

td_module_mega_menu Object
(
[post] => WP_Post Object
(
[ID] => 32230
[post_author] => 3410
[post_date] => 2026-06-03 13:02:08
[post_date_gmt] => 2026-06-03 03:02:08
[post_content] => The combination of an award wage rise and pay equity increase is set to deliver a meaningful lift in pharmacist remuneration.
Pharmacists are in line for a pay boost. AP investigates the drivers set to lift wages from 1 July.
Strong award wage increase
Pharmacists employed by the Pharmacy Industry Award 2020 will experience their largest annual wage rise in years. The Fair Work Commission (FWC) announced the 2026 Annual Wage Review decision yesterday (2 June), increasing modern award minimum wages by 4.75% from 1 July.
The increase is lower than the 6% wage rise supported by unions, but higher than the rate proposed by employer and business groups.
Along with the Australian Industry Group and the Australian Chamber of Commerce, the Pharmacy Guild of Australia advocated for a 3.5% wage increase, advising that this was the highest level businesses could accommodate.
However, the FWC’s decision was based on the challenges the nation is facing, including Reserve Bank interest rate tightening and the conflict in the Middle East.
In its decision, the FWC noted ‘ regrettably, … it is not practicable in the current uncertain circumstances to award a real wage increase for employees reliant on modern award wage rates that would be sufficient to close the real wage gap entirely’ compared to the post-pandemic inflation spike after July 2021.
‘I consistently hear from our members that remuneration is their number one concern, particularly given cost-of-living pressures,’ said PSA National President Professor Mark Naunton MPS. ‘For many community pharmacists, who make up a significant proportion of our membership, the award increase feels long overdue.’
Year 2 of the gender undervaluation correction
In 2025, the FWC’s Expert Panel for pay equity in the care and community sector determined that pharmacists have been subject to a total of 14.1% gender-based undervaluation.
‘Last year's gender discrimination determination was so important in addressing longstanding discrimination in the award rate for pharmacists, which was found to be directly linked to the high proportion of the workforce that is female,’ Prof Naunton said.
The Expert Panel issued a determination that there will be a total increase in the minimum wage rates of 14.1% over 3 years, with the second increase set to take place on 30 June 2026.
As AP pointed out last year, not all pharmacists will see a direct pay rise. The award sets a remuneration level, and Fair Work data indicates around 12.7% of pharmacists are paid at award rates only – though the proportion is likely higher in community pharmacy, where collective agreements are less common than in the hospital sector.
While pharmacy interns will see another pay rise, pharmacy assistants and pharmacy students are not included, as their rates were not found to be undervalued.
‘We welcome the next stage of the gender discrimination increase to the Pharmacy Industry Award, which will take effect from 1 July, in addition to the 4.75% increase from the annual wage review,’ Prof Naunton added.
What will the new award hourly rate be?
The exact updated pay rates will be confirmed when the FWC publishes new pay guides ahead of 1 July.
And while the wage increase is a relief for many pharmacists, Prof Naunton said PSA also recognised that there are other members who are being left further behind financially.
‘We continue to fight to increase remuneration for pharmacists who provide medication review services, who have had no fee increase in 7 years,’ he added.
[post_title] => Pharmacists in line for pay increase
[post_excerpt] => The combination of an award wage rise and pay equity increase is set to deliver a meaningful lift in pharmacist remuneration.
[post_status] => publish
[comment_status] => open
[ping_status] => open
[post_password] =>
[post_name] => pharmacists-in-line-for-pay-increase
[to_ping] =>
[pinged] =>
[post_modified] => 2026-06-03 15:41:29
[post_modified_gmt] => 2026-06-03 05:41:29
[post_content_filtered] =>
[post_parent] => 0
[guid] => https://www.australianpharmacist.com.au/?p=32230
[menu_order] => 0
[post_type] => post
[post_mime_type] =>
[comment_count] => 0
[filter] => raw
)
[title_attribute] => Pharmacists in line for pay increase
[title] => Pharmacists in line for pay increase
[href] => https://www.australianpharmacist.com.au/pharmacists-in-line-for-pay-increase/
[module_atts:td_module:private] => Array
(
)
[td_review:protected] => Array
(
)
[is_review:protected] =>
[post_thumb_id:protected] => 32232
[authorType] =>
)
td_module_mega_menu Object
(
[post] => WP_Post Object
(
[ID] => 32217
[post_author] => 12503
[post_date] => 2026-06-01 12:04:52
[post_date_gmt] => 2026-06-01 02:04:52
[post_content] => Four of Australia’s leading health peak bodies have united in calling for investment in general practice pharmacists to make medicine use safer and strengthen multidisciplinary care.
Speaking at an inter-professional panel session at the 2026 GP Pharmacist Symposium today on the Gold Coast, presidents from PSA, Australian Medical Association, Royal Australian College of General Practitioners and Australian Primary Health Care Nurses Association reinforced the clinical need and value of embedding pharmacists within general practice.
The session provided a real-life demonstration of cohesion, bringing together representatives from general practice, medicine, nursing and pharmacy.
Exploring how collaborative healthcare efforts can improve patient care, reduce pressure on hospitals and strengthen the primary care workforce, the panel was made up of extraordinary leaders, including:
td_module_mega_menu Object
(
[post] => WP_Post Object
(
[ID] => 32211
[post_author] => 3410
[post_date] => 2026-06-01 11:39:57
[post_date_gmt] => 2026-06-01 01:39:57
[post_content] => Australia's ADHD medicine supply has stabilised – for now. Here’s what pharmacists need to know before the next shortage hits.
