classified as primary or secondary, depending on the cause. Primary fissures are usually benign and caused by local trauma, such as the passage of hard stools, prolonged diarrhoea, vaginal delivery, repeated injury or anal penetration.4 Secondary fissures, which are often multiple and located off the midline, are uncommon, accounting for less than 1% of cases.5 They result from underlying conditions like previous anal surgery, inflammatory bowel disease (e.g. Crohn’s disease), colorectal malignancy, infections (e.g. HIV/AIDS), or dermatological conditions (e.g. psoriasis).6 Anal fissures are further described as acute or chronic, with chronic fissures lasting more than 6 weeks.7
[caption id="attachment_31774" align="alignright" width="200"] DR Amy Page (she/her) PhD, MClinPharm, GradDipBiostat, GCertHProfEd, GAICD, GStat, FSHPA, FPS is a consultant pharmacist, biostatistician, and the director of the Centre for Optimisation of Medicines at UWA’s School of Allied Health.[/caption]
DR Amy Page (she/her) PhD, MClinPharm, GradDipBiostat, GCertHProfEd, GAICD, GStat, FSHPA, FPS is a consultant pharmacist, biostatistician, and the director of the Centre for Optimisation of Medicines at UWA’s School of Allied Health.[/caption]
Anal fissures are often mistaken for haemorrhoids because of their overlapping symptoms. Haemorrhoids differ in that they are normal vascular cushions within the anal canal that maintain continence. Although part of normal anatomy, the term ‘haemorrhoid’ is commonly used when these vascular anal cushions, containing a rich arteriovenous network, become enlarged or displaced.8 Haemorrhoids may be classified as internal (located above the dentate line) or external (located below the dentate line). Factors such as constipation, straining during bowel movements and a low-fibre diet are commonly considered to increase the risk of haemorrhoids.9,10 However, the causal relationship between these factors and the development of haemorrhoids remains unclear. A possible genetic component that predisposes to smooth muscle, epithelial and connective tissue dysfunction, may contribute to haemorrhoids.11
It is important to note that certain medicines can contribute to or worsen constipation as an adverse effect, thereby aggravating haemorrhoids and anal fissures.9,10 Common examples include opioids, anticholinergics, and iron and calcium supplements.12 Regular medication review should be incorporated into comprehensive care to identify opportunities for deprescribing and prevent inappropriate prescribing cascades, with further guidance available in the new deprescribing guidelines.13
The symptoms of haemorrhoids may include rectal bleeding, anal pruritus, prolapse, faecal seepage and mucus discharge.7
Internal haemorrhoids are usually painless because the columnar epithelium is insensitive to touch and temperature.8 In contrast, external haemorrhoids are pain sensitive and may become thrombosed, often following straining during defecation.8
Anal fissures, by comparison, typically cause severe tearing pain that is provoked by defecation.4 A key distinguishing feature is that the pain of an anal fissure typically occurs during or immediately after defecation.

For diagnostic purposes, medical practitioners perform a physical examination with the individual lying on their side, gently parting the buttocks to inspect the posterior midline.4 Several other anorectal disorders may present with similar symptoms to anal fissures and haemorrhoids. Differential diagnoses include anal fistula and solitary rectal ulcer syndrome, which can often be distinguished based on their clinical appearance through careful physical examination. An anal fistula typically presents as a draining skin punctum.14 It is an abnormal hollow tract, most often arising from infection of an anal gland.15 Solitary rectal ulcer syndrome may present with anal pain, rectal bleeding, constipation, mucus discharge, excessive straining and a sensation of incomplete evacuation. Despite its name, the condition does not always involve an ulcer. Diagnosis requires a combination of endoscopic and histopathological findings, together with the individual’s reported symptoms.16
Haemorrhoids
[caption id="attachment_31775" align="alignright" width="200"] Hui Wen Quek (she/her) BPharm(Hons), GradCertAppPharmPrac is a pharmacist and PhD candidate at the University of Western Australia (UWA).[/caption]
Hui Wen Quek (she/her) BPharm(Hons), GradCertAppPharmPrac is a pharmacist and PhD candidate at the University of Western Australia (UWA).[/caption]
Non-pharmacological treatment options
First-line management for haemorrhoids usually involves lifestyle measures, which include ensuring adequate fluid and fibre intake, reducing prolonged time on the toilet and avoiding excessive straining, the use of sitz baths, and maintaining anal hygiene.10 In practice, dietary and lifestyle modifications are usually recommended as a conservative and preventive measure for haemorrhoids.17
Pharmacological treatment options
Over-the-counter therapies for internal haemorrhoids include ointments and suppositories that contain emollients, mild astringents, local anaesthetics and/or corticosteroids.8 These preparations temporarily relieve itching and discomfort. Prolonged use of topical corticosteroids is not recommended, as it increases the risk of local infections such as candidiasis.8 Similarly, prolonged use of anaesthetic-containing products should be avoided due to potential adverse effects, including skin sensitisation and dermatitis.8
Outpatient procedures and surgery
For individuals who do not respond to conservative measures, other treatment options for haemorrhoids include outpatient procedures and surgery.10 Outpatient procedures include rubber band ligation, infrared coagulation, bipolar probe, heater probe, sclerotherapy and cryotherapy.10 These are generally reserved for people with grade I or grade II haemorrhoids.10 Surgery can be considered for people who do not respond to or cannot tolerate outpatient procedures, as well as those with large external haemorrhoids, or combined internal and external haemorrhoids with prolapse.10
A recent meta-analysis found that surgical treatments, compared with conservative treatments, offer greater symptom relief, quicker recovery and lower recurrence rates.18 However, they do carry risks of procedure-related complications. In contrast, conservative treatments are safer and less invasive, but tend to provide slower symptom improvement and have higher recurrence rates.18 Therefore, treatment decisions should be individualised, taking into account symptom severity, individual preferences and risk tolerance.
Anal fissures
The primary aim of treatment for anal fissures is to reduce anal sphincter spasm, improve blood flow and promote healing.6 First-line management is usually non-operative and includes increasing dietary fibre, use of sitz baths and applying glyceryl trinitrate 0.2% ointment, three times daily for up to 4 weeks to relieve pain.19 Headache is a common adverse effect of glyceryl trinitrate due to systemic absorption, which could limit its use.20 Other topical agents used in clinical practice include lidocaine and hydrocortisone, although these are less effective than a combination of fibre intake and warm sitz baths.4
Botulinum toxin injections may also be considered and can achieve good healing rates.6,19 However, botulinum toxin may cause temporary reduced control of wind or bowel motions, which usually resolves as the effect of the injection wears off.4 Due to these adverse effects and the invasive nature of the procedure, botulinum toxin is generally reserved as a second-line therapy.4 If these treatment options are unsuccessful, surgical treatment may be required.6
Secondary or chronic anal fissures warrant additional investigation and multidisciplinary management is advised, especially when malignancy is suspected or confirmed.4
Pharmacists play a key role in recognising the symptoms of haemorrhoids and anal fissures, providing timely advice and suggesting a suitable course of action. Pharmacists can also educate individuals on first-line management strategies and provide lifestyle advice where appropriate to prevent recurrence. In addition, pharmacists can advocate for the safe use of over-the-counter treatments, advise on appropriate duration of use and counsel individuals on potential adverse effects.
Referral to a medical practitioner is advisable for individuals who21:
Anal fissures and haemorrhoids are common conditions encountered in primary care and can greatly impact quality of life. Pharmacists play a critical role in early management and referral, helping prevent complications and improve outcomes.
Case scenario continuedYou identify that Sarah has haemorrhoids and provide lifestyle advice, including increasing fluid and dietary fibre intake, avoiding prolonged straining during bowel movements and practising good anal hygiene. For symptomatic relief, you suggest a topical ointment containing cinchocaine 0.5% and zinc oxide 20%, to be applied as needed. You also advise her to consult her general practitioner if she experiences persistent bleeding, ongoing pain or worsening prolapse. With your support and advice, Sarah feels more confident in managing her condition effectively while reducing the risk of recurrence. |
[cpd_submit_answer_button]
Hui Wen Quek (she/her) BPharm(Hons), GradCertAppPharmPrac is a pharmacist and PhD candidate at the University of Western Australia (UWA).
DR Amy Page (she/her) PhD, MClinPharm, GradDipBiostat, GCertHProfEd, GAICD, GStat, FSHPA, FPS is a consultant pharmacist, biostatistician, and the director of the Centre for Optimisation of Medicines at UWA’s School of Allied Health.
Manisha Pillai (she/her) BPharm
Higuero T. Update on the management of anal fissure. J Visc Surg. 2015;152(2 Suppl):S37–S43.
Riss S, Weiser FA, Schwameis K, Riss T, Mittlböck M, Steiner G, et al. The prevalence of hemorrhoids in adults. Int J Colorectal Dis. 2012;27(2):215–220.
Vitton V, Bouchard D, Guingand M, Higuero T. Treatment of anal fissures: results from a national survey on French practice. Clin Res Hepatol Gastroenterol. 2022;46(4):101821.
Schlichtemeier S, Engel A. Anal fissure. Aust Prescr. 2016;39(1):14–17.
Zaghiyan KN, Fleshner P. Anal fissure. Clin Colon Rectal Surg. 2011;24(1):22–30.
Newman M, Collie M. Anal fissure: diagnosis, management, and referral in primary care. Br J Gen Pract. 2019;69(685):409–410.
Lyle V, Young C. Anal fissures: an update on treatment options. Aust J Gen Pract. 2024;53:33–35.
Therapeutic Guidelines Limited. Haemorrhoids. Melbourne: Therapeutic Guidelines Limited; 2022. Available from: https://www.tg.org.au
Lohsiriwat V. Hemorrhoids: from basic pathophysiology to clinical management. World J Gastroenterol. 2012;18(17):2009–2017.
Sandler RS, Peery AF. Rethinking what we know about hemorrhoids. Clin Gastroenterol Hepatol. 2019;17(1):8–15.
Zheng T, Ellinghaus D, Juzenas S, Cossais F, Burmeister G, Mayr G, et al. Genome-wide analysis of 944 133 individuals provides insights into the etiology of haemorrhoidal disease. Gut. 2021;70(8):1538–1549.
Therapeutic Guidelines Limited. Functional constipation in adults. Melbourne: Therapeutic Guidelines Limited; 2022. Available from: https://www.tg.org.au
Quek HW, Reus X, Lee K, Etherton-Beer C, Page A; Guideline Development Group. Deprescribing in older people: a clinical practice guideline. Perth (WA): The University of Western Australia; 2025.
Stewart D. Anal fissure: clinical manifestations, diagnosis, prevention. UpToDate. 2025. Available from: https://www.uptodate.com/contents/anal-fissure-clinical-manifestations-diagnosis-prevention
Therapeutic Guidelines Limited. Anorectal abscess and fistula. Melbourne: Therapeutic Guidelines Limited; 2022. Available from: https://www.tg.org.au
Sadeghi A, Biglari M, Forootan M, Adibi P. Solitary rectal ulcer syndrome: a narrative review. Middle East J Dig Dis. 2019;11(3):129–134.
Lohsiriwat V. Treatment of hemorrhoids: a coloproctologist's view. World J Gastroenterol. 2015;21(31):9245–9252.
Quan L, Bai X, Cheng F, Chen J, Ma H, Wang P, et al. Comparison of efficacy and safety between surgical and conservative treatments for hemorrhoids: a meta-analysis. BMC Gastroenterol. 2025;25(1):492.
Therapeutic Guidelines Limited. Anal fissure. Melbourne: Therapeutic Guidelines Limited; 2022. Available from: https://www.tg.org.au
Australian Medicines Handbook Pty Ltd. Glyceryl trinitrate (rectal). Adelaide: Australian Medicines Handbook Pty Ltd; 2025. Available from: https://amhonline.amh.net.au/
Allen S. Haemorrhoids and anal fissures. S Afr Pharm J. 2008;75(5):36.
td_module_mega_menu Object
(
[post] => WP_Post Object
(
[ID] => 31741
[post_author] => 3148
[post_date] => 2026-04-02 13:54:04
[post_date_gmt] => 2026-04-02 02:54:04
[post_content] => Community pharmacies are often the first place people turn when something feels ‘off’. But as pharmacists know, many requests for over-the-counter medicines are more complex than they may initially seem.
AP took a look at some presentations which might signal hidden red flags.
‘I just want a multivitamin’
When people present to community pharmacies, they often arrive with a solution already in mind. For example, ‘I need a strong painkiller,’ ‘I just want a multivitamin,’ or ‘What do you have for nerve pain?’
Nearly always, consistent with professional standards, these invite an open ended question to uncover the rationale for the request
 Gauri Godbole FPS[/caption]
Gauri Godbole FPS[/caption]
td_module_mega_menu Object
(
[post] => WP_Post Object
(
[ID] => 31597
[post_author] => 12189
[post_date] => 2026-04-09 16:34:44
[post_date_gmt] => 2026-04-09 06:34:44
[post_content] => Case scenario
Kurt, a 52-year-old slightly overweight electrician, comes into your pharmacy complaining of a burning feeling in his chest after meals, which has persisted for a few weeks.
[caption id="attachment_28205" align="alignright" width="388"] This CPD activity is sponsored by Reckitt. All content is the true, accurate and independent opinion of the speakers and the views expressed are entirely their own.[/caption]
You ask about other symptoms and he describes a burning sensation behind his sternum, generally mild but worse when he lies down after dinner, and the occasional sour taste in the back of the throat 1–2 times a week. He shares that he eats irregularly, is a heavy coffee drinker due to shift work and often has large late-evening meals. He finds late-night snacking, alcohol and spicy takeaways aggravate his symptoms. Kurt takes no other medicines, has no allergies and is a non-smoker.
This CPD activity is sponsored by Reckitt. All content is the true, accurate and independent opinion of the speakers and the views expressed are entirely their own.[/caption]
You ask about other symptoms and he describes a burning sensation behind his sternum, generally mild but worse when he lies down after dinner, and the occasional sour taste in the back of the throat 1–2 times a week. He shares that he eats irregularly, is a heavy coffee drinker due to shift work and often has large late-evening meals. He finds late-night snacking, alcohol and spicy takeaways aggravate his symptoms. Kurt takes no other medicines, has no allergies and is a non-smoker.
Learning objectivesAfter reading this article, pharmacists should be able to:
|
As accessible primary healthcare providers, community pharmacists are often the first healthcare provider consulted when a patient presents with symptoms of heartburn, regurgitation or reflux.
