The TGA is reviewing andrographis after hundreds of serious adverse events raise safety concerns.
It’s mid April and flu season is kicking into gear. According to Roy Morgan, almost half (44.5%) of Australians over 14 years of age rely on cold and flu medicines to help manage their symptoms.
While orally ingested phenylephrine products have come under scrutiny in recent years due to efficacy concerns, an ingredient in another cold and flu product has risen to the fore – andrographis paniculata.
The herbal ingredient, found in over 100 locally available products, is unscheduled, and is widely available in pharmacies, supermarkets and health food stores.
But this could potentially change, with the Therapeutic Goods Administration (TGA) proposing swift action following a safety review linking the herb to rare but potentially fatal anaphylaxis.
What did the TGA find?
A consistent pattern of serious allergic reactions over time, including:
- 287 reports of anaphylaxis
- 1,365 adverse event reports
- One reported fatal case.
In June 2024, 37-year-old Queensland father of two, Cale Agosta, died due to a suspected fatal anaphylactic reaction after taking the andrographis-containing BioCeuticals product ArmaForce, prompting the TGA to conduct an updated review.
The reactions experienced by consumers were unpredictable, often occurring on first use or after previous tolerance, and frequently in people with no prior history of allergy or anaphylaxis.
Carmel Wells told the ABC that she experienced yellowing of her skin and eyes in 2019 after taking a supplement containing andrographis paniculata. She has since been diagnosed with drug-induced autoimmune hepatitis.
‘For me, 6 years down the track, I’m having to manage my health still … so I’m back on regular blood testing,’ she said.
Why are labels not enough?
Since 2019, the TGA has required mandatory label warnings about allergic reactions (including anaphylaxis risk) to be included on all listed medicines containing andrographis. In 2024, following the TGA’s updated review – one sponsor introduced a more prominent warning label as an additional risk mitigation measure.
However, the TGA stated that these actions have not resulted in a meaningful reduction in reported cases of anaphylaxis.
What is the TGA proposing?
The removal of andrographis from the permitted ingredients list for listed medicines.
If adopted, this would mean products could no longer be sold as ‘low-risk’ complementary medicines and would need to be reformulated or withdrawn.
And the only way a product containing andrographis paniculata could be supplied in Australia would be after quality, safety and efficacy assessment by the TGA to be a registered product carrying an AUST R number.
When is a decision expected?
The TGA has not specified a timeline for its final decision, and will consider stakeholder feedback before determining whether to remove andrographis from the list of permitted ingredients.
What should pharmacists be on the lookout for now?
Factors that could increase the chance of adverse events and/or anaphylaxis include viral infections, anti‑inflammatory use (such as NSAIDs), alcohol use or exercising around the time of exposure to the allergen.
In many patients, symptoms began in around 30 minutes, including:
- difficult or noisy breathing
- swelling of the tongue and/or throat
- wheezing or coughing
- difficulty speaking or speaking in a hoarse voice
- dizziness or collapse
- stomach pain or vomiting.
Patients who experience these symptoms after taking the supplement are advised to seek emergency care.