After several years of disruption, Australia's ADHD medicine supply has largely recovered. But the shortages of lisdexamfetamine (Vyvanse) in 2023–24 and methylphenidate in 2025 exposed global supply chain vulnerabilities. With diagnosis rates rising, prescribing arrangements expanding and global manufacturing concentrated overseas, pharmacists may once again find themselves managing the fallout from supply interruptions.
[caption id="attachment_32071" align="alignright" width="300"] Yvette Anderson MPS[/caption]
Yvette Anderson MPS – founder of Spectrum Pharmacist, which sets out to bridge the gap for neurodivergent families – tells AP how preparation, collaboration and neuroaffirming care will be essential when the next shortage arrives.
Yvette Anderson MPS[/caption]
Yvette Anderson MPS – founder of Spectrum Pharmacist, which sets out to bridge the gap for neurodivergent families – tells AP how preparation, collaboration and neuroaffirming care will be essential when the next shortage arrives.
A fragile recovery
The shortages that dominated ADHD care over the past 3 years have largely eased, Ms Anderson said.
‘Except for one particular strength of long-acting methylphenidate (30 mg), which is a bit hit and miss.’
However, the situation remains precarious.
Australia relies heavily on overseas manufacturing for ADHD medicines, with production historically concentrated in Ireland and Germany. Supply is also influenced by annual manufacturing quotas established by the United States Drug Enforcement Administration (DEA), which shape global production volumes for controlled substances.
When the Vyvanse patent expired in the United States in 2023, demand for generic lisdexamfetamine surged – rapidly straining international supply chains. More recently, scrutiny of DEA production quotas under the Make America Healthy Again policy framework contributed to a significant reduction in manufacturing, creating flow-on effects internationally.
Due to international uproar, production recommenced. ‘But it's still not probably at the rate and quantity needed globally, so we're always going to sit in a bit of a precarious area,’ Ms Anderson said.
Demand is surging
While supply remains vulnerable, Australia's ADHD treatment landscape is also undergoing significant change.
Initiation of ADHD medicines was largely restricted to paediatricians and psychiatrists. Now, most states and territories have introduced pathways that allow appropriately trained GPs to diagnose and prescribe ADHD medicines.
'A number of GPs in different states have either already been trained, or their state or territory has put laws in place to allow them to be trained to diagnose and prescribe medications,' Ms Anderson said.
Diagnosis rates are also increasing due to changes in diagnosis criteria. More accessible and affordable treatment pathways will help to ensure patients can receive timely access to care.
Importantly, diagnosis does not automatically lead to pharmacological treatment.
'Just because there is potentially going to be an increase in the number of Australians getting diagnosed, that number won't directly correlate to the same number of increasing supply of medication,' Ms Anderson said. 'Medication is only one tiny piece of your management plan, and for some people, medicines aren't the way they want to go, or they don't suit them, or find them effective,'
Nevertheless, more diagnoses and more prescribers are likely to increase demand for medicines supplied through already fragile global supply chains.
Is it possible to get ahead of shortages?
Yes, and no.
Ms Anderson encourages pharmacies to establish systems that allow them to identify and respond to potential disruptions early. This includes monitoring Therapeutic Goods Administration (TGA) medicine shortage alerts.
Once a shortage is identified, pharmacies can use dispensing records to determine which patients may be affected and begin discussions with prescribers before supplies run out.
‘It's a matter of looking at who the prescribers are and reaching out to them and saying, “The TGA has alerted us to this. We know you have a number of patients on this medication. What can we do as a collaborative to support this transition and this shortage period?”’ Ms Anderson said.
As a hospital pharmacist, she was able to pass on information about ADHD medicine shortages to paediatricians – who opened telehealth appointments to turn scripts around quickly.
‘I also communicated with the community pharmacies in my area to ask: Who has stock? Who doesn't? Can we redirect patients?’ Ms Anderson said. ‘Working together as a community, making sure you're embedded in that multidisciplinary team really pays off when there are medication shortages.’
Switching medicines safely
When ADHD medicines are in short supply, patients may need to adjust their medicines; so it’s crucial to be aware of the various formulations, durations of action and release characteristics of different stimulant products.
‘If someone's stabilised on Ritalin, they may go on Ritalin LA or Concerta – but Concerta's duration of action is longer and Ritalin LA's is shorter – so we need to be considering: does this person work through to 6.00 pm? Do we need some immediate release on top of that?’ Ms Anderson said. ‘It's not a straight switch between the medicines.’
The challenges become greater when patients need to move between stimulant classes, such as from lisdexamfetamine to methylphenidate.
‘I've seen people that have quite significant adverse effects after switching to lisdexamfetamine and a couple of days later being unable to function,’ she said. ‘Even though we know someone might be getting a positive effect from stimulants, it doesn't mean it's going to be the same for all stimulants.’
Where stimulant options become unavailable, non-stimulant medicines such as atomoxetine, guanfacine (Intuniv) and clonidine (off-label) can provide alternative management pathways.
‘These non-stimulant options have really good evidence [of efficacy], but they're probably underutilised,' Ms Anderson said. ‘When we do come to another significant shortage, there's only a handful of stimulant medicines – so we need to be able to talk to patients about other options.’
Learn more about supporting patients through ADHD medicine shortages by attending the ADHD care Session at PSA26, held from 31 July to 2 August at the ICC in Sydney.
[post_title] => How to manage ADHD medicine shortages
[post_excerpt] => Australia's ADHD medicine supply has stabilised – for now. Here’s what pharmacists need to know before ADHD medicine shortages strike again.