Globally, the prevalence of heartburn (retrosternal burning or pain), dyspepsia (indigestion, upper abdomen discomfort) and regurgitation (perception of flow of refluxed stomach content into the mouth or hypopharynx) in adults varies widely. Depending on the definition used and geographical location, prevalence ranges from 2.5% to 52.1%.1
While often asymptomatic, it may result in symptoms caused by refluxate flowing backwards into the oesophagus. This condition, termed gastro-oesophageal reflux (GOR, or simply reflux), is a common physiological event that can occur at any age and is generally considered non-erosive.2
Reflux aetiology involves dysfunction of the lower oesophageal sphincter (LOS) and, in some patients, distal oesophageal motility issues.2,3 A pronounced gastric acid pocket – a highly acidic, unbuffered layer of stomach acid forming on top of food in the upper stomach after a meal, acting as a reservoir that can easily backflow into the oesophagus – significantly contributes to reflux symptoms.4
Other components of gastric juice, such as bile, digestive enzymes and microbial pathogens, can also cause oesophageal symptoms.5-7 Pepsin is an endopeptidase activated at low pH to break down proteins, and can play a pivotal role in damaging the oesophageal surface.5-7 Bile is a digestive fluid designed to break down fats. When bile contacts sensitive mucosa, it can cause painful inflammation and the burning sensation associated with heartburn.5-7 The combination of acid, pepsin and bile can collectively act to assault the oesophageal mucosa.5-7
Gastro-oesophageal reflux disease (GORD) is usually associated with defective LOS function.3 It can cause reflux sufficiently severe to damage mucosa and result in oesophagitis or stricture. A meta-analysis of GORD epidemiology suggested global pooled prevalence of 13.9%.8
Laryngopharyngeal reflux (LPR) occurs when refluxate travels to the throat or higher, and may be the underlying cause of atypical reflux symptoms such as hoarse throat, changes in voice quality, excessive throat clearing and coughing.9 Suspicion of atypical reflux or ‘alarm’ symptoms requires medical referral e.g. cardiac-like chest pain, difficulty swallowing, unintended weight loss, severe abdominal pain, sudden onset reflux symptoms in a person ≥65 years, haematemesis or melaena.3
Symptoms across these conditions overlap and do not distinguish non-erosive from erosive pathology, making it difficult to assign a diagnosis without medical investigation such as endoscopy.
As there is no simple, widely accepted screening tool to differentiate between reflux and GORD, and no correlation between the symptom severity and visible changes in the oesophageal mucosa,10 pharmacists must rely on structured history-taking to identify the most appropriate management.
The use of a mnemonic e.g. Chief Complaint History Taking (LOQQSAM),11 together with targeted age or life-event-related questions, are useful to guide symptom description and triage:
The aim of empiric reflux therapy is to control symptoms and improve quality of life. This review focuses on non-pregnant adults and young people, with the management approach dependent on symptom frequency and severity. Options include risk reduction and prevention through lifestyle modification and/or pharmacological treatment.
Lifestyle modifications
The pharmacist’s role includes assessing for reflux risk factors, particularly those that are modifiable, and advising on strategies to prevent or reduce risk. Non-pharmacological strategies to reduce reflux symptom frequency or severity in this cohort include12,13:
A systematic literature review of clinical trial evidence and clinical practice guidelines14 identified that the strongest levels of evidence for non-pharmacological strategies to improve reflux symptoms were for:
Evidence was categorised with a Grade, based on the National Health and Medical Research Council Levels of Evidence for each study type.15,16
Pharmacological treatment
Most national and international guidelines recommend that a formal diagnostic workup beyond patient history is not required to diagnose the majority of patients who present with upper gastrointestinal symptoms.12,14,17-22
Patient safety is not compromised by typical symptoms of acute reflux (or GORD) being initially managed by pharmacists empirically with a short over-the-counter (OTC) trial of acid suppressant and/or alginate medicines. In Australia, the four OTC therapeutic class options available are: antacids, alginates, histamine-2 receptor antagonists (H2RAs) and proton pump inhibitors (PPIs).
Antacids
Antacid formulations contain salts of magnesium, aluminium, calcium or sodium. Antacids neutralise hydrochloric acid secreted by gastric parietal cells, leading to increased pH of gastric contents – with potency generally expressed in terms of acid neutralising capacity (ANC).23
However, onset and duration of action depend on the dose forms (powder and liquid have a higher ANC than tablets) and extent of chewing (for tablets). Onset of action is rapid, often less than a minute, with duration of action ranging from 20–60 minutes to up to 2–3 hours, based on whether taken fasting or 1 hour after a meal, respectively.18 So, antacids need to be taken frequently during the day to provide efficient relief of persistent symptoms.
Antacids provide rapid but temporary symptom relief. In 2013, the World Gastroenterology Organisation developed guidelines for the community-based management of common gastrointestinal symptoms – recommending antacids as an appropriate OTC option for infrequent, mild or moderate symptoms of heartburn.12
While updated with a global focus in 2017, antacid +/- alginate therapy remained an empirical recommendation for self-care in ‘countries with limited resources and low Helicobacter pylori prevalence’.13 They generally have a good safety profile when used below the maximum daily dose.19
Most impact on bowel function is dose-related, with calcium and magnesium salts causing diarrhoea and aluminium salts causing constipation.18 However, due to the potential for systemic adverse effects with sodium bicarbonate, regular use should be avoided in older people, those with renal or liver impairment, or those on a sodium-restricted diet.24,25
Alginates/antacid-alginate combinations
Alginates are seaweed-derived, natural polysaccharides.26 Most formulations consist of three chemical components: sodium alginate, sodium bicarbonate and calcium carbonate. The latter two constituents react with stomach acid to yield carbon dioxide and free calcium ions, respectively.27
The combination produces a low-density viscous gel (‘raft’) that floats on top of the stomach contents, forming a physical barrier to reduce or prevent the postprandial acid pocket from inducing reflux symptoms.27 Alginates can bind bile salts in refluxate, which contributes to their mucoprotective effect.28
Onset of action, symptom relief and duration of action are brand or formulation specific; alginates typically act quickly after administration, and may provide symptom relief for several hours.27–29 Onset of action has been demonstrated to be slower than antacids but faster than either a PPI or H2RA.29 This attribute is particularly useful for nocturnal symptoms and post-meal reflux.
Antacid-alginate combinations have shown to provide rapid and effective relief of mild to moderate reflux symptoms.24 In 2017, Leiman and colleagues30 published a meta-analysis of randomised-controlled trials (RCTs) for treatment of symptomatic GORD (2,095 adult patients – nine studies [N=900] comparing alginate-based products to placebo or antacid, and five studies [N=1195] to PPIs or H2RAs).
Alginate-based products increased the odds of GORD symptom resolution when compared to placebo or antacids (OR:4.42; 95% CI 2.45–7.97), with a moderate degree of heterogeneity between studies (I2=71%, P=0.001). Alginates appeared less effective than PPIs or H2RAs, but the pooled estimate was not statistically significant (OR:0.58; 95% CI 0.27–1.22).30 While, in clinical trials, alginate caused adverse effects with a comparable rate to placebo,30 they are often combined with antacids or contain excipients that may cause adverse effects.
Histamine-2 receptor antagonists (H2RAs)
By blocking the histamine receptor and histamine stimulation of parietal cell acid secretion, H2RAs competitively suppress both stimulated and basal gastric acid secretion induced by histamine.32 H2RAs have good bioavailability.33
Onset of gastric relief is about 60 minutes (decreasing stomach acid production within 1 to 3 hours), with a dose-dependent duration of action of 4 to 12 hours.34 Although less effective than PPIs, H2RAs are useful for mild and/or intermittent symptoms or when PPIs are unsuitable.17,19 Ranitidine is the only H2RA available in short-course Schedule 2 packs, while others are Schedule 4, restricting OTC H2RA options.
Proton pump inhibitors (PPIs)
PPIs are the most potent OTC acid suppression therapy available.21 They are acid-labile pro-drugs. As food can decrease bioavailability, PPIs are best taken on an empty stomach, once daily 30–60 minutes before breakfast.19 Proton pumps become activated during meals, and administration prior to food intake will enhance efficacy.18,19
PPIs are selectively taken up by gastric parietal cells and concentrated within the acidic canalicular space where they become protonated and convert to an active sulfenamide form. The activated compound then covalently binds to cysteine residues on the luminal surface of the hydrogen/potassium ATPase.
By irreversibly inactivating this proton pump, PPIs suppress both basal and stimulated gastric acid secretion. Acid production remains reduced until new pumps are synthesised, a process that may take up to 36 hours.35
Once absorbed, onset of action is about 90 minutes.36 While PPIs have a short elimination half-life (1–2 hours), their pharmacodynamic effect to reliably maintain intragastric pH >4 can last 15 to 21 hours, due to their irreversible covalent bonding to the proton pump. This provides symptom relief for 24 hours or longer, after 1 to 3 days of treatment.37,38
The delay between PPI initiation and inhibition of acid secretion reaching steady state means that patients with frequent reflux symptoms may benefit from concomitant therapy with an agent that provides short-term relief, such as an antacid-alginate, while waiting for the PPI to take effect. Additionally, by co-localising to the postprandial acid pocket to create a mechanical barrier that suppresses gastric reflux at the LOS, and binding bile salts to contribute to the mucoprotective effect, antacid-alginates may be considered as adjunctive therapy in some patients with breakthrough symptoms, and in PPI deprescribing to help manage symptom rebound.39,40
OTC PPIs are indicated for the initial relief of reflux and GORD as:
In contrast, aligned with the Therapeutic Guidelines (TG), pharmacists approved for expanded scope can prescribe 4 weeks of ‘standard dose’ PPI (esomeprazole 20 mg, lansoprazole 30 mg, omeprazole 20 mg, pantoprazole 40 mg or rabeprazole 20 mg), 0.5–1 hour before a meal.17
In RCTs, 2–4-weeks of PPIs at doses approved for OTC use are usually effective in the management of those with frequent reflux symptoms.37,42,43 A non-response to a short course of acid-suppressive therapy warrants prompt medical consultation, as it can suggest other diagnoses.
Globally, guideline differences in pharmacist empirical adult management of reflux or GORD largely stem from regulatory limitations, differences in definition of symptom severity and frequency, and whether used for initial or maintenance therapy.
TG recommendations include OTC and prescription options from all four reflux medicines classes. For mild, intermittent symptoms (specified as 1–2 episodes weekly), options are an antacid-alginate, combined magnesium-aluminium antacid or H2RA (all when necessary), or standard dose PPI once daily.17
For frequent or severe typical GORD symptoms, TG recommend a 4–8 week initial course of standard dose PPI; then when symptom control is adequate, step down to maintenance therapy (second daily or half the daily dose to therapy only on days when symptoms arise).17
Unfortunately, TG does not provide guidance when reflux (not GORD) symptoms are more frequent or severe. The Australasian College of Pharmacy guidelines attempt a more nuanced, pharmacist-focused approach to initial management (Figure 1).14
[caption id="attachment_31817" align="alignnone" width="2453"] Sources: Australasian College of Pharmacy. Management of reflux: a guideline for pharmacists. Queensland Health. Queensland Community Pharmacy Gastro-oesophageal Reflux and Gastro-oesophageal Reflux Disease – Clinical Practice Guideline. NSW Health. NSW Pharmacist Practice Standards for gastro-oesophageal reflux and gastro-oesophageal reflux disease.[/caption]
Sources: Australasian College of Pharmacy. Management of reflux: a guideline for pharmacists. Queensland Health. Queensland Community Pharmacy Gastro-oesophageal Reflux and Gastro-oesophageal Reflux Disease – Clinical Practice Guideline. NSW Health. NSW Pharmacist Practice Standards for gastro-oesophageal reflux and gastro-oesophageal reflux disease.[/caption]
Irrespective of approach, there is general consensus that13,14,17-19:
Australian pharmacists are expanding their scope of practice, with full scope of practice gaining momentum. In 3 years, the vision has evolved from a 2022 Queensland Government election commitment to work with pharmacy stakeholders to design and implement a pilot of pharmacists practicing to their full scope (the Queensland Community Pharmacy Scope of Practice Pilot) to business-as-usual community pharmacy prescribing services.
Since 1 July 2025, pharmacists completing additional training and meeting the requirements of the Extended Practice Authority have been able to provide clinical advice and treatment, which may include prescribing medicines to adults (18–55 years) for a range of specified acute common conditions, in accordance with a tailored clinical practice guideline.44
NSW Health has also introduced Practice Standards (adults ≥18 to ≤50 years) for expanded pharmacist scope, informed by the Queensland Health clinical practice guidelines and NSW Health emergency care assessment and treatment protocols. It is hoped that a national approach will follow, as other states roll out extended scope services.45 Both jurisdictions list gastro-oesophageal reflux and GORD as an included condition and service, with a companion clinical practice guideline and Practice Standard, respectively.46,47
All pharmacists should take advantage of these reflux resources as they provide an evidence-informed framework for patient assessment and management, primarily aligned to the TG.16 Additional expanded scope training is beneficial to improve pharmacists’ clinical skills and confidence. However, a quality and consistent approach to clinical assessment, triage, and referral is embedded in the competency standards for all pharmacists.48
While extended scope imposes formal responsibilities for patient consultation, documentation and follow-up – pharmacists should undertake these quality care strategies. The main prescribing advantages of expanded scope is extending the initial standard dose PPI treatment period from 2–4 weeks, and, in Queensland, access to Schedule 4 H2RAs. Importantly, the Clinical Practice Guideline and Practice Standard both focus heavily on the use of non-pharmacological management options in reflux symptom management.
Pharmacists play a key role in assessing reflux symptoms through structured history-taking, identifying red flags, and selecting appropriate non-pharmacological and pharmacological options based on symptom frequency and severity. Judicious use of antacid–alginate therapy for rapid relief and adjunctive support during PPI initiation or step-down can optimise symptom control while promoting safe, evidence-based self-management.
Effective management of reflux and GORD in pharmacy practice requires a patient-centred approach that integrates lifestyle modification with tailored use of OTC and, where appropriate, expanded-scope prescribing options. By applying current guidelines and supporting timely review and referral, pharmacists can improve symptom outcomes and quality of life.