 This CPD activity is sponsored by Reckitt. All content is the true, accurate and independent opinion of the speakers and the views expressed are entirely their own.[/caption]
This CPD activity is sponsored by Reckitt. All content is the true, accurate and independent opinion of the speakers and the views expressed are entirely their own.[/caption]
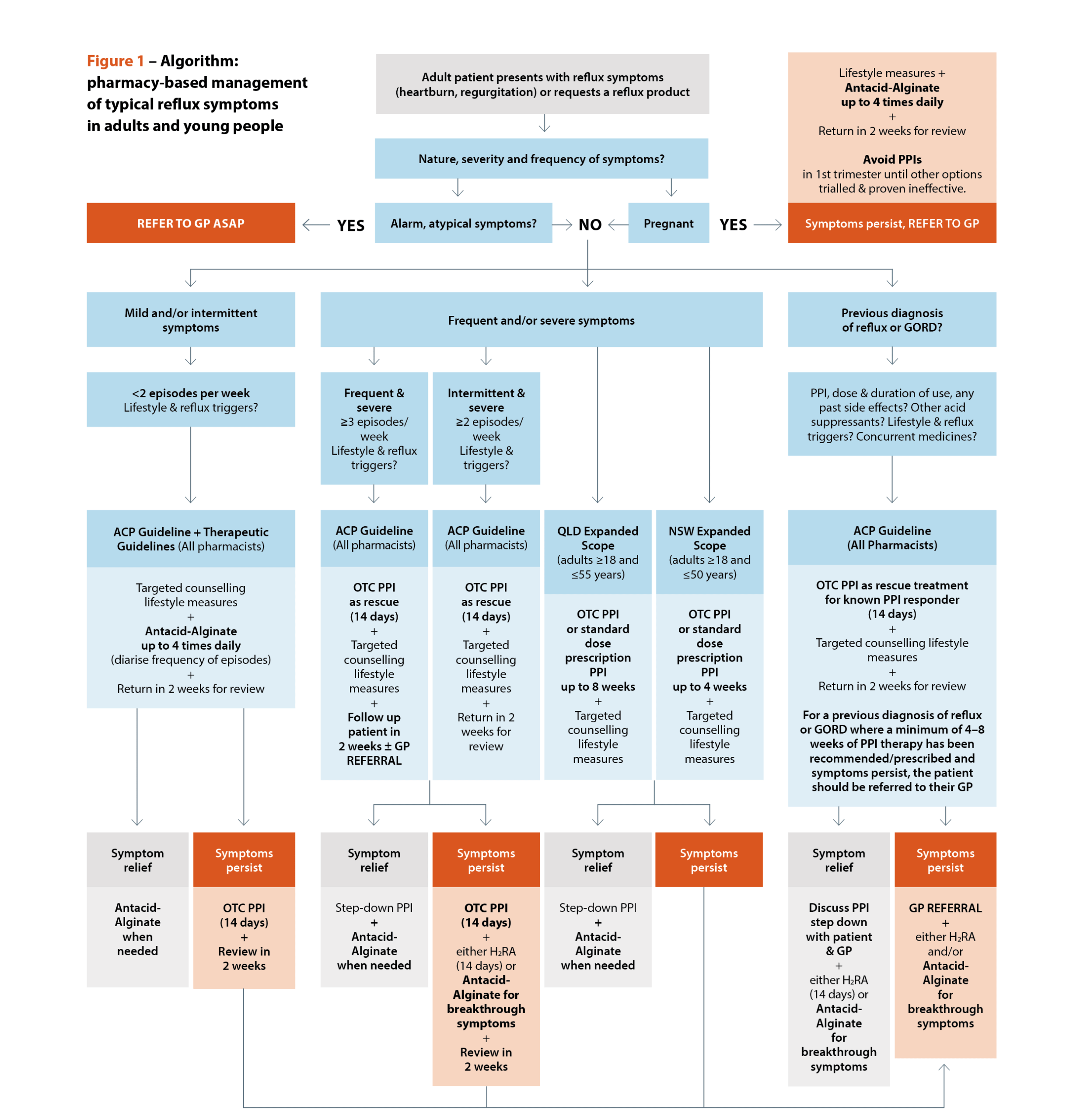 Sources: Australasian College of Pharmacy. Management of reflux: a guideline for pharmacists. Queensland Health. Queensland Community Pharmacy Gastro-oesophageal Reflux and Gastro-oesophageal Reflux Disease – Clinical Practice Guideline. NSW Health. NSW Pharmacist Practice Standards for gastro-oesophageal reflux and gastro-oesophageal reflux disease.[/caption]
Sources: Australasian College of Pharmacy. Management of reflux: a guideline for pharmacists. Queensland Health. Queensland Community Pharmacy Gastro-oesophageal Reflux and Gastro-oesophageal Reflux Disease – Clinical Practice Guideline. NSW Health. NSW Pharmacist Practice Standards for gastro-oesophageal reflux and gastro-oesophageal reflux disease.[/caption]

 Dr Ming S Soh PhD, BPharm (Hons)[/caption]
Dr Ming S Soh PhD, BPharm (Hons)[/caption]






 DR Amy Page (she/her) PhD, MClinPharm, GradDipBiostat, GCertHProfEd, GAICD, GStat, FSHPA, FPS is a consultant pharmacist, biostatistician, and the director of the Centre for Optimisation of Medicines at UWA’s School of Allied Health.[/caption]
DR Amy Page (she/her) PhD, MClinPharm, GradDipBiostat, GCertHProfEd, GAICD, GStat, FSHPA, FPS is a consultant pharmacist, biostatistician, and the director of the Centre for Optimisation of Medicines at UWA’s School of Allied Health.[/caption]

 Hui Wen Quek (she/her) BPharm(Hons), GradCertAppPharmPrac is a pharmacist and PhD candidate at the University of Western Australia (UWA).[/caption]
Hui Wen Quek (she/her) BPharm(Hons), GradCertAppPharmPrac is a pharmacist and PhD candidate at the University of Western Australia (UWA).[/caption]