[post_status] => publish
[comment_status] => open
[ping_status] => open
[post_password] =>
[post_name] => how-to-manage-adhd-medicine-shortages
[to_ping] =>
[pinged] =>
[post_modified] => 2026-06-01 15:15:51
[post_modified_gmt] => 2026-06-01 05:15:51
[post_content_filtered] =>
[post_parent] => 0
[guid] => https://www.australianpharmacist.com.au/?p=32211
[menu_order] => 0
[post_type] => post
[post_mime_type] =>
[comment_count] => 0
[filter] => raw
)
[title_attribute] => How to manage ADHD medicine shortages
[title] => How to manage ADHD medicine shortages
[href] => https://www.australianpharmacist.com.au/how-to-manage-adhd-medicine-shortages/
[module_atts:td_module:private] => Array
(
)
[td_review:protected] => Array
(
)
[is_review:protected] =>
[post_thumb_id:protected] => 32214
[authorType] =>
)
td_module_mega_menu Object
(
[post] => WP_Post Object
(
[ID] => 32207
[post_author] => 12503
[post_date] => 2026-06-01 11:03:42
[post_date_gmt] => 2026-06-01 01:03:42
[post_content] => Two pharmacists were honoured at PSA’s Consultant Pharmacist Conference (CPC26), held from 29–31 May 2026 on the Gold Coast.
Kelly Abbott MPS was awarded 2026 PSA MIMS Consultant Pharmacist of the Year for her outstanding contribution to consultant pharmacy practice and passion for improving patient care throughout her hometown of Gippsland, Victoria.
Tiernan McDonough MPS, based in South Australia, was named 2026 PSA AMH Aged Care Pharmacist of the Year for excellence, leadership, and innovation in aged care pharmacy.
Tireless HMR advocate
Highly regarded across the industry for her work in delivering Home Medicines Reviews, Residential Medication Management Reviews, and providing Quality Use of Medicines (QUM) services, Ms Abbott’s 15 years of service to consultant pharmacy make her a well-deserving winner of this year’s award.
PSA National President, Professor Mark Naunton MPS, said Ms Abbott epitomises many credentialed pharmacists as a passionate and relentless advocate for HMRs, her patients, and the greater credentialed pharmacist workforce.
‘Kelly has continued to be actively involved in grassroots advocacy, notably in recent months during the First Pharmacy Programs Agreement negotiations led by PSA, ensuring the voices of frontline pharmacists and patients were heard,’ he said.
‘Kelly is a loyal, responsive, and reliable contributor to PSA and the Consultant Pharmacist of Australia working groups, while balancing multiple professional roles and caring for her family.
‘Her contributions to PSA and the profession are honourable. We are proud to recognise Kelly as the PSA MIMS Consultant Pharmacist of the Year and celebrate her significant and ongoing impact.’
Aged care champion
Mr McDonough has been recognised for his ongoing dedication and commitment to aged care pharmacy practice through his support for residents and healthcare teams in delivering medication reviews and QUM services.
PSA National President, Professor Mark Naunton MPS, said Mr McDonough embodies the essential role pharmacists play in aged care.
‘Pharmacists working in aged care are critical to ensuring safe and effective medicines use, but Tiernan’s impact extends beyond the aged care sector in which he practices,’ he said.
‘Tiernan has led an important mentoring program that connects pharmacists within the residential aged care profession, to reduce professional isolation and support further workforce development.
‘His work demonstrates his compassion, leadership, and strong drive for improving care for older Australians.’
[post_title] => Consultant pharmacist excellence awarded
[post_excerpt] => Two pharmacists were honoured at PSA’s Consultant Pharmacist Conference (CPC26), held from 29–31 May 2026 on the Gold Coast.
[post_status] => publish
[comment_status] => open
[ping_status] => open
[post_password] =>
[post_name] => top-consultant-pharmacists-honoured-at-cpc26
[to_ping] =>
[pinged] =>
[post_modified] => 2026-06-01 15:18:21
[post_modified_gmt] => 2026-06-01 05:18:21
[post_content_filtered] =>
[post_parent] => 0
[guid] => https://www.australianpharmacist.com.au/?p=32207
[menu_order] => 0
[post_type] => post
[post_mime_type] =>
[comment_count] => 0
[filter] => raw
)
[title_attribute] => Consultant pharmacist excellence awarded
[title] => Consultant pharmacist excellence awarded
[href] => https://www.australianpharmacist.com.au/top-consultant-pharmacists-honoured-at-cpc26/
[module_atts:td_module:private] => Array
(
)
[td_review:protected] => Array
(
)
[is_review:protected] =>
[post_thumb_id:protected] => 32209
[authorType] =>
)
td_module_mega_menu Object
(
[post] => WP_Post Object
(
[ID] => 31968
[post_author] => 8963
[post_date] => 2026-06-01 09:56:44
[post_date_gmt] => 2026-05-31 23:56:44
[post_content] =>
Case scenarioJohn, 62, regularly visits your community pharmacy. Following his referral to a gastroenterologist, he presents to purchase a bowel cleansing kit and asks for advice about his upcoming endoscopy and colonoscopy. John reports a history of difficulty swallowing certain foods and says a family member recently told him that his grandfather had bowel cancer. He is feeling anxious about the procedures and the bowel preparation. |