Case scenario continuedYou explain to Kurt that his lifestyle is likely contributing to his symptoms and offer targeted counselling to reduce modifiable risks. You provide an OTC PPI once daily, taken 30–60 minutes before his first daily meal for 14 days, and an antacid-alginate, to use for 1–3 days before the PPI starts to work and for breakthrough symptoms, especially at night, when needed. You make an appointment with him to return in 2 weeks to check his progress, or earlier to you or his GP if symptoms worsen. At review, Kurt says he is much improved by his medicines and is avoiding his reflux triggers, especially raising the bedhead. He is happy to step-down to use of antacid-alginate when needed, and return if his symptoms worsen in the future. |
td_module_mega_menu Object
(
[post] => WP_Post Object
(
[ID] => 31434
[post_author] => 12254
[post_date] => 2026-04-09 14:39:38
[post_date_gmt] => 2026-04-09 04:39:38
[post_content] =>
Case scenarioStavros, a 35-year-old male with focal epilepsy, presents to the pharmacy to collect his medicines. He appears tired and mentions he has been having more frequent breakthrough seizures lately. As you chat, he reveals he recently started lorazepam 1 mg twice daily for generalised anxiety disorder (GAD), prescribed by his new GP. Stavros has been missing doses of his anti-seizure medicine, carbamazepine, as he feels it worsens his drowsiness and confusion. |
 Dr Ming S Soh PhD, BPharm (Hons)[/caption]
Dr Ming S Soh PhD, BPharm (Hons)[/caption]
Epilepsy is a chronic neurological condition characterised by a persistent tendency to produce seizures.1,2 According to the International League Against Epilepsy (ILAE), epilepsy is diagnosed when an individual experiences at least two unprovoked seizures more than 24 hours apart, one unprovoked seizure with a high probability (≥60%) of recurrence, or when an epilepsy syndrome is identified.3 Seizures result from abnormal, excessive electrical discharges in the brain, and vary in type, severity and frequency.4 While most seizures are brief, lasting seconds to minutes, they can become prolonged and continuous (i.e. status epilepticus), which requires urgent, intensive medical intervention.4 Globally, epilepsy affects around 50 million people.5,6 It can affect individuals across all age groups, races and ethnicities, and can significantly disrupt quality of life.7,8 Epilepsy is often associated with neurobiological, cognitive, psychological and social sequelae.1,2
Management of epilepsy primarily involves antiseizure medicines (ASM), administered as monotherapy or polytherapy depending on the patient’s response.8,9 In some cases, surgery, dietary therapy or neurostimulation may be considered.9 Treatment complexity, adverse drug reactions and non-adherence to medicines can hinder seizure control and prevent optimal outcomes.8
Learning OutcomesAfter reading this article, pharmacists should be able to:
|
In practice, pharmacists provide therapeutic drug monitoring (hospital setting), Home Medicines Reviews (HMR), MedsChecks and patient counselling, and they support the management of common comorbidities such as depression, anxiety and cognitive impairments, which can complicate treatment.6,8,9

Globally, the pooled incidence rate of epilepsy is estimated at 50–61.4 cases per 100,000 person-years.2,10,11 However, incidence varies by region and income level. In high-income countries, rates typically range from 40–70 per 100,000 person-years, while in low- and middle-income countries, incidence can reach 100–190 per 100,000 person-years, reflecting disparities in healthcare access, diagnostic capacity and exposure to risk factors.2,10 The highest incidence of epilepsy occurs in childhood, reflecting the higher seizure susceptibility of the developing brain.12
In Australia, the lifetime risk of developing epilepsy is estimated to be up to 4%, while approximately 0.6–1% of the population is living with epilepsy at any given time.13,14 Modelled data estimates that prevalence peaks in males aged 60–64 (8.54 per 1,000), and in females aged 50–54 (9.57 per 1,000).15 National surveillance data indicates that prevalence generally remains high into older age groups, largely due to increased rates of stroke, neurodegenerative diseases and brain tumours.2,14,15
Disparities are pronounced among Aboriginal and Torres Strait Islander people, who experience twice the prevalence of epilepsy compared to non-Aboriginal and Torres Strait Islander people.14,16 Aboriginal and Torres Strait Islander people also face higher rates of hospitalisation, reduced access to specialist care and ASM, and significantly elevated epilepsy-related mortality.16 The disability-adjusted life years for Aboriginal and Torres Strait Islander people with epilepsy are double those of non-Aboriginal and Torres Strait Islander people.16
Epilepsy has a multifactorial aetiology. The ILAE classifies epilepsy aetiologies into six main categories: genetic, structural, infectious, metabolic, immune and unknown (see Figure 1).17,18 Genetic mutations generally affect neuronal excitability, especially ion channels, and are linked to specific epilepsy syndromes.18–20 Structural causes include brain malformations, trauma, stroke and tumours. Infections like meningitis or encephalitis can provoke seizures through inflammation. Metabolic causes arise from systemic or inherited metabolic disorders that disrupt brain chemistry or energy production. Autoimmune responses, where the body’s immune system attacks brain tissue, can also lead to seizures. Unknown causes refer to cases where no clear aetiology can be identified despite thorough investigation. Neurodegenerative causes are increasingly recognised as important contributors to epilepsy.18 Despite structured classifications, many overlap. For example, an infection such as encephalitis may lead to structural brain damage.18 Risk factors include family history, perinatal insults, head trauma, brain infections, febrile seizures and socioeconomic disparities, which can influence both the likelihood of developing epilepsy as well as its clinical progression.21

Epilepsy involves several interconnected mechanisms that predominate in different seizure types and epilepsy syndromes and are targeted by specific ASM.
Neuronal hyperexcitability is increased neuronal responsiveness due to ion-channel dysfunction, resulting in spontaneous, excessive neuronal firing. This is a key mechanism underlying all seizure types, but is not the sole pathophysiological mechanism.4,22,23
Hypersynchrony is the abnormal synchronisation of neuronal activity which facilitates seizure propagation across brain regions, particularly in generalised epilepsies such as absence and tonic-clonic seizures.1,4,12,18
Excitatory/inhibitory neurotransmitter imbalance refers to the disruption of glutamate and gamma-aminobutyric acid (GABA) neurotransmission, shifting networks toward hyperexcitability and is central to both focal and generalised epilepsy.4,17
Neuroinflammation refers to the immune activation and cytokine release (e.g. IL-1 beta, TNF-alpha) that can enhance excitability, and are implicated in structural focal epilepsies, post-stroke epilepsy and autoimmune epilepsies. Anti-inflammatory and immunomodulatory therapies may be used in selected cases.4,24
mTOR pathway dysregulation is the abnormal mTOR signalling that alters synaptic plasticity, contributing to epileptogenesis, most notably in tuberous sclerosis complex–associated epilepsy, where mTOR inhibitors such as everolimus are used as targeted therapy.18
Oxidative stress is the excess reactive oxygen species that can damage neurons and lower seizure thresholds, contributing to seizures in metabolic and neurodegenerative epilepsies.24
Mitochondrial dysfunction refers to impaired energy metabolism, which reduces neuronal resilience and is characteristic of mitochondrial epilepsies and some childhood epileptic encephalopathies.24
Synaptic reorganisation refers to structural changes, including aberrant axon sprouting and new excitatory circuit formation, and are commonly seen in temporal lobe epilepsy following brain injury or prolonged seizures.4
Recognising underlying epileptogenic mechanisms aids interpretation of seizure type/classification and guides appropriate ASM selection.
Focal seizures (formerly known as partial)
Focal seizures originate in a specific area of one cerebral hemisphere. Depending on the brain area affected, they may present with localised motor activity like rhythmic jerking or muscle stiffening, sensory changes such as tingling or visual distortions, or autonomic signs, including flushing and gastrointestinal discomfort.12 Awareness may or may not be impaired during these seizures.25 Some focal seizures begin with an aura, such as déjà vu or intense emotions, during which a person remains alert. First-line treatment includes carbamazepine, lamotrigine or levetiracetam, where selection is guided by contraindications and other patient-specific factors.26
Generalised seizures
Generalised seizures involve widespread electrical activity across both cerebral hemispheres and typically include tonic-clonic, absence, myoclonic and atonic seizures.12,25
Tonic-clonic seizures (formerly known as grand mal)
Tonic-clonic seizures are characterised by a sudden loss of consciousness, muscle stiffening followed by rhythmic jerking, and a post-seizure phase with confusion, agitation or fatigue. They can sometimes be fatal due to associated complications. Some tonic-clonic seizures begin as focal seizures with preserved awareness, but progress to loss of consciousness as they generalise across both hemispheres of the brain.12 First-line treatment is sodium valproate, unless contraindicated or otherwise inappropriate for the patient (e.g. individuals of childbearing potential).26
Absence seizures (formerly known as petit mal)
Absence seizures are typically characterised by brief episodes of staring or unresponsiveness, and are often unnoticed or mistaken for daydreaming.12,25 First-line treatment includes ethosuximide or sodium valproate where selection is guided by contraindications and other patient-specific factors.26
Myoclonic seizures
Myoclonic seizures present as sudden and brief muscle jerks.12,25 First-line treatment is sodium valproate unless contraindicated or otherwise inappropriate (e.g. individuals of childbearing potential).26
Atonic seizures
Atonic seizures result in a sudden loss of muscle tone, leading to falls.12,25
For further information, see Australian Medicines Handbook: Epilepsy.26
Diagnosis of epilepsy involves detailed clinical history, neurological examination and supportive investigations.12,17 Electroencephalogram (EEG) is central for detecting epileptiform discharges, while neuroimaging techniques such as computed tomography (CT) and magnetic resonance imaging (MRI) can help identify structural causes.12 In complex cases, additional tools including positron emission tomography (PET)/single photon emission computed tomography (SPECT), magnetoencephalography (MEG) and genetic testing can support diagnosis and guide treatment. The advanced neuroimaging modalities are especially useful for presurgical evaluations in specialised centres.12 Neuropsychological assessments can also provide insight into cognitive impacts.1 ILAE classification recommends a structured, three-level diagnostic approach, identifying seizure type, epilepsy type and specific epilepsy syndrome, while integrating aetiology at each stage to inform treatment decisions.3 Syncope, psychogenic non-epileptic seizures, transient ischaemic attacks, migraine auras and sleep disorders can mimic epileptic seizures but differ in aetiology and treatment.12,27
Prognosis varies widely, depending on seizure type, aetiology, age of onset and treatment response.12 Many patients can achieve seizure control with ASM, and some enter long-term remission. However, around one-third of patients develop drug-resistant epilepsy, unresponsive to two or more ASM.12 Early age of onset, especially in infancy, and epilepsy with structural lesions such as hippocampal sclerosis, are often linked to poorer outcomes.28,29 Sudden unexpected death in epilepsy remains a serious risk,30 particularly in patients with uncontrolled generalised tonic-clonic seizures and early-onset syndromes such as Dravet syndrome (severe infantile-onset developmental epileptic encephalopathy), which has a 3–5-fold higher mortality risk than the general epilepsy population.31 Early diagnosis and individualised treatment strategies improve outcomes.
The primary aim is to achieve seizure freedom with minimal side effects, improving quality of life. Treatment recommendations follow a stepwise approach, guided by seizure classification, epilepsy type, underlying aetiology, side effect profile, cost, potential for measuring serum drug concentration, pharmacokinetics and patient-specific factors such as age, pregnancy, medicine interactions and comorbidities.17,26,32,33
Initial management involves monotherapy with an appropriate ASM. Dose is generally started low and titrated slowly to achieve seizure control. If control is not achieved with maximally tolerated doses of a single agent, a second agent may be added. The second agent may be considered for monotherapy if the patient and prescriber are willing to gradually withdraw the first agent. If two agents do not promote seizure control, the second agent may be withdrawn gradually and replaced with a third.34
Treatment with an ASM should never be ceased abruptly.34
Key treatment considerations
For further information, see Therapeutic Guidelines: Neurology.34
Evidence is limited for cannabis-derived products. Cannabidiol is approved for specific syndromes but requires specialist oversight.33,34
Non-pharmacological management includes surgery following EEG/MRI evaluation, vagus nerve and deep brain stimulation, ketogenic diet and gene therapy. Yoga, meditation and acupuncture have limited evidence for use.17,31–35
One of the most common challenges during epilepsy treatment is adherence. Missed doses or abrupt discontinuation can trigger recurrent seizures which significantly increase mortality risk.33 Medicine interactions are another significant concern, particularly with older ASM such as phenytoin and carbamazepine, which are enzyme inducers and can alter the efficacy of other medicines.33 Patients should be informed that some medicines can lower their seizure threshold. These include theophylline, pethidine, certain antidepressants (uncommon), and some antipsychotics. Some important medicine interactions are shown in Table 1.

Treatment complications range from mild to severe. Common side effects include fatigue, dizziness, cognitive issues such as memory or attention difficulties, and abdominal upset including pain, nausea and vomiting.33
More serious but less frequent reactions include liver and renal toxicity, bone marrow suppression and skin reactions.26 Long-term use of certain ASM including carbamazepine and phenytoin may also reduce bone density, increasing fracture risk.12,33 Additionally, some ASM including levetiracetam and topiramate can worsen mood disorders, contributing to anxiety and depression.26,33
Age also influences management.32,33 Children often require weight-based dosing and closer monitoring due to variable metabolism, while older adults are more susceptible to adverse effects and medicine interactions due to comorbidities and declining organ function.34
To manage these risks, routine monitoring, including liver and renal function tests, therapeutic drug monitoring, bone health assessments and mental health screening, is essential.
Epilepsy treatment requires regular follow-up to assess seizure control, treatment efficacy and adverse effects. Monitoring for drugs with narrow therapeutic index should also be considered. Treatment plans should be reviewed annually, or sooner, to discuss any changes in seizure occurrence.
Patients in Australia can access further advice and support through multiple channels, including:
Epilepsy Action Australia – education, telehealth and resources
Epilepsy Foundation – seizure management and support services
National Epilepsy Support Service/Epilepsy Smart Australia – 1300 761 487 Monday to Friday, from 9 am to 5 pm (AEST).
Pharmacists support epilepsy management across both community and hospital settings. Hospital pharmacists contribute by implementing therapeutic drug monitoring, especially for ASM with narrow therapeutic windows, collaborating with multidisciplinary teams to optimise treatment plans, monitoring for adverse drug reactions, and managing complex cases involving polytherapy or comorbidities. Community pharmacists can provide ongoing support and medicines counselling, and conduct MedsChecks, detect side effects early, and address mental health concerns. Together, pharmacists provide personalised care that can help improve seizure control, enhance treatment safety and reduce mortality.
With appropriate treatment, many people with epilepsy can achieve good seizure control and lead full, active lives. However, outcomes depend on early intervention, consistent follow-up, monitoring and personalised care. Pharmacist involvement helps reduce complications, improves medicine adherence and supports overall wellbeing.
Case scenario continuedYou recognise that emotional stress, non-adherence and adverse effects are likely precipitating Stavros’ seizures. Concomitant use of lorazepam and carbamazepine contributes to additive CNS depression, causing sedation. As Stavros prefers pharmacological treatment for GAD, you call his GP and suggest sertraline 50 mg daily, noting that carbamazepine may reduce sertraline concentrations via enzyme induction, and clinical response should be monitored. You recommend ceasing lorazepam and encourage cognitive behavioural therapy. The GP reviews Stavros and he returns with a cessation plan for lorazepam and a prescription for sertraline. You counsel on treatment expectations and the importance of adherence. A month later, Stavros returns, feeling alert with no recent seizures. |

DR Ming S Soh PhD, BPharm (Hons) is a Research Fellow at the Florey Institute of Neuroscience and Mental Health, University of Melbourne, with expertise in preclinical models of genetic epilepsy and therapeutic development.
Fisher RS, van Emde Boas W, Blume W, et al. Epileptic seizures and epilepsy: definitions proposed by the International League Against Epilepsy (ILAE) and the International Bureau for Epilepsy (IBE). Epilepsia. 2005;46(4):470–472.
Beghi E. The epidemiology of epilepsy. Neuroepidemiology. 2020;54(2):185–191.
Scheffer IE, Berkovic S, Capovilla G, et al. ILAE classification of the epilepsies: position paper of the ILAE commission for classification and terminology. Epilepsia. 2017;58(4):512–521.
Scharfman HE. The neurobiology of epilepsy. Curr Neurol Neurosci Rep. 2007;7(4):348–354.
