The Therapeutic Goods Administration (TGA) has requested comments from interested parties on the addition of a new advisory statement for labels of non-prescription medicines containing benzocaine for topical oral use.
The invitation to comment on proposed changes to the Required Advisory Statements for Medicine Labels (RASML) document comes following the safety alert issued in the United States in May.
A safety alert was issued by the Food and Drug Administration (FDA) that over-the-counter (OTC) oral products containing benzocaine should not be used to treat infants and children younger than 2 years due to the risk of methaemoglobinaemia.
Due to the risk posed by the life-threatening condition, and the negligible benefit that oral products containing benzocaine provides to teething infants, the FDA has requested manufacturers stop marketing OTC oral benzocaine products for this purpose.
There are no benzocaine-containing products registered in Australia specifically for use in infant teething, however the TGA is proposing updating the entry in the RASML statements for medicines containing benzocaine to read:
Do not give to children under 6 years of age, unless recommended by a doctor, pharmacist or dentist.
In preparations for topical oral use EXCEPT lozenges: Do not give to children under 2 years of age.
Submissions are invited with suggestions on the wording of the RASML statement and feedback on how the proposed change will affect relevant parties, including quantified costs and benefits.
Enquiries can be made to rasml@tga.gov.au and submissions at the TGA website.



 This CPD activity is sponsored by Reckitt. All content is the true, accurate and independent opinion of the speakers and the views expressed are entirely their own.[/caption]
This CPD activity is sponsored by Reckitt. All content is the true, accurate and independent opinion of the speakers and the views expressed are entirely their own.[/caption]
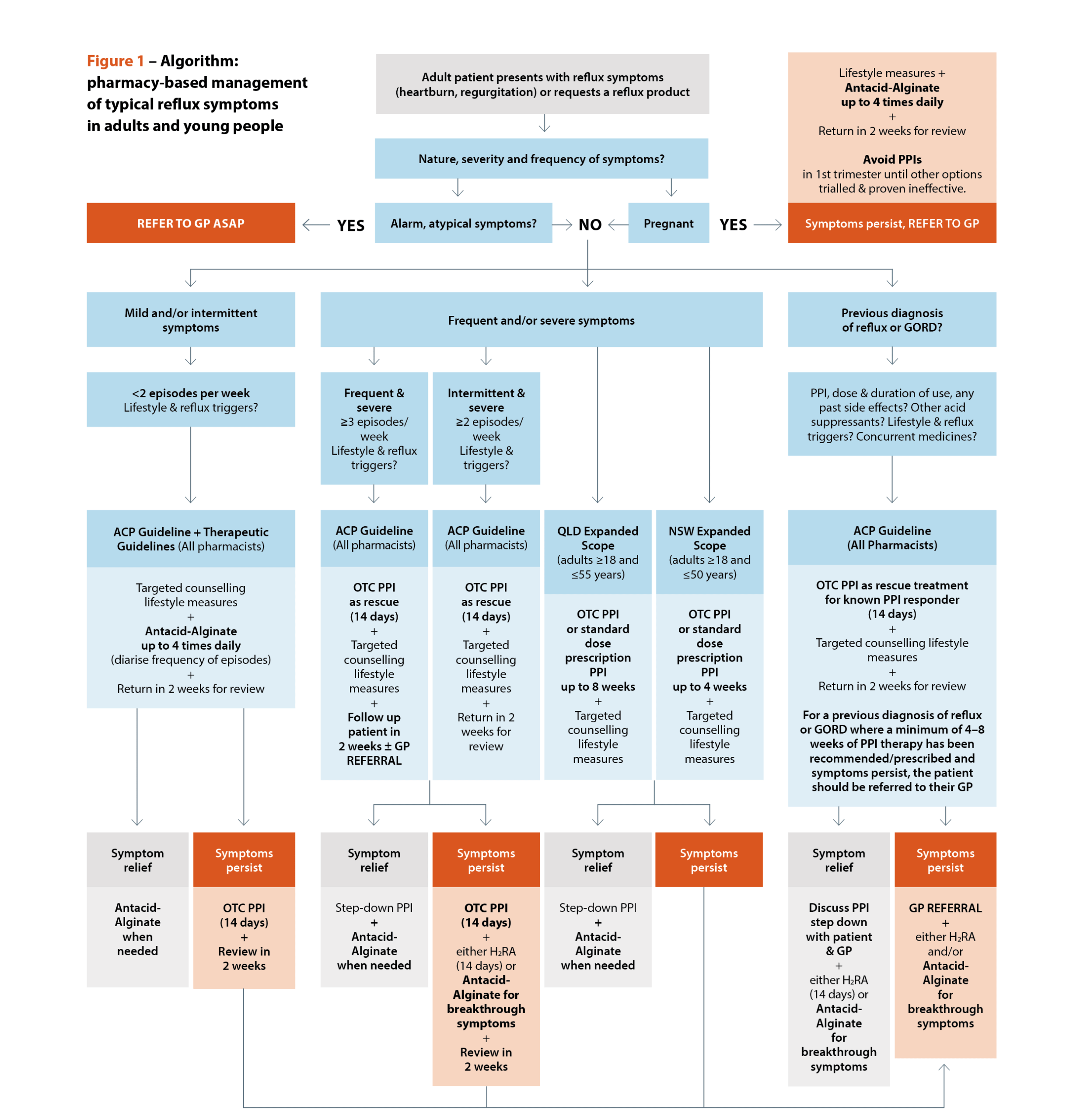 Sources: Australasian College of Pharmacy. Management of reflux: a guideline for pharmacists. Queensland Health. Queensland Community Pharmacy Gastro-oesophageal Reflux and Gastro-oesophageal Reflux Disease – Clinical Practice Guideline. NSW Health. NSW Pharmacist Practice Standards for gastro-oesophageal reflux and gastro-oesophageal reflux disease.[/caption]
Sources: Australasian College of Pharmacy. Management of reflux: a guideline for pharmacists. Queensland Health. Queensland Community Pharmacy Gastro-oesophageal Reflux and Gastro-oesophageal Reflux Disease – Clinical Practice Guideline. NSW Health. NSW Pharmacist Practice Standards for gastro-oesophageal reflux and gastro-oesophageal reflux disease.[/caption]

 Dr Ming S Soh PhD, BPharm (Hons)[/caption]
Dr Ming S Soh PhD, BPharm (Hons)[/caption]






 DR Amy Page (she/her) PhD, MClinPharm, GradDipBiostat, GCertHProfEd, GAICD, GStat, FSHPA, FPS is a consultant pharmacist, biostatistician, and the director of the Centre for Optimisation of Medicines at UWA’s School of Allied Health.[/caption]
DR Amy Page (she/her) PhD, MClinPharm, GradDipBiostat, GCertHProfEd, GAICD, GStat, FSHPA, FPS is a consultant pharmacist, biostatistician, and the director of the Centre for Optimisation of Medicines at UWA’s School of Allied Health.[/caption]

 Hui Wen Quek (she/her) BPharm(Hons), GradCertAppPharmPrac is a pharmacist and PhD candidate at the University of Western Australia (UWA).[/caption]
Hui Wen Quek (she/her) BPharm(Hons), GradCertAppPharmPrac is a pharmacist and PhD candidate at the University of Western Australia (UWA).[/caption]

 Gauri Godbole FPS[/caption]
Gauri Godbole FPS[/caption]