Endoscopy and colonoscopy are commonly performed gastrointestinal procedures that pharmacists may encounter. Patients may require counselling before and after investigations. Endoscopy refers to procedures that use an endoscope – a thin, flexible tube with a light and camera – to examine internal organs. However, in both clinical and public contexts, particularly in gastroenterology, the term endoscopy is generally used to refer specifically to oesophagoscopy, gastroscopy or duodenoscopy, which involves examining the upper gastrointestinal tract (oesophagus, stomach and duodenum). Colonoscopy is the term used specifically for the examination of the lower gastrointestinal tract (colon/bowel) and includes the entire colon, rectum and terminal ileum. Both procedures are usually performed by gastroenterologists, although colonoscopies may also be performed by colorectal surgeons.1
Learning objectivesAfter reading this article, pharmacists should be able to:
Competency standards (2016) addressed: 1.1, 1.4, 1.5, 2.2, 3.1, 3.5 Accreditation number: PSAAP2606BW Accreditation expiry: 31/05/2028 Accreditation points: 1.5 Group 2 credits |
Both endoscopy and colonoscopy may be used for diagnostic and therapeutic purposes. Standard diagnostic functions include inspection, biopsy, photography and videorecording; diagnostic observations may include focal benign or malignant lesions, mucosal changes, luminal obstruction, motility and strictures. Common therapeutic interventions include polypectomy, dilation of strictures, stent placement, removal of foreign bodies, gastrostomy and treatment of gastrointestinal bleeding.2

EndoscopyOesophagogastroduodenoscopy, more commonly referred to as endoscopy, is a valuable diagnostic procedure in the upper gastrointestinal tract, and is used mainly for detection and surveillance of premalignant conditions and cancers.3 It provides a clear view of mucosal surfaces of the oesophagus, stomach and proximal duodenum.2 It is being increasingly used for gastric cancer and oesophageal adenocarcinoma, the latter having an endoscopically detectable (and potentially curable) precursor lesion, Barrett’s oesophagus.3 Other forms of oesophagitis include eosinophilic oesophagitis, a chronic immune-mediated inflammatory disease of the oesophagus that causes dysphagia, food impaction of the oesophagus, and oesophageal strictures.4 |
Endoscopy with biopsy is mainly used for diagnosing stomach and oesophageal cancers. It is usually performed as day surgery and requires patients to fast for 4–6 hours before the procedure. Before insertion of the endoscope, the throat is sprayed with a local anaesthetic, and a sedative or appropriate anaesthesia is given to the patient. If any suspicious-looking areas are seen, a biopsy may be taken for examination by a pathologist, with results usually available within a few days.1
An endoscopy generally takes about 15 minutes. The patient may have a sore throat afterwards and feel a little bloated. An endoscopic ultrasound is sometimes performed at the same time as a standard endoscopy. This procedure helps determine whether a cancer has spread into the oesophageal wall, nearby tissues or lymph nodes, and is also used to determine whether the patient is a suitable candidate for surgery.1
Endoscopies have a low risk of adverse events. The risk of mortality is rated as 1 in 10,000.5 Possible adverse events include infection, perforation, bleeding, aspiration and cardiopulmonary events.5
ColonoscopyColonoscopy is the main procedure used to investigate or diagnose bowel cancer and is usually performed as day surgery. In 2023–24, there were 691,514 Medicare-subsidised colonoscopies in Australia, of which approximately 20% were repeat procedures performed within 2 years and 10 months of a previous colonoscopy.6 A colonoscope is a tiny camera on a long, flexible tube that is used to examine the entire length of the large bowel. It is inserted into the anus and up into the rectum and colon. Air or carbon dioxide is passed into the colon. Any abnormal tissue can be detected, polyps removed and biopsies taken for further examination. The process usually takes about 20–30 minutes.1 |
Indications for colonoscopy may include:
Australians aged 50–74 years with an average risk of bowel cancer should participate in Australia’s National Bowel Cancer Screening Program. Since 1 July 2024, people aged 45–49 years can also participate on request. A free screening kit is mailed to participants. This is a faecal occult blood test which uses a small stool sample that can be easily collected at home and returned for laboratory testing.1
Before insertion of the colonoscope, an appropriate anaesthesia is given. Patients may feel drowsy or weak prior to and particularly after the colonoscopy, therefore it is recommended that patients do not drive or travel alone. An occasional side effect of a colonoscopy is temporary wind pain and flatulence, especially if air rather than carbon dioxide is passed into the bowel during the procedure.1 More serious but rare complications include perforation (1 in 1,000 risk), infection and risks of sedation and anaesthesia.7
High-quality colonoscopy is required to ensure that cancers and adenomas are not missed, and to avoid adverse patient experiences. The Australian Colonoscopy Clinical Care Standard lists nine key factors, three of which are relevant to pharmacists7:
Informed decision making and patient consent: this is essentially between the patient and gastroenterologist, but patients may seek further advice.
Adequate bowel preparation: bowel cleansing products are generally provided by pharmacies. Reporting and patient follow-up: this is essentially between the patient and gastroenterologist, but patients may seek further advice.
