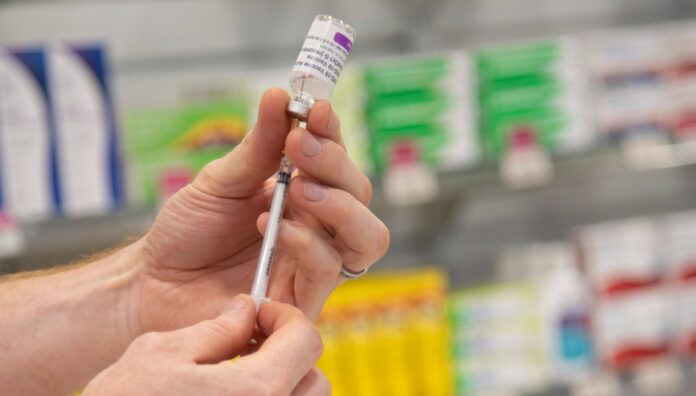
Each week, pharmacists across the country ring PSA’s Pharmacist to Pharmacist advice line looking for guidance on everything from practice-related queries to ethical dilemmas. In our occasional series, Australian Pharmacist will speak to the experts and answer some of the most frequently asked questions.
This month, we’ll again focus on COVID-19 vaccination questions answered by Amanda Fairjones, Professional Support Advisor.
Have a question of your own? PSA members can call 1300 369 772 to receive professional advice and support.
1. What do I do when someone under 60 years of age chooses to receive the AstraZeneca vaccine in a community pharmacy?
This is dependent on where you practice, as pharmacists must both adhere to relevant state/territory legislation and guidance, and follow the advice of the Australian Technical Advisory Group on Immunisation (ATAGI) and the Therapeutic Goods Administration (TGA).
For example, in NSW pharmacist immunisers may administer the COVID-19 AstraZeneca vaccine to individuals aged 18 years and older (regardless of geographical location) providing they do not have a precaution or contraindication to COVID-19 vaccination, and after obtaining fully informed consent. Informed consent requires discussion of the risks and benefits of vaccination using the latest information and advice from ATAGI and the TGA. Pharmacist immunisers must ensure they remain up to date with the advice from ATAGI or the TGA in relation to COVID-19 vaccination.
For further information in relation to the state or territory in which you practice please visit www.psa.org.au/coronavirus for more details, or PSA members can contact our professional support line on 1300 369 772.
2. What symptoms should persons be advised to look out for post-vaccination in relation to the AstraZeneca (AZ) vaccine?
Persons should be advised to present for medical advice if they develop any severe, persistent symptoms after vaccination. Persons should be provided with advice to immediately seek medical review for any new, severe persistent headache, stomach (abdominal pain), chest pain, vomiting or visual symptoms, bruising or petechial haemorrhages between 4 and 30 days after vaccination with the Covid-19 AZ vaccine.
Any patient with concerning signs or symptoms potentially related to thrombosis with thrombocytopenia syndrome (TTS) following receipt of the COVID-19 AZ vaccine should be referred to an emergency department for assessment and investigation.
3. What happens if the second dose of AZ is missed or given late?
If the second dose of the AZ vaccine is overdue (i.e. past the recommended 12-week interval for AZ), it should be given as soon as possible. A single dose likely only provides short-term protection. The second dose will be effective regardless of how late it is given. Even if the second dose is late, the current advice is no vaccine doses need to be repeated.
The role and evidence for additional booster doses is still emerging. However, it is likely these will be required into the future. It is clear that pharmacists in Australia will play a continued role in COVID immunisation, ensuring adequate booster immunisation rates should they be required.
4. Can pharmacists in suitable community pharmacies administer the second AZ dose if the first is given in a general practice or vaccination clinic?
Yes, this is perfectly okay. In fact, early results are showing, up to 30% of AZ vaccinations given by pharmacists in community pharmacy have been for the second AZ dose, with the first dose being given in a GP or vaccination clinic.
5. When should I upload COVID-19 vaccination information to AIR?
COVID-19 vaccine encounters should be uploaded into AIR within 24 hours from vaccine administration (including the patient’s individual Medicare reference number), or as soon as possible, to ensure consumer immunisation information is up to date.
It is mandatory under the Australian Immunisation Register Act 2015 to report all COVID-vaccine encounters to the Australian Immunisation Register (AIR). It is recommended pharmacist immunisers also check a patient’s immunisation history in AIR prior to administering the COVID-19 vaccine to ensure the correct vaccine is given within the appropriate timeframe (in some jurisdictions this is mandatory).
The ways to report a vaccination to the AIR include:
- Pharmacy professional services software integrated with AIR (both MedAdvisor and GuildCare have this function)
- Direct upload into AIR via the Clinician Vaccine Integrated Platform (operated by the Australian Digital Health Agency) or HPOS on the AIR website.
Pharmacist immunisers should confirm with their software provider that their existing professional services software will report COVID-19 vaccinations to the AIR and also how to enable the AIR viewing function on their software.
6. Should antipyretics and analgesics be used prior to vaccination?
Prophylactic use of antipyretics and analgesics is not recommended before receiving a COVID-19 vaccine; however, antipyretics and analgesics can be taken after vaccination for management of vaccine-related side effects such as fever and myalgia. Both paracetamol and ibuprofen have been shown to be effective post vaccination to reduce this discomfort.
7. Can I count CPD credits for completing the Australian Government COVID-19 vaccination training?
Yes. Continuing Professional Development activities with assessment, such as the Australian Government COVID-19 vaccination training can be recorded as a Group 2 activity, and two CPD credits per hour of activity can be counted. PSA members are able to self-record this training to their CPD plan.
In accordance with the Pharmacy Board Registration Standards, activities that address your professional development needs, are relevant to your scope of practice and are conducted by qualified persons can be counted towards your continuing professional development credits.
For further information, the Pharmacy Board document answers frequently asked questions in relation to CPD activities.



 Katie Phillips MPS[/caption]
Katie Phillips MPS[/caption]
 Erica Stephenson MPS with her kids[/caption]
Erica Stephenson MPS with her kids[/caption]
 Katie Phillip MPS with her two daughters[/caption]
Katie Phillip MPS with her two daughters[/caption]
 Deborah Hawthorne FPS with her two daughters[/caption]
Deborah Hawthorne FPS with her two daughters[/caption]


 Dr Amy Page (she/her) PhD, MClinPharm, GradDipBiostat, GradCertHProfEd, GradCertDMED, GAICD[/caption]
Dr Amy Page (she/her) PhD, MClinPharm, GradDipBiostat, GradCertHProfEd, GradCertDMED, GAICD[/caption]

 Amanda Quek (she/her) BPharm(Hons), GradCertAppPharmPrac[/caption]
Amanda Quek (she/her) BPharm(Hons), GradCertAppPharmPrac[/caption]


 ‘The Home of Pharmacists theme signifies belonging, connection, and purpose, uniting pharmacists across all areas of practice,’ said PSA National President Mark Naunton MPS.
‘The Home of Pharmacists theme signifies belonging, connection, and purpose, uniting pharmacists across all areas of practice,’ said PSA National President Mark Naunton MPS.


 Adjunct Professor Manya Angley FPS (CredPharm MMR) Credentialed and Disability Pharmacist
Adjunct Professor Manya Angley FPS (CredPharm MMR) Credentialed and Disability Pharmacist Penny Beirne MPS
Penny Beirne MPS