World Health Organization. Epilepsy. 2022. Available from: www.who.int/news-room/fact-sheets/detail/epilepsy
Petrides M, Peletidi A, Nena E, et al. The role of pharmacists in enhancing epilepsy care: a systematic review of community and outpatient interventions. J Pharm Policy Pract. 2025;18(1):2487046.
Zack MM. National and state estimates of the numbers of adults and children with active epilepsy—United States, 2015. MMWR Morb Mortal Wkly Rep. 2017;66.
Bacci JL, Zaraa S, Stergachis A, et al. Community pharmacists’ role in caring for people living with epilepsy: a scoping review. Epilepsy Behav. 2021;117:107850.
Reis TM, Campos M, Nagai MM, et al. Contributions of pharmacists in the treatment of epilepsy: a systematic review. Am J Pharm Benefits. 2016;8(3):e55–e60.
Bell GS, Sander JW. The epidemiology of epilepsy: the size of the problem. Seizure. 2001;10(4):306–316.
Fiest KM, Sauro KM, Wiebe S, et al. Prevalence and incidence of epilepsy: a systematic review and meta-analysis of international studies. Neurology. 2017;88(3):296–303.
Stafstrom CE, Carmant L. Seizures and epilepsy: an overview for neuroscientists. Cold Spring Harb Perspect Med. 2015;5(6):a022426.
Mohanannair Geethadevi G, Chen Z, et al. Impact of epilepsy on productivity and quality of life: the Australian epilepsy project. Neurology. 2025;105(5):e214011.
Australian Institute of Health and Welfare. Epilepsy in Australia. Canberra: AIHW; 2022. Available from: www.aihw.gov.au/reports/chronic-disease/epilepsy-in-australia/contents/about
Foster E, Chen Z, Zomer E, et al. The costs of epilepsy in Australia: a productivity-based analysis. Neurology. 2020;95(24):e3221–e3231.
Keenan NF, Aitchison SG, Jetté N, et al. Epilepsy in the Indigenous peoples in Canada, Australia, New Zealand, and the USA: a systematic scoping review. Lancet Glob Health. 2025;13(4):e656–e668.
Vera-González A. Pathophysiological mechanisms underlying the etiologies of seizures and epilepsy. In: Czuczwar SJ, editor. Epilepsy. Brisbane: Exon Publications; 2022. p. 1–13.
Balestrini S, Arzimanoglou A, Blümcke I, et al. The aetiologies of epilepsy. Epileptic Disord. 2021;23(1):1–16.
Perucca P, Bahlo M, Berkovic SF. The genetics of epilepsy. Annu Rev Genomics Hum Genet. 2020;21(1):205–230.
Scheffer IE, Berkovic SF. The genetics of human epilepsy. Trends Pharmacol Sci. 2003;24(8):428–433.
Bhalla D, Godet B, Druet-Cabanac M, Preux P-M. Etiologies of epilepsy: a comprehensive review. Expert Rev Neurother. 2011;11(6):861–876.
Reid CA. Preface: ion channels and genetic epilepsy. J Neurochem. 2024;168(12):3829–3830.
Oyrer J, Maljevic S, Scheffer IE, et al. Ion channels in genetic epilepsy: from genes and mechanisms to disease-targeted therapies. Pharmacol Rev. 2018;70(1):142–173.
Fabisiak T, Patel M. Crosstalk between neuroinflammation and oxidative stress in epilepsy. Front Cell Dev Biol. 2022;10:976953.
Fisher RS, Cross JH, French JA, et al. Operational classification of seizure types by the International League Against Epilepsy: position paper of the ILAE commission for classification and terminology. Epilepsia. 2017;58(4):522–530.
Buckley N, editor. Australian medicines handbook. Adelaide: Australian Medicines Handbook Pty Ltd; 2025. Available from: https://amhonline.amh.net.au/
Cook M. Differential diagnosis of epilepsy. In: Shorvon S, Perucca E, Engel J Jr, editors. The treatment of epilepsy. 4th ed. Hoboken (NJ): John Wiley & Sons; 2015. p. 24–37.
Semah F, Picot M-C, Adam C, et al. Is the underlying cause of epilepsy a major prognostic factor for recurrence? Neurology. 1998;51(5):1256–1262.
Nickels K. Earlier is not always better: outcomes when epilepsy occurs in early life versus adolescence. Epilepsy Curr. 2020;20(1):27–29.
Devinsky O, Hesdorffer DC, Thurman DJ, et al. Sudden unexpected death in epilepsy: epidemiology, mechanisms, and prevention. Lancet Neurol. 2016;15(10):1075–1088.
Cooper MS, McIntosh A, Crompton DE, et al. Mortality in Dravet syndrome. Epilepsy Res. 2016;128:43–47.
Perucca E. General principles of medical management. In: Shorvon S, Perucca E, Engel J Jr, editors. The treatment of epilepsy. 4th ed. Hoboken (NJ): John Wiley & Sons; 2015. p. 110–123.
Perucca P, Scheffer IE, Kiley M. The management of epilepsy in children and adults. Med J Aust. 2018;208(5):226–233.
Therapeutic Guidelines Limited. eTG complete. Melbourne: Therapeutic Guidelines; 2025. Available from: www.tg.org.au
Baxendale S. Complementary and alternative treatments for epilepsy. In: Shorvon S, Perucca E, Engel J Jr, editors. The treatment of epilepsy. Hoboken (NJ): John Wiley & Sons; 2015. p. 298–310.
Johannessen SI, Johannessen Landmark C. Antiepileptic drug interactions: principles and clinical implications. Curr Neuropharmacol. 2010;8(3):254–267.
Buchanan N. Medications which may lower seizure threshold. Aust Prescr. 2001;24(1):8–9.
td_module_mega_menu Object
(
[post] => WP_Post Object
(
[ID] => 31751
[post_author] => 4862
[post_date] => 2026-04-08 11:44:33
[post_date_gmt] => 2026-04-08 01:44:33
[post_content] => New avenues for nurse prescribing have the potential to reshape frontline care. For pharmacists, it’s a live example of how scope expansion translates into real impact.
Nurse prescribing is entering a new phase in Australia, and pharmacists should be paying close attention.
While nurse practitioners and endorsed midwives have been able to prescribe Pharmaceutical Benefits Schedule (PBS) subsidised medicines since 2010, a new pathway will soon expand prescribing to a broader group of experienced nurses.
‘A lot of people wouldn't even know that nurses are already prescribing medications that are funded on the PBS,’ said Denise Lyons, President of the Australian Primary Health Care Nurses Association. ‘But there are more than 3,000 nurse practitioners and endorsed midwives in Australia now who are already prescribing.’
Under a new registration standard which came into effect in September last year, suitably qualified registered nurses will be able to prescribe medicines in Schedules 2, 3, 4 and 8 in partnership with an authorised prescriber.
‘It took until this year for universities to start the course that enables registered nurses to meet [the new standard],’ Ms Lyons said. ‘Most are 6-month postgraduate courses … So it will probably be around July 2026 that we will see the first [wave] of nurses completing the educational requirement.’
For pharmacists, this shift offers a live view of how prescribing reform is built – through legislation, governance, defined scope and, crucially, access to the PBS.
What’s changing?
The new framework is deliberately cautious. To become a designated RN prescriber, a nurse must:
‘Pharmacists and nurses are often people's first point of contact with the healthcare system, and it's really good if we can work to the top of our scope and do what we know we can do, with the right guardrails in place.' denise lyons
td_module_mega_menu Object
(
[post] => WP_Post Object
(
[ID] => 31438
[post_author] => 10832
[post_date] => 2026-04-07 14:50:18
[post_date_gmt] => 2026-04-07 04:50:18
[post_content] => Case scenario
Sarah, a 42-year-old woman, presents to the pharmacy reporting a small lump near the anus, occasional bright red bleeding on toilet paper after bowel movements, anal itching and discomfort. She mentions feeling embarrassed to seek advice and has been self-managing with sitz baths. She reports a history of chronic constipation with frequent training during defecation. She is not taking any regular medicines and requests an over-the-counter product to relieve her symptoms. You take the time to explore Sarah’s symptoms in more detail, to help identify any red flags that would require immediate referral. You ask about the duration, severity and other symptoms such as fatigue, unintentional weight loss and changes in bowel habits, all of which she denies.
Learning objectivesAfter reading this article, pharmacists should be able to:
|
Anal fissures and haemorrhoids are commonly encountered, but their epidemiology is not well documented. Haemorrhoids represent the most frequent reason for proctology consultations, followed by anal fissures.1 Haemorrhoids are estimated to affect up to 40% of adults in the general population, although many individuals are asymptomatic.2 Anal fissures occur in approximately 10–15% of the population.3

An anal fissure is a longitudinal tear in the anal canal, most often occurring in the posterior midline, just below the dentate line.4 Anal fissures are typically classified as primary or secondary, depending on the cause. Primary fissures are usually benign and caused by local trauma, such as the passage of hard stools, prolonged diarrhoea, vaginal delivery, repeated injury or anal penetration.4 Secondary fissures, which are often multiple and located off the midline, are uncommon, accounting for less than 1% of cases.5 They result from underlying conditions like previous anal surgery, inflammatory bowel disease (e.g. Crohn’s disease), colorectal malignancy, infections (e.g. HIV/AIDS), or dermatological conditions (e.g. psoriasis).6 Anal fissures are further described as acute or chronic, with chronic fissures lasting more than 6 weeks.7
[caption id="attachment_31774" align="alignright" width="200"] DR Amy Page (she/her) PhD, MClinPharm, GradDipBiostat, GCertHProfEd, GAICD, GStat, FSHPA, FPS is a consultant pharmacist, biostatistician, and the director of the Centre for Optimisation of Medicines at UWA’s School of Allied Health.[/caption]
DR Amy Page (she/her) PhD, MClinPharm, GradDipBiostat, GCertHProfEd, GAICD, GStat, FSHPA, FPS is a consultant pharmacist, biostatistician, and the director of the Centre for Optimisation of Medicines at UWA’s School of Allied Health.[/caption]
Anal fissures are often mistaken for haemorrhoids because of their overlapping symptoms. Haemorrhoids differ in that they are normal vascular cushions within the anal canal that maintain continence. Although part of normal anatomy, the term ‘haemorrhoid’ is commonly used when these vascular anal cushions, containing a rich arteriovenous network, become enlarged or displaced.8 Haemorrhoids may be classified as internal (located above the dentate line) or external (located below the dentate line). Factors such as constipation, straining during bowel movements and a low-fibre diet are commonly considered to increase the risk of haemorrhoids.9,10 However, the causal relationship between these factors and the development of haemorrhoids remains unclear. A possible genetic component that predisposes to smooth muscle, epithelial and connective tissue dysfunction, may contribute to haemorrhoids.11
It is important to note that certain medicines can contribute to or worsen constipation as an adverse effect, thereby aggravating haemorrhoids and anal fissures.9,10 Common examples include opioids, anticholinergics, and iron and calcium supplements.12 Regular medication review should be incorporated into comprehensive care to identify opportunities for deprescribing and prevent inappropriate prescribing cascades, with further guidance available in the new deprescribing guidelines.13
The symptoms of haemorrhoids may include rectal bleeding, anal pruritus, prolapse, faecal seepage and mucus discharge.7
Internal haemorrhoids are usually painless because the columnar epithelium is insensitive to touch and temperature.8 In contrast, external haemorrhoids are pain sensitive and may become thrombosed, often following straining during defecation.8
Anal fissures, by comparison, typically cause severe tearing pain that is provoked by defecation.4 A key distinguishing feature is that the pain of an anal fissure typically occurs during or immediately after defecation.

For diagnostic purposes, medical practitioners perform a physical examination with the individual lying on their side, gently parting the buttocks to inspect the posterior midline.4 Several other anorectal disorders may present with similar symptoms to anal fissures and haemorrhoids. Differential diagnoses include anal fistula and solitary rectal ulcer syndrome, which can often be distinguished based on their clinical appearance through careful physical examination. An anal fistula typically presents as a draining skin punctum.14 It is an abnormal hollow tract, most often arising from infection of an anal gland.15 Solitary rectal ulcer syndrome may present with anal pain, rectal bleeding, constipation, mucus discharge, excessive straining and a sensation of incomplete evacuation. Despite its name, the condition does not always involve an ulcer. Diagnosis requires a combination of endoscopic and histopathological findings, together with the individual’s reported symptoms.16
Haemorrhoids
[caption id="attachment_31775" align="alignright" width="200"] Hui Wen Quek (she/her) BPharm(Hons), GradCertAppPharmPrac is a pharmacist and PhD candidate at the University of Western Australia (UWA).[/caption]
Hui Wen Quek (she/her) BPharm(Hons), GradCertAppPharmPrac is a pharmacist and PhD candidate at the University of Western Australia (UWA).[/caption]
Non-pharmacological treatment options
First-line management for haemorrhoids usually involves lifestyle measures, which include ensuring adequate fluid and fibre intake, reducing prolonged time on the toilet and avoiding excessive straining, the use of sitz baths, and maintaining anal hygiene.10 In practice, dietary and lifestyle modifications are usually recommended as a conservative and preventive measure for haemorrhoids.17
Pharmacological treatment options
Over-the-counter therapies for internal haemorrhoids include ointments and suppositories that contain emollients, mild astringents, local anaesthetics and/or corticosteroids.8 These preparations temporarily relieve itching and discomfort. Prolonged use of topical corticosteroids is not recommended, as it increases the risk of local infections such as candidiasis.8 Similarly, prolonged use of anaesthetic-containing products should be avoided due to potential adverse effects, including skin sensitisation and dermatitis.8
Outpatient procedures and surgery
For individuals who do not respond to conservative measures, other treatment options for haemorrhoids include outpatient procedures and surgery.10 Outpatient procedures include rubber band ligation, infrared coagulation, bipolar probe, heater probe, sclerotherapy and cryotherapy.10 These are generally reserved for people with grade I or grade II haemorrhoids.10 Surgery can be considered for people who do not respond to or cannot tolerate outpatient procedures, as well as those with large external haemorrhoids, or combined internal and external haemorrhoids with prolapse.10
A recent meta-analysis found that surgical treatments, compared with conservative treatments, offer greater symptom relief, quicker recovery and lower recurrence rates.18 However, they do carry risks of procedure-related complications. In contrast, conservative treatments are safer and less invasive, but tend to provide slower symptom improvement and have higher recurrence rates.18 Therefore, treatment decisions should be individualised, taking into account symptom severity, individual preferences and risk tolerance.