For patients, bowel preparation can be an unpleasant experience.7 However, the importance of adequate bowel preparation and following instructions must be emphasised to patients to ensure that there is a clear view of the mucosa to detect adenomas.8 If the bowel preparation is inadequate, there is not only a lower likelihood of adenoma detection, but it may also result in longer procedures, repeat procedures, more risk to the patient and higher costs.7
Suboptimal bowel preparation has been reported in up to 25% of patients.9 Factors which may affect the bowel preparation include product choice, timing, diet and patient compliance.8
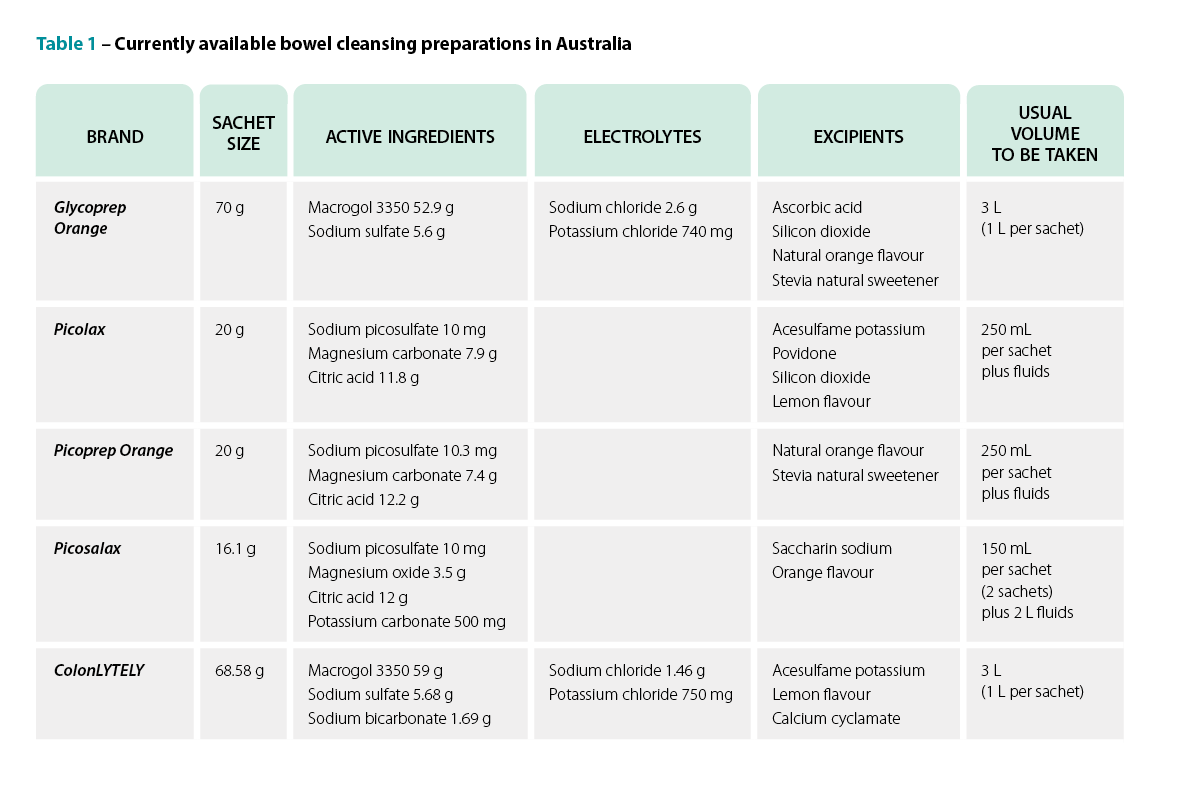
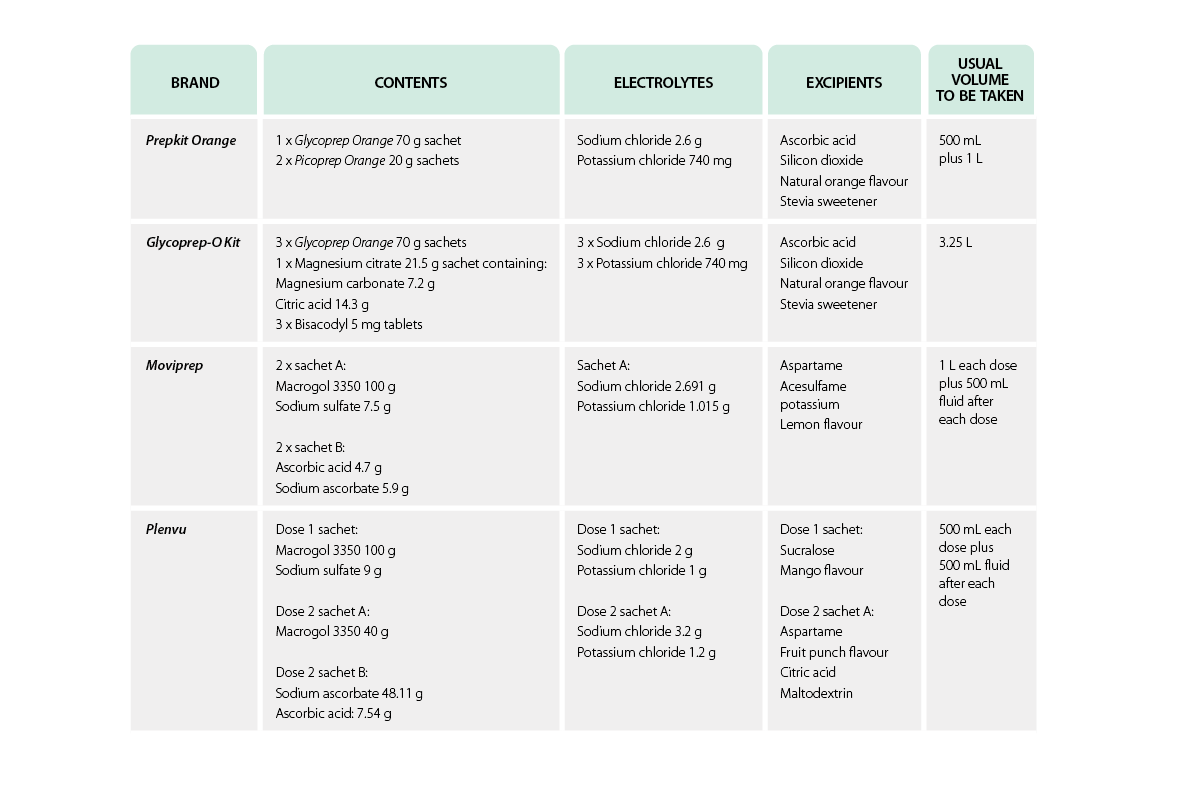
Product selection
Various products are available for bowel cleansing, and there is no consensus on which product is best.7 Over the years, many products have been used, including enemas, mannitol and large-volume solutions. The use of polyethylene glycol 3350 (PEG), also known as macrogol, represented a breakthrough in 1980. This was followed by sodium phosphate preparations in 1990, but these are no longer used because of the risk of renal toxicity.9 Today, many products are based on PEG with the addition of a balanced electrolyte solution and sometimes combined with sodium sulfate, bisacodyl or sodium ascorbate/ascorbic acid.
