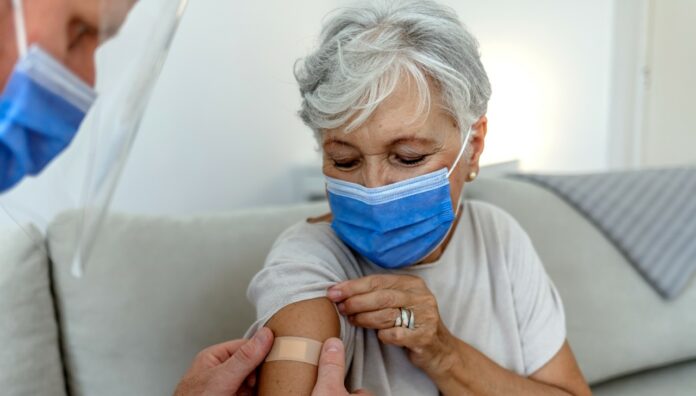
Third primary doses of COVID-19 vaccines for severely immunocompromised people and booster doses for the general population were both approved last month, but pharmacists report patients have trouble distinguishing between the two.
Since the Australian Technical Advisory Group on Immunisation (ATAGI) recommended third primary COVID-19 vaccine doses for immunocompromised people in early October, Melbourne-based pharmacist May Chang MPS has been approached by many patients requesting a ‘booster’ dose.
Only three, however, have actually been eligible to receive a third dose of a COVID-19 vaccine.
‘We were eased into it gently because our first [patient], who had received an organ transplant, actually had a letter from the Royal Melbourne Hospital requesting that she be given a third dose,’ Ms Chang told Australian Pharmacist.
At a glance
|
Eligible patients include those over 12 years of age with certain conditions, including active haematological malignancy, solid organ transplant with immunosuppressive therapy and haematopoietic stem cell transplant recipients. Patients who are prescribed immunosuppressive medicines, such as high-dose corticosteroids, are also eligible.
Those whose conditions are not listed in the ATAGI recommendations should be considered eligible only when a doctor has determined a comparative level of immunocompromise.
Third doses should be administered 2–6 months after a patient’s second dose, although a 4-week interval can be considered in special circumstances, such as during COVID-19 outbreaks or an acceleration of immunosuppression.
While Ms Chang’s patient had previously been administered two doses of the AstraZeneca vaccine, she explained that the third dose would be Moderna.
AstraZeneca can be given to patients who received this vaccine for their first two doses when there are no contraindications, significant adverse reactions or precautions for use, but ATAGI recommends an mRNA vaccine for the third dose.
Clear protocols for third doses

There are multiple ways for pharmacist immunisers to determine if someone is eligible for a third primary dose due to severe immunocompromise.
For example, Ms Chang has been presented with letters describing an eligible condition from their doctor or specialist.
These letters must contain reasonable evidence of the patient’s health condition as the basis for the third primary dose request.
As independent decision makers to administer COVID-19 vaccines, pharmacist immunisers could also make an assessment on the appropriateness of a third primary dose by checking My Health Record or the patient’s dispensing history.
This is what Ms Chang did when a request came through from a patient with multiple sclerosis.
‘We could see from her dispense records that she was on Gilenya, [which is] an immunosuppressant,’ she said.
For new patients, Ms Chang recommends broaching the topic in conversation.
‘When you ask them, “What medicines are you on?” They are usually quite forthcoming,’ she said.
One form of evidence, such as identifying medicines used following transplants, should be sufficient. But if you are unsure, for example, if a letter from the doctor doesn’t disclose a patient’s eligible medical condition, you can request another.
Earlier this week, Ms Chang was forced to turn away a patient who came in with a medicine summary that did not contain information suggesting severe immunosuppression from her GP, and no letter of request.
If patients are are not able to provide further evidence, they can sign a declaration using the Department of Health form.
Before administering the dose, Ms Chang also recommends checking when their last dose was administered.
‘Do your due diligence by logging onto PRODA to check they’re not rushing the dates on you,’ she said.
Post vaccine, Ms Chang regularly recorded third dose administrations as another second dose in the Australian Immunisation Register (AIR). But now, AIR provides a ‘dose three’ option, where both third primary doses and booster doses can be recorded.
Addressing confusion over ‘boosters’
While pharmacy staff were made aware of the difference between third primary doses and booster doses early on, Ms Chang said there is plenty of confusion among patients.
‘Everybody was coming in saying, “What’s this about booster shots?” So that’s when we had to work out a way of telling people,’ she said.
Ms Chang explains that when a person’s immune system is compromised, it takes three doses of the vaccine to do the same job as a healthy immune system would accomplish with two.
‘[I’ll say], “You know how much work you can do when you’re well, imagine if you’re having a sick day, you obviously won’t accomplish the same amount of work, so same goes with our immune system”,’ she said.
Ms Chang then explains that booster doses are available to patients who do not have compromised immune systems 6 months after their primary course.
The role of boosters, or a fourth dose, in people with immunocompromise is yet to be determined by ATAGI.
‘Politicians are not doctors, so they sometimes use the word booster in a non-medical manner, so that’s where I have to differentiate and go through that spiel with them as to who’s allowed the third [dose] right now.’
Once she explains the difference in medical terminology between third primary and booster doses, patients relax.
‘A lot of them come in flustered and worried, so it helps them to be a bit more receptive if you speak in a considered tone, using visual analogies which are easier to grasp,’ she said. ‘It’s just about trying to stop the panic.’



 Yvette Anderson MPS[/caption]
Yvette Anderson MPS[/caption]


 Diabetes medicines and peri-procedural blood glucose management
Diabetes medicines and peri-procedural blood glucose management
 Katie Phillips MPS[/caption]
Katie Phillips MPS[/caption]
 Erica Stephenson MPS with her kids[/caption]
Erica Stephenson MPS with her kids[/caption]
 Katie Phillip MPS with her two daughters[/caption]
Katie Phillip MPS with her two daughters[/caption]
 Deborah Hawthorne FPS with her two daughters[/caption]
Deborah Hawthorne FPS with her two daughters[/caption]


 Dr Amy Page (she/her) PhD, MClinPharm, GradDipBiostat, GradCertHProfEd, GradCertDMED, GAICD[/caption]
Dr Amy Page (she/her) PhD, MClinPharm, GradDipBiostat, GradCertHProfEd, GradCertDMED, GAICD[/caption]

 Amanda Quek (she/her) BPharm(Hons), GradCertAppPharmPrac[/caption]
Amanda Quek (she/her) BPharm(Hons), GradCertAppPharmPrac[/caption]


 ‘The Home of Pharmacists theme signifies belonging, connection, and purpose, uniting pharmacists across all areas of practice,’ said PSA National President Mark Naunton MPS.
‘The Home of Pharmacists theme signifies belonging, connection, and purpose, uniting pharmacists across all areas of practice,’ said PSA National President Mark Naunton MPS.