Anal fissures
The primary aim of treatment for anal fissures is to reduce anal sphincter spasm, improve blood flow and promote healing.6 First-line management is usually non-operative and includes increasing dietary fibre, use of sitz baths and applying glyceryl trinitrate 0.2% ointment, three times daily for up to 4 weeks to relieve pain.19 Headache is a common adverse effect of glyceryl trinitrate due to systemic absorption, which could limit its use.20 Other topical agents used in clinical practice include lidocaine and hydrocortisone, although these are less effective than a combination of fibre intake and warm sitz baths.4
Botulinum toxin injections may also be considered and can achieve good healing rates.6,19 However, botulinum toxin may cause temporary reduced control of wind or bowel motions, which usually resolves as the effect of the injection wears off.4 Due to these adverse effects and the invasive nature of the procedure, botulinum toxin is generally reserved as a second-line therapy.4 If these treatment options are unsuccessful, surgical treatment may be required.6
Secondary or chronic anal fissures warrant additional investigation and multidisciplinary management is advised, especially when malignancy is suspected or confirmed.4
Pharmacists play a key role in recognising the symptoms of haemorrhoids and anal fissures, providing timely advice and suggesting a suitable course of action. Pharmacists can also educate individuals on first-line management strategies and provide lifestyle advice where appropriate to prevent recurrence. In addition, pharmacists can advocate for the safe use of over-the-counter treatments, advise on appropriate duration of use and counsel individuals on potential adverse effects.
Referral to a medical practitioner is advisable for individuals who21:
Anal fissures and haemorrhoids are common conditions encountered in primary care and can greatly impact quality of life. Pharmacists play a critical role in early management and referral, helping prevent complications and improve outcomes.
Case scenario continuedYou identify that Sarah has haemorrhoids and provide lifestyle advice, including increasing fluid and dietary fibre intake, avoiding prolonged straining during bowel movements and practising good anal hygiene. For symptomatic relief, you suggest a topical ointment containing cinchocaine 0.5% and zinc oxide 20%, to be applied as needed. You also advise her to consult her general practitioner if she experiences persistent bleeding, ongoing pain or worsening prolapse. With your support and advice, Sarah feels more confident in managing her condition effectively while reducing the risk of recurrence. |
[cpd_submit_answer_button]
Hui Wen Quek (she/her) BPharm(Hons), GradCertAppPharmPrac is a pharmacist and PhD candidate at the University of Western Australia (UWA).
DR Amy Page (she/her) PhD, MClinPharm, GradDipBiostat, GCertHProfEd, GAICD, GStat, FSHPA, FPS is a consultant pharmacist, biostatistician, and the director of the Centre for Optimisation of Medicines at UWA’s School of Allied Health.
Manisha Pillai (she/her) BPharm
Higuero T. Update on the management of anal fissure. J Visc Surg. 2015;152(2 Suppl):S37–S43.
Riss S, Weiser FA, Schwameis K, Riss T, Mittlböck M, Steiner G, et al. The prevalence of hemorrhoids in adults. Int J Colorectal Dis. 2012;27(2):215–220.
Vitton V, Bouchard D, Guingand M, Higuero T. Treatment of anal fissures: results from a national survey on French practice. Clin Res Hepatol Gastroenterol. 2022;46(4):101821.
Schlichtemeier S, Engel A. Anal fissure. Aust Prescr. 2016;39(1):14–17.
Zaghiyan KN, Fleshner P. Anal fissure. Clin Colon Rectal Surg. 2011;24(1):22–30.
Newman M, Collie M. Anal fissure: diagnosis, management, and referral in primary care. Br J Gen Pract. 2019;69(685):409–410.
Lyle V, Young C. Anal fissures: an update on treatment options. Aust J Gen Pract. 2024;53:33–35.
Therapeutic Guidelines Limited. Haemorrhoids. Melbourne: Therapeutic Guidelines Limited; 2022. Available from: https://www.tg.org.au
Lohsiriwat V. Hemorrhoids: from basic pathophysiology to clinical management. World J Gastroenterol. 2012;18(17):2009–2017.
Sandler RS, Peery AF. Rethinking what we know about hemorrhoids. Clin Gastroenterol Hepatol. 2019;17(1):8–15.
Zheng T, Ellinghaus D, Juzenas S, Cossais F, Burmeister G, Mayr G, et al. Genome-wide analysis of 944 133 individuals provides insights into the etiology of haemorrhoidal disease. Gut. 2021;70(8):1538–1549.
Therapeutic Guidelines Limited. Functional constipation in adults. Melbourne: Therapeutic Guidelines Limited; 2022. Available from: https://www.tg.org.au
Quek HW, Reus X, Lee K, Etherton-Beer C, Page A; Guideline Development Group. Deprescribing in older people: a clinical practice guideline. Perth (WA): The University of Western Australia; 2025.
Stewart D. Anal fissure: clinical manifestations, diagnosis, prevention. UpToDate. 2025. Available from: https://www.uptodate.com/contents/anal-fissure-clinical-manifestations-diagnosis-prevention
Therapeutic Guidelines Limited. Anorectal abscess and fistula. Melbourne: Therapeutic Guidelines Limited; 2022. Available from: https://www.tg.org.au
Sadeghi A, Biglari M, Forootan M, Adibi P. Solitary rectal ulcer syndrome: a narrative review. Middle East J Dig Dis. 2019;11(3):129–134.
Lohsiriwat V. Treatment of hemorrhoids: a coloproctologist's view. World J Gastroenterol. 2015;21(31):9245–9252.
Quan L, Bai X, Cheng F, Chen J, Ma H, Wang P, et al. Comparison of efficacy and safety between surgical and conservative treatments for hemorrhoids: a meta-analysis. BMC Gastroenterol. 2025;25(1):492.
Therapeutic Guidelines Limited. Anal fissure. Melbourne: Therapeutic Guidelines Limited; 2022. Available from: https://www.tg.org.au
Australian Medicines Handbook Pty Ltd. Glyceryl trinitrate (rectal). Adelaide: Australian Medicines Handbook Pty Ltd; 2025. Available from: https://amhonline.amh.net.au/
Allen S. Haemorrhoids and anal fissures. S Afr Pharm J. 2008;75(5):36.
td_module_mega_menu Object
(
[post] => WP_Post Object
(
[ID] => 31741
[post_author] => 3148
[post_date] => 2026-04-02 13:54:04
[post_date_gmt] => 2026-04-02 02:54:04
[post_content] => Community pharmacies are often the first place people turn when something feels ‘off’. But as pharmacists know, many requests for over-the-counter medicines are more complex than they may initially seem.
AP took a look at some presentations which might signal hidden red flags.
‘I just want a multivitamin’
When people present to community pharmacies, they often arrive with a solution already in mind. For example, ‘I need a strong painkiller,’ ‘I just want a multivitamin,’ or ‘What do you have for nerve pain?’
Nearly always, consistent with professional standards, these invite an open ended question to uncover the rationale for the request
 Gauri Godbole FPS[/caption]
Gauri Godbole FPS[/caption]
td_module_mega_menu Object
(
[post] => WP_Post Object
(
[ID] => 31597
[post_author] => 12189
[post_date] => 2026-04-09 16:34:44
[post_date_gmt] => 2026-04-09 06:34:44
[post_content] => Case scenario
Kurt, a 52-year-old slightly overweight electrician, comes into your pharmacy complaining of a burning feeling in his chest after meals, which has persisted for a few weeks.
[caption id="attachment_28205" align="alignright" width="388"] This CPD activity is sponsored by Reckitt. All content is the true, accurate and independent opinion of the speakers and the views expressed are entirely their own.[/caption]
You ask about other symptoms and he describes a burning sensation behind his sternum, generally mild but worse when he lies down after dinner, and the occasional sour taste in the back of the throat 1–2 times a week. He shares that he eats irregularly, is a heavy coffee drinker due to shift work and often has large late-evening meals. He finds late-night snacking, alcohol and spicy takeaways aggravate his symptoms. Kurt takes no other medicines, has no allergies and is a non-smoker.
This CPD activity is sponsored by Reckitt. All content is the true, accurate and independent opinion of the speakers and the views expressed are entirely their own.[/caption]
You ask about other symptoms and he describes a burning sensation behind his sternum, generally mild but worse when he lies down after dinner, and the occasional sour taste in the back of the throat 1–2 times a week. He shares that he eats irregularly, is a heavy coffee drinker due to shift work and often has large late-evening meals. He finds late-night snacking, alcohol and spicy takeaways aggravate his symptoms. Kurt takes no other medicines, has no allergies and is a non-smoker.
Learning objectivesAfter reading this article, pharmacists should be able to:
|
As accessible primary healthcare providers, community pharmacists are often the first healthcare provider consulted when a patient presents with symptoms of heartburn, regurgitation or reflux.
Globally, the prevalence of heartburn (retrosternal burning or pain), dyspepsia (indigestion, upper abdomen discomfort) and regurgitation (perception of flow of refluxed stomach content into the mouth or hypopharynx) in adults varies widely. Depending on the definition used and geographical location, prevalence ranges from 2.5% to 52.1%.1
While often asymptomatic, it may result in symptoms caused by refluxate flowing backwards into the oesophagus. This condition, termed gastro-oesophageal reflux (GOR, or simply reflux), is a common physiological event that can occur at any age and is generally considered non-erosive.2
Reflux aetiology involves dysfunction of the lower oesophageal sphincter (LOS) and, in some patients, distal oesophageal motility issues.2,3 A pronounced gastric acid pocket – a highly acidic, unbuffered layer of stomach acid forming on top of food in the upper stomach after a meal, acting as a reservoir that can easily backflow into the oesophagus – significantly contributes to reflux symptoms.4
Other components of gastric juice, such as bile, digestive enzymes and microbial pathogens, can also cause oesophageal symptoms.5-7 Pepsin is an endopeptidase activated at low pH to break down proteins, and can play a pivotal role in damaging the oesophageal surface.5-7 Bile is a digestive fluid designed to break down fats. When bile contacts sensitive mucosa, it can cause painful inflammation and the burning sensation associated with heartburn.5-7 The combination of acid, pepsin and bile can collectively act to assault the oesophageal mucosa.5-7
Gastro-oesophageal reflux disease (GORD) is usually associated with defective LOS function.3 It can cause reflux sufficiently severe to damage mucosa and result in oesophagitis or stricture. A meta-analysis of GORD epidemiology suggested global pooled prevalence of 13.9%.8
Laryngopharyngeal reflux (LPR) occurs when refluxate travels to the throat or higher, and may be the underlying cause of atypical reflux symptoms such as hoarse throat, changes in voice quality, excessive throat clearing and coughing.9 Suspicion of atypical reflux or ‘alarm’ symptoms requires medical referral e.g. cardiac-like chest pain, difficulty swallowing, unintended weight loss, severe abdominal pain, sudden onset reflux symptoms in a person ≥65 years, haematemesis or melaena.3
Symptoms across these conditions overlap and do not distinguish non-erosive from erosive pathology, making it difficult to assign a diagnosis without medical investigation such as endoscopy.
As there is no simple, widely accepted screening tool to differentiate between reflux and GORD, and no correlation between the symptom severity and visible changes in the oesophageal mucosa,10 pharmacists must rely on structured history-taking to identify the most appropriate management.
The use of a mnemonic e.g. Chief Complaint History Taking (LOQQSAM),11 together with targeted age or life-event-related questions, are useful to guide symptom description and triage:
The aim of empiric reflux therapy is to control symptoms and improve quality of life. This review focuses on non-pregnant adults and young people, with the management approach dependent on symptom frequency and severity. Options include risk reduction and prevention through lifestyle modification and/or pharmacological treatment.
Lifestyle modifications
The pharmacist’s role includes assessing for reflux risk factors, particularly those that are modifiable, and advising on strategies to prevent or reduce risk. Non-pharmacological strategies to reduce reflux symptom frequency or severity in this cohort include12,13:
A systematic literature review of clinical trial evidence and clinical practice guidelines14 identified that the strongest levels of evidence for non-pharmacological strategies to improve reflux symptoms were for:
Evidence was categorised with a Grade, based on the National Health and Medical Research Council Levels of Evidence for each study type.15,16
Pharmacological treatment
Most national and international guidelines recommend that a formal diagnostic workup beyond patient history is not required to diagnose the majority of patients who present with upper gastrointestinal symptoms.12,14,17-22
Patient safety is not compromised by typical symptoms of acute reflux (or GORD) being initially managed by pharmacists empirically with a short over-the-counter (OTC) trial of acid suppressant and/or alginate medicines. In Australia, the four OTC therapeutic class options available are: antacids, alginates, histamine-2 receptor antagonists (H2RAs) and proton pump inhibitors (PPIs).
Antacids
Antacid formulations contain salts of magnesium, aluminium, calcium or sodium. Antacids neutralise hydrochloric acid secreted by gastric parietal cells, leading to increased pH of gastric contents – with potency generally expressed in terms of acid neutralising capacity (ANC).23
However, onset and duration of action depend on the dose forms (powder and liquid have a higher ANC than tablets) and extent of chewing (for tablets). Onset of action is rapid, often less than a minute, with duration of action ranging from 20–60 minutes to up to 2–3 hours, based on whether taken fasting or 1 hour after a meal, respectively.18 So, antacids need to be taken frequently during the day to provide efficient relief of persistent symptoms.
Antacids provide rapid but temporary symptom relief. In 2013, the World Gastroenterology Organisation developed guidelines for the community-based management of common gastrointestinal symptoms – recommending antacids as an appropriate OTC option for infrequent, mild or moderate symptoms of heartburn.12
While updated with a global focus in 2017, antacid +/- alginate therapy remained an empirical recommendation for self-care in ‘countries with limited resources and low Helicobacter pylori prevalence’.13 They generally have a good safety profile when used below the maximum daily dose.19
Most impact on bowel function is dose-related, with calcium and magnesium salts causing diarrhoea and aluminium salts causing constipation.18 However, due to the potential for systemic adverse effects with sodium bicarbonate, regular use should be avoided in older people, those with renal or liver impairment, or those on a sodium-restricted diet.24,25
Alginates/antacid-alginate combinations
Alginates are seaweed-derived, natural polysaccharides.26 Most formulations consist of three chemical components: sodium alginate, sodium bicarbonate and calcium carbonate. The latter two constituents react with stomach acid to yield carbon dioxide and free calcium ions, respectively.27
The combination produces a low-density viscous gel (‘raft’) that floats on top of the stomach contents, forming a physical barrier to reduce or prevent the postprandial acid pocket from inducing reflux symptoms.27 Alginates can bind bile salts in refluxate, which contributes to their mucoprotective effect.28
Onset of action, symptom relief and duration of action are brand or formulation specific; alginates typically act quickly after administration, and may provide symptom relief for several hours.27–29 Onset of action has been demonstrated to be slower than antacids but faster than either a PPI or H2RA.29 This attribute is particularly useful for nocturnal symptoms and post-meal reflux.
Antacid-alginate combinations have shown to provide rapid and effective relief of mild to moderate reflux symptoms.24 In 2017, Leiman and colleagues30 published a meta-analysis of randomised-controlled trials (RCTs) for treatment of symptomatic GORD (2,095 adult patients – nine studies [N=900] comparing alginate-based products to placebo or antacid, and five studies [N=1195] to PPIs or H2RAs).
Alginate-based products increased the odds of GORD symptom resolution when compared to placebo or antacids (OR:4.42; 95% CI 2.45–7.97), with a moderate degree of heterogeneity between studies (I2=71%, P=0.001). Alginates appeared less effective than PPIs or H2RAs, but the pooled estimate was not statistically significant (OR:0.58; 95% CI 0.27–1.22).30 While, in clinical trials, alginate caused adverse effects with a comparable rate to placebo,30 they are often combined with antacids or contain excipients that may cause adverse effects.