PEG does not alter the histology of the mucosa and rarely causes electrolyte disturbances.9 The volume to be consumed is an important consideration, with split dosing highly recommended for 4 L solutions (2 L the evening before and 2 L 5–6 hours before the procedure).7,9
The addition of ascorbic acid allows for lower-volume PEG formulations, as its osmotic effect enhances bowel cleansing. These preparations are generally better tolerated due to the reduced volume and improved palatability (typically ~3 L).9
Products based on magnesium citrate combined with sodium picosulfate are also available. These are as effective as PEG-based products and may be better tolerated.9 However, they need to be used with caution in elderly patients, those with renal impairment or heart disease, and those taking certain medications such as diuretics.9 It should be noted that bisacodyl and picosulfate are both converted in the gut to the same active metabolite.9 The decision regarding choice of product is generally made by the gastroenterologist depending on the patient characteristics.
Dietary modification
For a few days before the colonoscopy, patients may be advised to avoid high-fibre foods (such as fruit, vegetables, brown rice, wholegrain pasta, bran, cereals, nuts and seeds). Instead, low-fibre foods (such as white rice, white bread, meat, chicken, fish, yoghurt, cheese, potato and pumpkin) may be recommended.1 Patients may also be advised to drink only fluids, such as water, clear broth, black tea and coffee, and clear fruit juice (without pulp) for 12–24 hours before the procedure to prevent dehydration.1
Special populations and precautions
Patients requiring special care include those with lower gastrointestinal bleeding, chronic kidney disease, inflammatory bowel disease, heart failure, chronic constipation, those who have had previous bowel resection or are pregnant or lactating, as well as hospital inpatients and older people.8 Prevention of dehydration in patients undergoing bowel preparation minimises the risk of severe adverse events.9
Adverse events following bowel preparation are uncommon but may be potentially serious. The product selection decision is an important one because the risk of adverse effects is increased if there is inadequate hydration, inappropriate dosing and inappropriate patient selection.9
Hyponatraemia, because of vomiting or diarrhoea, can develop after taking any colonoscopy preparation. Vomiting may reduce plasma volume and increase thirst. Patients with renal insufficiency, hypothyroidism, mineralocorticoid deficiency, liver cirrhosis, heart failure, or those on diuretics, NSAIDs or ACEIs are at an increased risk of hyponatraemia.9
Improving tolerability
Various methods have been promoted to improve the tolerability and palatability of bowel cleansing preparations. These include chilling the preparation, drinking the preparation through a straw, drinking clear pulp-free juices or approved herbal teas, and chewing gum or various confectioneries concomitantly with the preparation.8
Post colonoscopy syndrome occurs in a third of patients and is more common in females and after lengthy procedures. Symptoms, which include abdominal pain, discomfort and bloating, persist for up to 48 hours following the procedure and may be caused by transient changes in the gut microbiota due to bowel cleansing.8
If a colonoscopy reveals anything of concern, a biopsy is taken for testing by a pathologist. Imaging (such as abdominal scans) may be required. If bowel cancer is diagnosed, scans will determine what stage it is at. Tissue taken at the time of colonoscopy may be tested for genetic changes in the cancer cells, which can guide the choice of treatment.1
Pharmacists are frequently involved in supporting patients undergoing endoscopy and colonoscopy, particularly in the community setting. Patients may seek reassurance about the purpose, safety and expected after-effects of these procedures. Providing clear, evidence-based information can help reduce anxiety. Pharmacists can provide advice regarding colonoscopy preparation by counselling on correct use of bowel cleansing products, split dosing, diet and hydration. They should identify patients at high risk of complications, including older adults, those with renal or cardiac impairment, or those taking diuretics, NSAIDs or ACE inhibitors, and provide tailored advice. Following the procedure, pharmacists can reassure patients about common after effects.
Endoscopy and colonoscopy are widely performed diagnostic and therapeutic procedures that play a central role in the detection and management of gastrointestinal disease, including malignancy. Although generally safe, both procedures require appropriate patient education and awareness of potential adverse effects.
Pharmacists are well positioned to support patients before and after these procedures by providing counselling, identifying individuals at increased risk of complications, and promoting effective bowel preparation for colonoscopy. Through proactive engagement, pharmacists can contribute to improved patient experience and procedural outcomes.