Histamine-2 receptor antagonists (H2RAs)
By blocking the histamine receptor and histamine stimulation of parietal cell acid secretion, H2RAs competitively suppress both stimulated and basal gastric acid secretion induced by histamine.32 H2RAs have good bioavailability.33
Onset of gastric relief is about 60 minutes (decreasing stomach acid production within 1 to 3 hours), with a dose-dependent duration of action of 4 to 12 hours.34 Although less effective than PPIs, H2RAs are useful for mild and/or intermittent symptoms or when PPIs are unsuitable.17,19 Ranitidine is the only H2RA available in short-course Schedule 2 packs, while others are Schedule 4, restricting OTC H2RA options.
Proton pump inhibitors (PPIs)
PPIs are the most potent OTC acid suppression therapy available.21 They are acid-labile pro-drugs. As food can decrease bioavailability, PPIs are best taken on an empty stomach, once daily 30–60 minutes before breakfast.19 Proton pumps become activated during meals, and administration prior to food intake will enhance efficacy.18,19
PPIs are selectively taken up by gastric parietal cells and concentrated within the acidic canalicular space where they become protonated and convert to an active sulfenamide form. The activated compound then covalently binds to cysteine residues on the luminal surface of the hydrogen/potassium ATPase.
By irreversibly inactivating this proton pump, PPIs suppress both basal and stimulated gastric acid secretion. Acid production remains reduced until new pumps are synthesised, a process that may take up to 36 hours.35
Once absorbed, onset of action is about 90 minutes.36 While PPIs have a short elimination half-life (1–2 hours), their pharmacodynamic effect to reliably maintain intragastric pH >4 can last 15 to 21 hours, due to their irreversible covalent bonding to the proton pump. This provides symptom relief for 24 hours or longer, after 1 to 3 days of treatment.37,38
The delay between PPI initiation and inhibition of acid secretion reaching steady state means that patients with frequent reflux symptoms may benefit from concomitant therapy with an agent that provides short-term relief, such as an antacid-alginate, while waiting for the PPI to take effect. Additionally, by co-localising to the postprandial acid pocket to create a mechanical barrier that suppresses gastric reflux at the LOS, and binding bile salts to contribute to the mucoprotective effect, antacid-alginates may be considered as adjunctive therapy in some patients with breakthrough symptoms, and in PPI deprescribing to help manage symptom rebound.39,40
OTC PPIs are indicated for the initial relief of reflux and GORD as:
In contrast, aligned with the Therapeutic Guidelines (TG), pharmacists approved for expanded scope can prescribe 4 weeks of ‘standard dose’ PPI (esomeprazole 20 mg, lansoprazole 30 mg, omeprazole 20 mg, pantoprazole 40 mg or rabeprazole 20 mg), 0.5–1 hour before a meal.17
In RCTs, 2–4-weeks of PPIs at doses approved for OTC use are usually effective in the management of those with frequent reflux symptoms.37,42,43 A non-response to a short course of acid-suppressive therapy warrants prompt medical consultation, as it can suggest other diagnoses.
Globally, guideline differences in pharmacist empirical adult management of reflux or GORD largely stem from regulatory limitations, differences in definition of symptom severity and frequency, and whether used for initial or maintenance therapy.
TG recommendations include OTC and prescription options from all four reflux medicines classes. For mild, intermittent symptoms (specified as 1–2 episodes weekly), options are an antacid-alginate, combined magnesium-aluminium antacid or H2RA (all when necessary), or standard dose PPI once daily.17
For frequent or severe typical GORD symptoms, TG recommend a 4–8 week initial course of standard dose PPI; then when symptom control is adequate, step down to maintenance therapy (second daily or half the daily dose to therapy only on days when symptoms arise).17
Unfortunately, TG does not provide guidance when reflux (not GORD) symptoms are more frequent or severe. The Australasian College of Pharmacy guidelines attempt a more nuanced, pharmacist-focused approach to initial management (Figure 1).14
[caption id="attachment_31817" align="alignnone" width="2453"] Sources: Australasian College of Pharmacy. Management of reflux: a guideline for pharmacists. Queensland Health. Queensland Community Pharmacy Gastro-oesophageal Reflux and Gastro-oesophageal Reflux Disease – Clinical Practice Guideline. NSW Health. NSW Pharmacist Practice Standards for gastro-oesophageal reflux and gastro-oesophageal reflux disease.[/caption]
Sources: Australasian College of Pharmacy. Management of reflux: a guideline for pharmacists. Queensland Health. Queensland Community Pharmacy Gastro-oesophageal Reflux and Gastro-oesophageal Reflux Disease – Clinical Practice Guideline. NSW Health. NSW Pharmacist Practice Standards for gastro-oesophageal reflux and gastro-oesophageal reflux disease.[/caption]
Irrespective of approach, there is general consensus that13,14,17-19:
Australian pharmacists are expanding their scope of practice, with full scope of practice gaining momentum. In 3 years, the vision has evolved from a 2022 Queensland Government election commitment to work with pharmacy stakeholders to design and implement a pilot of pharmacists practicing to their full scope (the Queensland Community Pharmacy Scope of Practice Pilot) to business-as-usual community pharmacy prescribing services.
Since 1 July 2025, pharmacists completing additional training and meeting the requirements of the Extended Practice Authority have been able to provide clinical advice and treatment, which may include prescribing medicines to adults (18–55 years) for a range of specified acute common conditions, in accordance with a tailored clinical practice guideline.44
NSW Health has also introduced Practice Standards (adults ≥18 to ≤50 years) for expanded pharmacist scope, informed by the Queensland Health clinical practice guidelines and NSW Health emergency care assessment and treatment protocols. It is hoped that a national approach will follow, as other states roll out extended scope services.45 Both jurisdictions list gastro-oesophageal reflux and GORD as an included condition and service, with a companion clinical practice guideline and Practice Standard, respectively.46,47
All pharmacists should take advantage of these reflux resources as they provide an evidence-informed framework for patient assessment and management, primarily aligned to the TG.16 Additional expanded scope training is beneficial to improve pharmacists’ clinical skills and confidence. However, a quality and consistent approach to clinical assessment, triage, and referral is embedded in the competency standards for all pharmacists.48
While extended scope imposes formal responsibilities for patient consultation, documentation and follow-up – pharmacists should undertake these quality care strategies. The main prescribing advantages of expanded scope is extending the initial standard dose PPI treatment period from 2–4 weeks, and, in Queensland, access to Schedule 4 H2RAs. Importantly, the Clinical Practice Guideline and Practice Standard both focus heavily on the use of non-pharmacological management options in reflux symptom management.
Pharmacists play a key role in assessing reflux symptoms through structured history-taking, identifying red flags, and selecting appropriate non-pharmacological and pharmacological options based on symptom frequency and severity. Judicious use of antacid–alginate therapy for rapid relief and adjunctive support during PPI initiation or step-down can optimise symptom control while promoting safe, evidence-based self-management.
Effective management of reflux and GORD in pharmacy practice requires a patient-centred approach that integrates lifestyle modification with tailored use of OTC and, where appropriate, expanded-scope prescribing options. By applying current guidelines and supporting timely review and referral, pharmacists can improve symptom outcomes and quality of life.
Case scenario continuedYou explain to Kurt that his lifestyle is likely contributing to his symptoms and offer targeted counselling to reduce modifiable risks. You provide an OTC PPI once daily, taken 30–60 minutes before his first daily meal for 14 days, and an antacid-alginate, to use for 1–3 days before the PPI starts to work and for breakthrough symptoms, especially at night, when needed. You make an appointment with him to return in 2 weeks to check his progress, or earlier to you or his GP if symptoms worsen. At review, Kurt says he is much improved by his medicines and is avoiding his reflux triggers, especially raising the bedhead. He is happy to step-down to use of antacid-alginate when needed, and return if his symptoms worsen in the future. |
td_module_mega_menu Object
(
[post] => WP_Post Object
(
[ID] => 31434
[post_author] => 12254
[post_date] => 2026-04-09 14:39:38
[post_date_gmt] => 2026-04-09 04:39:38
[post_content] =>
Case scenarioStavros, a 35-year-old male with focal epilepsy, presents to the pharmacy to collect his medicines. He appears tired and mentions he has been having more frequent breakthrough seizures lately. As you chat, he reveals he recently started lorazepam 1 mg twice daily for generalised anxiety disorder (GAD), prescribed by his new GP. Stavros has been missing doses of his anti-seizure medicine, carbamazepine, as he feels it worsens his drowsiness and confusion. |
 Dr Ming S Soh PhD, BPharm (Hons)[/caption]
Dr Ming S Soh PhD, BPharm (Hons)[/caption]
Epilepsy is a chronic neurological condition characterised by a persistent tendency to produce seizures.1,2 According to the International League Against Epilepsy (ILAE), epilepsy is diagnosed when an individual experiences at least two unprovoked seizures more than 24 hours apart, one unprovoked seizure with a high probability (≥60%) of recurrence, or when an epilepsy syndrome is identified.3 Seizures result from abnormal, excessive electrical discharges in the brain, and vary in type, severity and frequency.4 While most seizures are brief, lasting seconds to minutes, they can become prolonged and continuous (i.e. status epilepticus), which requires urgent, intensive medical intervention.4 Globally, epilepsy affects around 50 million people.5,6 It can affect individuals across all age groups, races and ethnicities, and can significantly disrupt quality of life.7,8 Epilepsy is often associated with neurobiological, cognitive, psychological and social sequelae.1,2
Management of epilepsy primarily involves antiseizure medicines (ASM), administered as monotherapy or polytherapy depending on the patient’s response.8,9 In some cases, surgery, dietary therapy or neurostimulation may be considered.9 Treatment complexity, adverse drug reactions and non-adherence to medicines can hinder seizure control and prevent optimal outcomes.8
Learning OutcomesAfter reading this article, pharmacists should be able to:
|
In practice, pharmacists provide therapeutic drug monitoring (hospital setting), Home Medicines Reviews (HMR), MedsChecks and patient counselling, and they support the management of common comorbidities such as depression, anxiety and cognitive impairments, which can complicate treatment.6,8,9

Globally, the pooled incidence rate of epilepsy is estimated at 50–61.4 cases per 100,000 person-years.2,10,11 However, incidence varies by region and income level. In high-income countries, rates typically range from 40–70 per 100,000 person-years, while in low- and middle-income countries, incidence can reach 100–190 per 100,000 person-years, reflecting disparities in healthcare access, diagnostic capacity and exposure to risk factors.2,10 The highest incidence of epilepsy occurs in childhood, reflecting the higher seizure susceptibility of the developing brain.12
In Australia, the lifetime risk of developing epilepsy is estimated to be up to 4%, while approximately 0.6–1% of the population is living with epilepsy at any given time.13,14 Modelled data estimates that prevalence peaks in males aged 60–64 (8.54 per 1,000), and in females aged 50–54 (9.57 per 1,000).15 National surveillance data indicates that prevalence generally remains high into older age groups, largely due to increased rates of stroke, neurodegenerative diseases and brain tumours.2,14,15
Disparities are pronounced among Aboriginal and Torres Strait Islander people, who experience twice the prevalence of epilepsy compared to non-Aboriginal and Torres Strait Islander people.14,16 Aboriginal and Torres Strait Islander people also face higher rates of hospitalisation, reduced access to specialist care and ASM, and significantly elevated epilepsy-related mortality.16 The disability-adjusted life years for Aboriginal and Torres Strait Islander people with epilepsy are double those of non-Aboriginal and Torres Strait Islander people.16
Epilepsy has a multifactorial aetiology. The ILAE classifies epilepsy aetiologies into six main categories: genetic, structural, infectious, metabolic, immune and unknown (see Figure 1).17,18 Genetic mutations generally affect neuronal excitability, especially ion channels, and are linked to specific epilepsy syndromes.18–20 Structural causes include brain malformations, trauma, stroke and tumours. Infections like meningitis or encephalitis can provoke seizures through inflammation. Metabolic causes arise from systemic or inherited metabolic disorders that disrupt brain chemistry or energy production. Autoimmune responses, where the body’s immune system attacks brain tissue, can also lead to seizures. Unknown causes refer to cases where no clear aetiology can be identified despite thorough investigation. Neurodegenerative causes are increasingly recognised as important contributors to epilepsy.18 Despite structured classifications, many overlap. For example, an infection such as encephalitis may lead to structural brain damage.18 Risk factors include family history, perinatal insults, head trauma, brain infections, febrile seizures and socioeconomic disparities, which can influence both the likelihood of developing epilepsy as well as its clinical progression.21

Epilepsy involves several interconnected mechanisms that predominate in different seizure types and epilepsy syndromes and are targeted by specific ASM.
Neuronal hyperexcitability is increased neuronal responsiveness due to ion-channel dysfunction, resulting in spontaneous, excessive neuronal firing. This is a key mechanism underlying all seizure types, but is not the sole pathophysiological mechanism.4,22,23
Hypersynchrony is the abnormal synchronisation of neuronal activity which facilitates seizure propagation across brain regions, particularly in generalised epilepsies such as absence and tonic-clonic seizures.1,4,12,18
Excitatory/inhibitory neurotransmitter imbalance refers to the disruption of glutamate and gamma-aminobutyric acid (GABA) neurotransmission, shifting networks toward hyperexcitability and is central to both focal and generalised epilepsy.4,17
Neuroinflammation refers to the immune activation and cytokine release (e.g. IL-1 beta, TNF-alpha) that can enhance excitability, and are implicated in structural focal epilepsies, post-stroke epilepsy and autoimmune epilepsies. Anti-inflammatory and immunomodulatory therapies may be used in selected cases.4,24
mTOR pathway dysregulation is the abnormal mTOR signalling that alters synaptic plasticity, contributing to epileptogenesis, most notably in tuberous sclerosis complex–associated epilepsy, where mTOR inhibitors such as everolimus are used as targeted therapy.18
Oxidative stress is the excess reactive oxygen species that can damage neurons and lower seizure thresholds, contributing to seizures in metabolic and neurodegenerative epilepsies.24
Mitochondrial dysfunction refers to impaired energy metabolism, which reduces neuronal resilience and is characteristic of mitochondrial epilepsies and some childhood epileptic encephalopathies.24
Synaptic reorganisation refers to structural changes, including aberrant axon sprouting and new excitatory circuit formation, and are commonly seen in temporal lobe epilepsy following brain injury or prolonged seizures.4
Recognising underlying epileptogenic mechanisms aids interpretation of seizure type/classification and guides appropriate ASM selection.
Focal seizures (formerly known as partial)
Focal seizures originate in a specific area of one cerebral hemisphere. Depending on the brain area affected, they may present with localised motor activity like rhythmic jerking or muscle stiffening, sensory changes such as tingling or visual distortions, or autonomic signs, including flushing and gastrointestinal discomfort.12 Awareness may or may not be impaired during these seizures.25 Some focal seizures begin with an aura, such as déjà vu or intense emotions, during which a person remains alert. First-line treatment includes carbamazepine, lamotrigine or levetiracetam, where selection is guided by contraindications and other patient-specific factors.26
Generalised seizures
Generalised seizures involve widespread electrical activity across both cerebral hemispheres and typically include tonic-clonic, absence, myoclonic and atonic seizures.12,25
Tonic-clonic seizures (formerly known as grand mal)
Tonic-clonic seizures are characterised by a sudden loss of consciousness, muscle stiffening followed by rhythmic jerking, and a post-seizure phase with confusion, agitation or fatigue. They can sometimes be fatal due to associated complications. Some tonic-clonic seizures begin as focal seizures with preserved awareness, but progress to loss of consciousness as they generalise across both hemispheres of the brain.12 First-line treatment is sodium valproate, unless contraindicated or otherwise inappropriate for the patient (e.g. individuals of childbearing potential).26
Absence seizures (formerly known as petit mal)
Absence seizures are typically characterised by brief episodes of staring or unresponsiveness, and are often unnoticed or mistaken for daydreaming.12,25 First-line treatment includes ethosuximide or sodium valproate where selection is guided by contraindications and other patient-specific factors.26
Myoclonic seizures
Myoclonic seizures present as sudden and brief muscle jerks.12,25 First-line treatment is sodium valproate unless contraindicated or otherwise inappropriate (e.g. individuals of childbearing potential).26
Atonic seizures
Atonic seizures result in a sudden loss of muscle tone, leading to falls.12,25
For further information, see Australian Medicines Handbook: Epilepsy.26
Diagnosis of epilepsy involves detailed clinical history, neurological examination and supportive investigations.12,17 Electroencephalogram (EEG) is central for detecting epileptiform discharges, while neuroimaging techniques such as computed tomography (CT) and magnetic resonance imaging (MRI) can help identify structural causes.12 In complex cases, additional tools including positron emission tomography (PET)/single photon emission computed tomography (SPECT), magnetoencephalography (MEG) and genetic testing can support diagnosis and guide treatment. The advanced neuroimaging modalities are especially useful for presurgical evaluations in specialised centres.12 Neuropsychological assessments can also provide insight into cognitive impacts.1 ILAE classification recommends a structured, three-level diagnostic approach, identifying seizure type, epilepsy type and specific epilepsy syndrome, while integrating aetiology at each stage to inform treatment decisions.3 Syncope, psychogenic non-epileptic seizures, transient ischaemic attacks, migraine auras and sleep disorders can mimic epileptic seizures but differ in aetiology and treatment.12,27
Prognosis varies widely, depending on seizure type, aetiology, age of onset and treatment response.12 Many patients can achieve seizure control with ASM, and some enter long-term remission. However, around one-third of patients develop drug-resistant epilepsy, unresponsive to two or more ASM.12 Early age of onset, especially in infancy, and epilepsy with structural lesions such as hippocampal sclerosis, are often linked to poorer outcomes.28,29 Sudden unexpected death in epilepsy remains a serious risk,30 particularly in patients with uncontrolled generalised tonic-clonic seizures and early-onset syndromes such as Dravet syndrome (severe infantile-onset developmental epileptic encephalopathy), which has a 3–5-fold higher mortality risk than the general epilepsy population.31 Early diagnosis and individualised treatment strategies improve outcomes.
The primary aim is to achieve seizure freedom with minimal side effects, improving quality of life. Treatment recommendations follow a stepwise approach, guided by seizure classification, epilepsy type, underlying aetiology, side effect profile, cost, potential for measuring serum drug concentration, pharmacokinetics and patient-specific factors such as age, pregnancy, medicine interactions and comorbidities.17,26,32,33
Initial management involves monotherapy with an appropriate ASM. Dose is generally started low and titrated slowly to achieve seizure control. If control is not achieved with maximally tolerated doses of a single agent, a second agent may be added. The second agent may be considered for monotherapy if the patient and prescriber are willing to gradually withdraw the first agent. If two agents do not promote seizure control, the second agent may be withdrawn gradually and replaced with a third.34
Treatment with an ASM should never be ceased abruptly.34
Key treatment considerations
For further information, see Therapeutic Guidelines: Neurology.34
Evidence is limited for cannabis-derived products. Cannabidiol is approved for specific syndromes but requires specialist oversight.33,34
Non-pharmacological management includes surgery following EEG/MRI evaluation, vagus nerve and deep brain stimulation, ketogenic diet and gene therapy. Yoga, meditation and acupuncture have limited evidence for use.17,31–35
One of the most common challenges during epilepsy treatment is adherence. Missed doses or abrupt discontinuation can trigger recurrent seizures which significantly increase mortality risk.33 Medicine interactions are another significant concern, particularly with older ASM such as phenytoin and carbamazepine, which are enzyme inducers and can alter the efficacy of other medicines.33 Patients should be informed that some medicines can lower their seizure threshold. These include theophylline, pethidine, certain antidepressants (uncommon), and some antipsychotics. Some important medicine interactions are shown in Table 1.

Treatment complications range from mild to severe. Common side effects include fatigue, dizziness, cognitive issues such as memory or attention difficulties, and abdominal upset including pain, nausea and vomiting.33
More serious but less frequent reactions include liver and renal toxicity, bone marrow suppression and skin reactions.26 Long-term use of certain ASM including carbamazepine and phenytoin may also reduce bone density, increasing fracture risk.12,33 Additionally, some ASM including levetiracetam and topiramate can worsen mood disorders, contributing to anxiety and depression.26,33
Age also influences management.32,33 Children often require weight-based dosing and closer monitoring due to variable metabolism, while older adults are more susceptible to adverse effects and medicine interactions due to comorbidities and declining organ function.34
To manage these risks, routine monitoring, including liver and renal function tests, therapeutic drug monitoring, bone health assessments and mental health screening, is essential.
Epilepsy treatment requires regular follow-up to assess seizure control, treatment efficacy and adverse effects. Monitoring for drugs with narrow therapeutic index should also be considered. Treatment plans should be reviewed annually, or sooner, to discuss any changes in seizure occurrence.
Patients in Australia can access further advice and support through multiple channels, including:
Epilepsy Action Australia – education, telehealth and resources
Epilepsy Foundation – seizure management and support services
National Epilepsy Support Service/Epilepsy Smart Australia – 1300 761 487 Monday to Friday, from 9 am to 5 pm (AEST).
Pharmacists support epilepsy management across both community and hospital settings. Hospital pharmacists contribute by implementing therapeutic drug monitoring, especially for ASM with narrow therapeutic windows, collaborating with multidisciplinary teams to optimise treatment plans, monitoring for adverse drug reactions, and managing complex cases involving polytherapy or comorbidities. Community pharmacists can provide ongoing support and medicines counselling, and conduct MedsChecks, detect side effects early, and address mental health concerns. Together, pharmacists provide personalised care that can help improve seizure control, enhance treatment safety and reduce mortality.
With appropriate treatment, many people with epilepsy can achieve good seizure control and lead full, active lives. However, outcomes depend on early intervention, consistent follow-up, monitoring and personalised care. Pharmacist involvement helps reduce complications, improves medicine adherence and supports overall wellbeing.
Case scenario continuedYou recognise that emotional stress, non-adherence and adverse effects are likely precipitating Stavros’ seizures. Concomitant use of lorazepam and carbamazepine contributes to additive CNS depression, causing sedation. As Stavros prefers pharmacological treatment for GAD, you call his GP and suggest sertraline 50 mg daily, noting that carbamazepine may reduce sertraline concentrations via enzyme induction, and clinical response should be monitored. You recommend ceasing lorazepam and encourage cognitive behavioural therapy. The GP reviews Stavros and he returns with a cessation plan for lorazepam and a prescription for sertraline. You counsel on treatment expectations and the importance of adherence. A month later, Stavros returns, feeling alert with no recent seizures. |

DR Ming S Soh PhD, BPharm (Hons) is a Research Fellow at the Florey Institute of Neuroscience and Mental Health, University of Melbourne, with expertise in preclinical models of genetic epilepsy and therapeutic development.
Fisher RS, van Emde Boas W, Blume W, et al. Epileptic seizures and epilepsy: definitions proposed by the International League Against Epilepsy (ILAE) and the International Bureau for Epilepsy (IBE). Epilepsia. 2005;46(4):470–472.
Beghi E. The epidemiology of epilepsy. Neuroepidemiology. 2020;54(2):185–191.
Scheffer IE, Berkovic S, Capovilla G, et al. ILAE classification of the epilepsies: position paper of the ILAE commission for classification and terminology. Epilepsia. 2017;58(4):512–521.
Scharfman HE. The neurobiology of epilepsy. Curr Neurol Neurosci Rep. 2007;7(4):348–354.
World Health Organization. Epilepsy. 2022. Available from: www.who.int/news-room/fact-sheets/detail/epilepsy
Petrides M, Peletidi A, Nena E, et al. The role of pharmacists in enhancing epilepsy care: a systematic review of community and outpatient interventions. J Pharm Policy Pract. 2025;18(1):2487046.
Zack MM. National and state estimates of the numbers of adults and children with active epilepsy—United States, 2015. MMWR Morb Mortal Wkly Rep. 2017;66.
Bacci JL, Zaraa S, Stergachis A, et al. Community pharmacists’ role in caring for people living with epilepsy: a scoping review. Epilepsy Behav. 2021;117:107850.
Reis TM, Campos M, Nagai MM, et al. Contributions of pharmacists in the treatment of epilepsy: a systematic review. Am J Pharm Benefits. 2016;8(3):e55–e60.
Bell GS, Sander JW. The epidemiology of epilepsy: the size of the problem. Seizure. 2001;10(4):306–316.
Fiest KM, Sauro KM, Wiebe S, et al. Prevalence and incidence of epilepsy: a systematic review and meta-analysis of international studies. Neurology. 2017;88(3):296–303.
Stafstrom CE, Carmant L. Seizures and epilepsy: an overview for neuroscientists. Cold Spring Harb Perspect Med. 2015;5(6):a022426.
Mohanannair Geethadevi G, Chen Z, et al. Impact of epilepsy on productivity and quality of life: the Australian epilepsy project. Neurology. 2025;105(5):e214011.
Australian Institute of Health and Welfare. Epilepsy in Australia. Canberra: AIHW; 2022. Available from: www.aihw.gov.au/reports/chronic-disease/epilepsy-in-australia/contents/about
Foster E, Chen Z, Zomer E, et al. The costs of epilepsy in Australia: a productivity-based analysis. Neurology. 2020;95(24):e3221–e3231.
Keenan NF, Aitchison SG, Jetté N, et al. Epilepsy in the Indigenous peoples in Canada, Australia, New Zealand, and the USA: a systematic scoping review. Lancet Glob Health. 2025;13(4):e656–e668.
Vera-González A. Pathophysiological mechanisms underlying the etiologies of seizures and epilepsy. In: Czuczwar SJ, editor. Epilepsy. Brisbane: Exon Publications; 2022. p. 1–13.
Balestrini S, Arzimanoglou A, Blümcke I, et al. The aetiologies of epilepsy. Epileptic Disord. 2021;23(1):1–16.
Perucca P, Bahlo M, Berkovic SF. The genetics of epilepsy. Annu Rev Genomics Hum Genet. 2020;21(1):205–230.
Scheffer IE, Berkovic SF. The genetics of human epilepsy. Trends Pharmacol Sci. 2003;24(8):428–433.
Bhalla D, Godet B, Druet-Cabanac M, Preux P-M. Etiologies of epilepsy: a comprehensive review. Expert Rev Neurother. 2011;11(6):861–876.
Reid CA. Preface: ion channels and genetic epilepsy. J Neurochem. 2024;168(12):3829–3830.
Oyrer J, Maljevic S, Scheffer IE, et al. Ion channels in genetic epilepsy: from genes and mechanisms to disease-targeted therapies. Pharmacol Rev. 2018;70(1):142–173.
Fabisiak T, Patel M. Crosstalk between neuroinflammation and oxidative stress in epilepsy. Front Cell Dev Biol. 2022;10:976953.
Fisher RS, Cross JH, French JA, et al. Operational classification of seizure types by the International League Against Epilepsy: position paper of the ILAE commission for classification and terminology. Epilepsia. 2017;58(4):522–530.
Buckley N, editor. Australian medicines handbook. Adelaide: Australian Medicines Handbook Pty Ltd; 2025. Available from: https://amhonline.amh.net.au/
Cook M. Differential diagnosis of epilepsy. In: Shorvon S, Perucca E, Engel J Jr, editors. The treatment of epilepsy. 4th ed. Hoboken (NJ): John Wiley & Sons; 2015. p. 24–37.
Semah F, Picot M-C, Adam C, et al. Is the underlying cause of epilepsy a major prognostic factor for recurrence? Neurology. 1998;51(5):1256–1262.
Nickels K. Earlier is not always better: outcomes when epilepsy occurs in early life versus adolescence. Epilepsy Curr. 2020;20(1):27–29.
Devinsky O, Hesdorffer DC, Thurman DJ, et al. Sudden unexpected death in epilepsy: epidemiology, mechanisms, and prevention. Lancet Neurol. 2016;15(10):1075–1088.
Cooper MS, McIntosh A, Crompton DE, et al. Mortality in Dravet syndrome. Epilepsy Res. 2016;128:43–47.
Perucca E. General principles of medical management. In: Shorvon S, Perucca E, Engel J Jr, editors. The treatment of epilepsy. 4th ed. Hoboken (NJ): John Wiley & Sons; 2015. p. 110–123.
Perucca P, Scheffer IE, Kiley M. The management of epilepsy in children and adults. Med J Aust. 2018;208(5):226–233.
Therapeutic Guidelines Limited. eTG complete. Melbourne: Therapeutic Guidelines; 2025. Available from: www.tg.org.au
Baxendale S. Complementary and alternative treatments for epilepsy. In: Shorvon S, Perucca E, Engel J Jr, editors. The treatment of epilepsy. Hoboken (NJ): John Wiley & Sons; 2015. p. 298–310.
Johannessen SI, Johannessen Landmark C. Antiepileptic drug interactions: principles and clinical implications. Curr Neuropharmacol. 2010;8(3):254–267.
Buchanan N. Medications which may lower seizure threshold. Aust Prescr. 2001;24(1):8–9.
td_module_mega_menu Object
(
[post] => WP_Post Object
(
[ID] => 31751
[post_author] => 4862
[post_date] => 2026-04-08 11:44:33
[post_date_gmt] => 2026-04-08 01:44:33
[post_content] => New avenues for nurse prescribing have the potential to reshape frontline care. For pharmacists, it’s a live example of how scope expansion translates into real impact.
Nurse prescribing is entering a new phase in Australia, and pharmacists should be paying close attention.
While nurse practitioners and endorsed midwives have been able to prescribe Pharmaceutical Benefits Schedule (PBS) subsidised medicines since 2010, a new pathway will soon expand prescribing to a broader group of experienced nurses.
‘A lot of people wouldn't even know that nurses are already prescribing medications that are funded on the PBS,’ said Denise Lyons, President of the Australian Primary Health Care Nurses Association. ‘But there are more than 3,000 nurse practitioners and endorsed midwives in Australia now who are already prescribing.’