Case scenario continuedYou explain the purpose of both procedures and reassure John that they are commonly performed and generally low risk. You counsel him on how to use the bowel cleansing kit, emphasising the importance of following the instructions carefully. You also discuss the dietary and fluid modifications required in the days leading up to the colonoscopy and provide tips to improve tolerability of the preparation. Following the procedures, John returns to the pharmacy with a prescription for budesonide 1 mg orally dispersible tablets. He explains that he has been diagnosed with eosinophilic oesophagitis and that his oesophagus has been dilated. He also mentions that two polyps were removed from his colon. You reassure John and provide counselling on the correct use of his new medicine. |
[cpd_submit_answer_button]
DR Barry Werth PhD, BPharm (Hons), MBA, MPS is a pharmacist with over 50 years’ experience in the Australian pharmaceutical industry, holding a PhD in epidemiology/pharmacoepidemiology, an honours degree in pharmacy and an MBA. He is a Research Affiliate of the University of Sydney and holds senior advisory roles with MND Australia, alongside ongoing consultancy work for pharmaceutical companies.
Rose-marie Pennisi (she/her), BPharm, MBA, MPS
Cancer Council Australia. Cancer Council. Sydney: Cancer Council Australia; 2025. At: www.cancer.org.au
Early DS, Ben-Menachem T, Decker GA, et al. Appropriate use of GI endoscopy. Gastrointest Endosc. 2012;75(6):1127–31.
Januszewicz W, Kaminski MF. Quality indicators in diagnostic upper gastrointestinal endoscopy. Ther Adv Gastroenterol. 2020;13:1756284820916693.
Muir A, Falk GW. Eosinophilic esophagitis: a review. JAMA. 2021;326(13):1310–8.
Ben-Menachem T, Decker GA, Early DS, et al. Adverse events of upper GI endoscopy. Gastrointest Endosc. 2012;76(4):707–18.
Australian Commission on Safety and Quality in Health Care. Atlas focus report: colonoscopy. Sydney: Australian Commission on Safety and Quality in Health Care; 2025. At: www.safetyandquality.gov.au
Duggan A, Skinner IJ, Bhasale AL. All colonoscopies are not created equal: why Australia now has a clinical care standard for colonoscopy. Med J Aust. 2018;209(10):427–30.
Di Leo M, Iannone A, Arena M, et al. Novel frontiers of agents for bowel cleansing for colonoscopy. World J Gastroenterol. 2021;27(45):7748.
Adamcewicz M, Bearelly D, Porat G, et al. Mechanism of action and toxicities of purgatives used for colonoscopy preparation. Expert Opin Drug Metab Toxicol. 2011;7(1):89–101.
td_module_mega_menu Object
(
[post] => WP_Post Object
(
[ID] => 32230
[post_author] => 3410
[post_date] => 2026-06-03 13:02:08
[post_date_gmt] => 2026-06-03 03:02:08
[post_content] => The combination of an award wage rise and pay equity increase is set to deliver a meaningful lift in pharmacist remuneration.
Pharmacists are in line for a pay boost. AP investigates the drivers set to lift wages from 1 July.
Strong award wage increase
Pharmacists employed by the Pharmacy Industry Award 2020 will experience their largest annual wage rise in years. The Fair Work Commission (FWC) announced the 2026 Annual Wage Review decision yesterday (2 June), increasing modern award minimum wages by 4.75% from 1 July.
The increase is lower than the 6% wage rise supported by unions, but higher than the rate proposed by employer and business groups.
Along with the Australian Industry Group and the Australian Chamber of Commerce, the Pharmacy Guild of Australia advocated for a 3.5% wage increase, advising that this was the highest level businesses could accommodate.
However, the FWC’s decision was based on the challenges the nation is facing, including Reserve Bank interest rate tightening and the conflict in the Middle East.
In its decision, the FWC noted ‘ regrettably, … it is not practicable in the current uncertain circumstances to award a real wage increase for employees reliant on modern award wage rates that would be sufficient to close the real wage gap entirely’ compared to the post-pandemic inflation spike after July 2021.
‘I consistently hear from our members that remuneration is their number one concern, particularly given cost-of-living pressures,’ said PSA National President Professor Mark Naunton MPS. ‘For many community pharmacists, who make up a significant proportion of our membership, the award increase feels long overdue.’
Year 2 of the gender undervaluation correction
In 2025, the FWC’s Expert Panel for pay equity in the care and community sector determined that pharmacists have been subject to a total of 14.1% gender-based undervaluation.
‘Last year's gender discrimination determination was so important in addressing longstanding discrimination in the award rate for pharmacists, which was found to be directly linked to the high proportion of the workforce that is female,’ Prof Naunton said.
The Expert Panel issued a determination that there will be a total increase in the minimum wage rates of 14.1% over 3 years, with the second increase set to take place on 30 June 2026.
As AP pointed out last year, not all pharmacists will see a direct pay rise. The award sets a remuneration level, and Fair Work data indicates around 12.7% of pharmacists are paid at award rates only – though the proportion is likely higher in community pharmacy, where collective agreements are less common than in the hospital sector.
While pharmacy interns will see another pay rise, pharmacy assistants and pharmacy students are not included, as their rates were not found to be undervalued.
‘We welcome the next stage of the gender discrimination increase to the Pharmacy Industry Award, which will take effect from 1 July, in addition to the 4.75% increase from the annual wage review,’ Prof Naunton added.
What will the new award hourly rate be?
The exact updated pay rates will be confirmed when the FWC publishes new pay guides ahead of 1 July.
And while the wage increase is a relief for many pharmacists, Prof Naunton said PSA also recognised that there are other members who are being left further behind financially.
‘We continue to fight to increase remuneration for pharmacists who provide medication review services, who have had no fee increase in 7 years,’ he added.