Under a new registration standard which came into effect in September last year, suitably qualified registered nurses will be able to prescribe medicines in Schedules 2, 3, 4 and 8 in partnership with an authorised prescriber.
‘It took until this year for universities to start the course that enables registered nurses to meet [the new standard],’ Ms Lyons said. ‘Most are 6-month postgraduate courses … So it will probably be around July 2026 that we will see the first [wave] of nurses completing the educational requirement.’
For pharmacists, this shift offers a live view of how prescribing reform is built – through legislation, governance, defined scope and, crucially, access to the PBS.
What’s changing?
The new framework is deliberately cautious. To become a designated RN prescriber, a nurse must:
‘Pharmacists and nurses are often people's first point of contact with the healthcare system, and it's really good if we can work to the top of our scope and do what we know we can do, with the right guardrails in place.' denise lyons
td_module_mega_menu Object
(
[post] => WP_Post Object
(
[ID] => 31438
[post_author] => 10832
[post_date] => 2026-04-07 14:50:18
[post_date_gmt] => 2026-04-07 04:50:18
[post_content] => Case scenario
Sarah, a 42-year-old woman, presents to the pharmacy reporting a small lump near the anus, occasional bright red bleeding on toilet paper after bowel movements, anal itching and discomfort. She mentions feeling embarrassed to seek advice and has been self-managing with sitz baths. She reports a history of chronic constipation with frequent training during defecation. She is not taking any regular medicines and requests an over-the-counter product to relieve her symptoms. You take the time to explore Sarah’s symptoms in more detail, to help identify any red flags that would require immediate referral. You ask about the duration, severity and other symptoms such as fatigue, unintentional weight loss and changes in bowel habits, all of which she denies.
Learning objectivesAfter reading this article, pharmacists should be able to:
|
Anal fissures and haemorrhoids are commonly encountered, but their epidemiology is not well documented. Haemorrhoids represent the most frequent reason for proctology consultations, followed by anal fissures.1 Haemorrhoids are estimated to affect up to 40% of adults in the general population, although many individuals are asymptomatic.2 Anal fissures occur in approximately 10–15% of the population.3

An anal fissure is a longitudinal tear in the anal canal, most often occurring in the posterior midline, just below the dentate line.4 Anal fissures are typically classified as primary or secondary, depending on the cause. Primary fissures are usually benign and caused by local trauma, such as the passage of hard stools, prolonged diarrhoea, vaginal delivery, repeated injury or anal penetration.4 Secondary fissures, which are often multiple and located off the midline, are uncommon, accounting for less than 1% of cases.5 They result from underlying conditions like previous anal surgery, inflammatory bowel disease (e.g. Crohn’s disease), colorectal malignancy, infections (e.g. HIV/AIDS), or dermatological conditions (e.g. psoriasis).6 Anal fissures are further described as acute or chronic, with chronic fissures lasting more than 6 weeks.7
[caption id="attachment_31774" align="alignright" width="200"] DR Amy Page (she/her) PhD, MClinPharm, GradDipBiostat, GCertHProfEd, GAICD, GStat, FSHPA, FPS is a consultant pharmacist, biostatistician, and the director of the Centre for Optimisation of Medicines at UWA’s School of Allied Health.[/caption]
DR Amy Page (she/her) PhD, MClinPharm, GradDipBiostat, GCertHProfEd, GAICD, GStat, FSHPA, FPS is a consultant pharmacist, biostatistician, and the director of the Centre for Optimisation of Medicines at UWA’s School of Allied Health.[/caption]
Anal fissures are often mistaken for haemorrhoids because of their overlapping symptoms. Haemorrhoids differ in that they are normal vascular cushions within the anal canal that maintain continence. Although part of normal anatomy, the term ‘haemorrhoid’ is commonly used when these vascular anal cushions, containing a rich arteriovenous network, become enlarged or displaced.8 Haemorrhoids may be classified as internal (located above the dentate line) or external (located below the dentate line). Factors such as constipation, straining during bowel movements and a low-fibre diet are commonly considered to increase the risk of haemorrhoids.9,10 However, the causal relationship between these factors and the development of haemorrhoids remains unclear. A possible genetic component that predisposes to smooth muscle, epithelial and connective tissue dysfunction, may contribute to haemorrhoids.11
It is important to note that certain medicines can contribute to or worsen constipation as an adverse effect, thereby aggravating haemorrhoids and anal fissures.9,10 Common examples include opioids, anticholinergics, and iron and calcium supplements.12 Regular medication review should be incorporated into comprehensive care to identify opportunities for deprescribing and prevent inappropriate prescribing cascades, with further guidance available in the new deprescribing guidelines.13
The symptoms of haemorrhoids may include rectal bleeding, anal pruritus, prolapse, faecal seepage and mucus discharge.7
Internal haemorrhoids are usually painless because the columnar epithelium is insensitive to touch and temperature.8 In contrast, external haemorrhoids are pain sensitive and may become thrombosed, often following straining during defecation.8
Anal fissures, by comparison, typically cause severe tearing pain that is provoked by defecation.4 A key distinguishing feature is that the pain of an anal fissure typically occurs during or immediately after defecation.

For diagnostic purposes, medical practitioners perform a physical examination with the individual lying on their side, gently parting the buttocks to inspect the posterior midline.4 Several other anorectal disorders may present with similar symptoms to anal fissures and haemorrhoids. Differential diagnoses include anal fistula and solitary rectal ulcer syndrome, which can often be distinguished based on their clinical appearance through careful physical examination. An anal fistula typically presents as a draining skin punctum.14 It is an abnormal hollow tract, most often arising from infection of an anal gland.15 Solitary rectal ulcer syndrome may present with anal pain, rectal bleeding, constipation, mucus discharge, excessive straining and a sensation of incomplete evacuation. Despite its name, the condition does not always involve an ulcer. Diagnosis requires a combination of endoscopic and histopathological findings, together with the individual’s reported symptoms.16
Haemorrhoids
[caption id="attachment_31775" align="alignright" width="200"] Hui Wen Quek (she/her) BPharm(Hons), GradCertAppPharmPrac is a pharmacist and PhD candidate at the University of Western Australia (UWA).[/caption]
Hui Wen Quek (she/her) BPharm(Hons), GradCertAppPharmPrac is a pharmacist and PhD candidate at the University of Western Australia (UWA).[/caption]
Non-pharmacological treatment options
First-line management for haemorrhoids usually involves lifestyle measures, which include ensuring adequate fluid and fibre intake, reducing prolonged time on the toilet and avoiding excessive straining, the use of sitz baths, and maintaining anal hygiene.10 In practice, dietary and lifestyle modifications are usually recommended as a conservative and preventive measure for haemorrhoids.17
Pharmacological treatment options
Over-the-counter therapies for internal haemorrhoids include ointments and suppositories that contain emollients, mild astringents, local anaesthetics and/or corticosteroids.8 These preparations temporarily relieve itching and discomfort. Prolonged use of topical corticosteroids is not recommended, as it increases the risk of local infections such as candidiasis.8 Similarly, prolonged use of anaesthetic-containing products should be avoided due to potential adverse effects, including skin sensitisation and dermatitis.8
Outpatient procedures and surgery
For individuals who do not respond to conservative measures, other treatment options for haemorrhoids include outpatient procedures and surgery.10 Outpatient procedures include rubber band ligation, infrared coagulation, bipolar probe, heater probe, sclerotherapy and cryotherapy.10 These are generally reserved for people with grade I or grade II haemorrhoids.10 Surgery can be considered for people who do not respond to or cannot tolerate outpatient procedures, as well as those with large external haemorrhoids, or combined internal and external haemorrhoids with prolapse.10
A recent meta-analysis found that surgical treatments, compared with conservative treatments, offer greater symptom relief, quicker recovery and lower recurrence rates.18 However, they do carry risks of procedure-related complications. In contrast, conservative treatments are safer and less invasive, but tend to provide slower symptom improvement and have higher recurrence rates.18 Therefore, treatment decisions should be individualised, taking into account symptom severity, individual preferences and risk tolerance.
Anal fissures
The primary aim of treatment for anal fissures is to reduce anal sphincter spasm, improve blood flow and promote healing.6 First-line management is usually non-operative and includes increasing dietary fibre, use of sitz baths and applying glyceryl trinitrate 0.2% ointment, three times daily for up to 4 weeks to relieve pain.19 Headache is a common adverse effect of glyceryl trinitrate due to systemic absorption, which could limit its use.20 Other topical agents used in clinical practice include lidocaine and hydrocortisone, although these are less effective than a combination of fibre intake and warm sitz baths.4
Botulinum toxin injections may also be considered and can achieve good healing rates.6,19 However, botulinum toxin may cause temporary reduced control of wind or bowel motions, which usually resolves as the effect of the injection wears off.4 Due to these adverse effects and the invasive nature of the procedure, botulinum toxin is generally reserved as a second-line therapy.4 If these treatment options are unsuccessful, surgical treatment may be required.6
Secondary or chronic anal fissures warrant additional investigation and multidisciplinary management is advised, especially when malignancy is suspected or confirmed.4
Pharmacists play a key role in recognising the symptoms of haemorrhoids and anal fissures, providing timely advice and suggesting a suitable course of action. Pharmacists can also educate individuals on first-line management strategies and provide lifestyle advice where appropriate to prevent recurrence. In addition, pharmacists can advocate for the safe use of over-the-counter treatments, advise on appropriate duration of use and counsel individuals on potential adverse effects.
Referral to a medical practitioner is advisable for individuals who21:
Anal fissures and haemorrhoids are common conditions encountered in primary care and can greatly impact quality of life. Pharmacists play a critical role in early management and referral, helping prevent complications and improve outcomes.
Case scenario continuedYou identify that Sarah has haemorrhoids and provide lifestyle advice, including increasing fluid and dietary fibre intake, avoiding prolonged straining during bowel movements and practising good anal hygiene. For symptomatic relief, you suggest a topical ointment containing cinchocaine 0.5% and zinc oxide 20%, to be applied as needed. You also advise her to consult her general practitioner if she experiences persistent bleeding, ongoing pain or worsening prolapse. With your support and advice, Sarah feels more confident in managing her condition effectively while reducing the risk of recurrence. |
[cpd_submit_answer_button]
Hui Wen Quek (she/her) BPharm(Hons), GradCertAppPharmPrac is a pharmacist and PhD candidate at the University of Western Australia (UWA).
DR Amy Page (she/her) PhD, MClinPharm, GradDipBiostat, GCertHProfEd, GAICD, GStat, FSHPA, FPS is a consultant pharmacist, biostatistician, and the director of the Centre for Optimisation of Medicines at UWA’s School of Allied Health.
Manisha Pillai (she/her) BPharm
Higuero T. Update on the management of anal fissure. J Visc Surg. 2015;152(2 Suppl):S37–S43.
Riss S, Weiser FA, Schwameis K, Riss T, Mittlböck M, Steiner G, et al. The prevalence of hemorrhoids in adults. Int J Colorectal Dis. 2012;27(2):215–220.
Vitton V, Bouchard D, Guingand M, Higuero T. Treatment of anal fissures: results from a national survey on French practice. Clin Res Hepatol Gastroenterol. 2022;46(4):101821.
Schlichtemeier S, Engel A. Anal fissure. Aust Prescr. 2016;39(1):14–17.
Zaghiyan KN, Fleshner P. Anal fissure. Clin Colon Rectal Surg. 2011;24(1):22–30.
Newman M, Collie M. Anal fissure: diagnosis, management, and referral in primary care. Br J Gen Pract. 2019;69(685):409–410.
Lyle V, Young C. Anal fissures: an update on treatment options. Aust J Gen Pract. 2024;53:33–35.
Therapeutic Guidelines Limited. Haemorrhoids. Melbourne: Therapeutic Guidelines Limited; 2022. Available from: https://www.tg.org.au
Lohsiriwat V. Hemorrhoids: from basic pathophysiology to clinical management. World J Gastroenterol. 2012;18(17):2009–2017.
Sandler RS, Peery AF. Rethinking what we know about hemorrhoids. Clin Gastroenterol Hepatol. 2019;17(1):8–15.
Zheng T, Ellinghaus D, Juzenas S, Cossais F, Burmeister G, Mayr G, et al. Genome-wide analysis of 944 133 individuals provides insights into the etiology of haemorrhoidal disease. Gut. 2021;70(8):1538–1549.
Therapeutic Guidelines Limited. Functional constipation in adults. Melbourne: Therapeutic Guidelines Limited; 2022. Available from: https://www.tg.org.au
Quek HW, Reus X, Lee K, Etherton-Beer C, Page A; Guideline Development Group. Deprescribing in older people: a clinical practice guideline. Perth (WA): The University of Western Australia; 2025.
Stewart D. Anal fissure: clinical manifestations, diagnosis, prevention. UpToDate. 2025. Available from: https://www.uptodate.com/contents/anal-fissure-clinical-manifestations-diagnosis-prevention
Therapeutic Guidelines Limited. Anorectal abscess and fistula. Melbourne: Therapeutic Guidelines Limited; 2022. Available from: https://www.tg.org.au
Sadeghi A, Biglari M, Forootan M, Adibi P. Solitary rectal ulcer syndrome: a narrative review. Middle East J Dig Dis. 2019;11(3):129–134.
Lohsiriwat V. Treatment of hemorrhoids: a coloproctologist's view. World J Gastroenterol. 2015;21(31):9245–9252.
Quan L, Bai X, Cheng F, Chen J, Ma H, Wang P, et al. Comparison of efficacy and safety between surgical and conservative treatments for hemorrhoids: a meta-analysis. BMC Gastroenterol. 2025;25(1):492.
Therapeutic Guidelines Limited. Anal fissure. Melbourne: Therapeutic Guidelines Limited; 2022. Available from: https://www.tg.org.au
Australian Medicines Handbook Pty Ltd. Glyceryl trinitrate (rectal). Adelaide: Australian Medicines Handbook Pty Ltd; 2025. Available from: https://amhonline.amh.net.au/
Allen S. Haemorrhoids and anal fissures. S Afr Pharm J. 2008;75(5):36.
td_module_mega_menu Object
(
[post] => WP_Post Object
(
[ID] => 31741
[post_author] => 3148
[post_date] => 2026-04-02 13:54:04
[post_date_gmt] => 2026-04-02 02:54:04
[post_content] => Community pharmacies are often the first place people turn when something feels ‘off’. But as pharmacists know, many requests for over-the-counter medicines are more complex than they may initially seem.
AP took a look at some presentations which might signal hidden red flags.
‘I just want a multivitamin’
When people present to community pharmacies, they often arrive with a solution already in mind. For example, ‘I need a strong painkiller,’ ‘I just want a multivitamin,’ or ‘What do you have for nerve pain?’
Nearly always, consistent with professional standards, these invite an open ended question to uncover the rationale for the request
 Gauri Godbole FPS[/caption]
Gauri Godbole FPS[/caption]
Get your weekly dose of the news and research you need to help advance your practice.
Protected by Google reCAPTCHA v3.

Australian Pharmacist is the official journal for Pharmaceutical Society of Australia Ltd.