[post_title] => Pharmacists in line for pay increase
[post_excerpt] => The combination of an award wage rise and pay equity increase is set to deliver a meaningful lift in pharmacist remuneration.
[post_status] => publish
[comment_status] => open
[ping_status] => open
[post_password] =>
[post_name] => pharmacists-in-line-for-pay-increase
[to_ping] =>
[pinged] =>
[post_modified] => 2026-06-03 15:41:29
[post_modified_gmt] => 2026-06-03 05:41:29
[post_content_filtered] =>
[post_parent] => 0
[guid] => https://www.australianpharmacist.com.au/?p=32230
[menu_order] => 0
[post_type] => post
[post_mime_type] =>
[comment_count] => 0
[filter] => raw
)
[title_attribute] => Pharmacists in line for pay increase
[title] => Pharmacists in line for pay increase
[href] => https://www.australianpharmacist.com.au/pharmacists-in-line-for-pay-increase/
[module_atts:td_module:private] => Array
(
)
[td_review:protected] => Array
(
)
[is_review:protected] =>
[post_thumb_id:protected] => 32232
[authorType] =>
)
td_module_mega_menu Object
(
[post] => WP_Post Object
(
[ID] => 32217
[post_author] => 12503
[post_date] => 2026-06-01 12:04:52
[post_date_gmt] => 2026-06-01 02:04:52
[post_content] => Four of Australia’s leading health peak bodies have united in calling for investment in general practice pharmacists to make medicine use safer and strengthen multidisciplinary care.
Speaking at an inter-professional panel session at the 2026 GP Pharmacist Symposium today on the Gold Coast, presidents from PSA, Australian Medical Association, Royal Australian College of General Practitioners and Australian Primary Health Care Nurses Association reinforced the clinical need and value of embedding pharmacists within general practice.
The session provided a real-life demonstration of cohesion, bringing together representatives from general practice, medicine, nursing and pharmacy.
Exploring how collaborative healthcare efforts can improve patient care, reduce pressure on hospitals and strengthen the primary care workforce, the panel was made up of extraordinary leaders, including:
td_module_mega_menu Object
(
[post] => WP_Post Object
(
[ID] => 32211
[post_author] => 3410
[post_date] => 2026-06-01 11:39:57
[post_date_gmt] => 2026-06-01 01:39:57
[post_content] => Australia's ADHD medicine supply has stabilised – for now. Here’s what pharmacists need to know before the next shortage hits.
After several years of disruption, Australia's ADHD medicine supply has largely recovered. But the shortages of lisdexamfetamine (Vyvanse) in 2023–24 and methylphenidate in 2025 exposed global supply chain vulnerabilities. With diagnosis rates rising, prescribing arrangements expanding and global manufacturing concentrated overseas, pharmacists may once again find themselves managing the fallout from supply interruptions.
[caption id="attachment_32071" align="alignright" width="300"] Yvette Anderson MPS[/caption]
Yvette Anderson MPS – founder of Spectrum Pharmacist, which sets out to bridge the gap for neurodivergent families – tells AP how preparation, collaboration and neuroaffirming care will be essential when the next shortage arrives.
Yvette Anderson MPS[/caption]
Yvette Anderson MPS – founder of Spectrum Pharmacist, which sets out to bridge the gap for neurodivergent families – tells AP how preparation, collaboration and neuroaffirming care will be essential when the next shortage arrives.
A fragile recovery
The shortages that dominated ADHD care over the past 3 years have largely eased, Ms Anderson said.
‘Except for one particular strength of long-acting methylphenidate (30 mg), which is a bit hit and miss.’
However, the situation remains precarious.
Australia relies heavily on overseas manufacturing for ADHD medicines, with production historically concentrated in Ireland and Germany. Supply is also influenced by annual manufacturing quotas established by the United States Drug Enforcement Administration (DEA), which shape global production volumes for controlled substances.
When the Vyvanse patent expired in the United States in 2023, demand for generic lisdexamfetamine surged – rapidly straining international supply chains. More recently, scrutiny of DEA production quotas under the Make America Healthy Again policy framework contributed to a significant reduction in manufacturing, creating flow-on effects internationally.
Due to international uproar, production recommenced. ‘But it's still not probably at the rate and quantity needed globally, so we're always going to sit in a bit of a precarious area,’ Ms Anderson said.
Demand is surging
While supply remains vulnerable, Australia's ADHD treatment landscape is also undergoing significant change.
Initiation of ADHD medicines was largely restricted to paediatricians and psychiatrists. Now, most states and territories have introduced pathways that allow appropriately trained GPs to diagnose and prescribe ADHD medicines.
'A number of GPs in different states have either already been trained, or their state or territory has put laws in place to allow them to be trained to diagnose and prescribe medications,' Ms Anderson said.
Diagnosis rates are also increasing due to changes in diagnosis criteria. More accessible and affordable treatment pathways will help to ensure patients can receive timely access to care.
Importantly, diagnosis does not automatically lead to pharmacological treatment.
'Just because there is potentially going to be an increase in the number of Australians getting diagnosed, that number won't directly correlate to the same number of increasing supply of medication,' Ms Anderson said. 'Medication is only one tiny piece of your management plan, and for some people, medicines aren't the way they want to go, or they don't suit them, or find them effective,'
Nevertheless, more diagnoses and more prescribers are likely to increase demand for medicines supplied through already fragile global supply chains.
Is it possible to get ahead of shortages?
Yes, and no.
Ms Anderson encourages pharmacies to establish systems that allow them to identify and respond to potential disruptions early. This includes monitoring Therapeutic Goods Administration (TGA) medicine shortage alerts.
Once a shortage is identified, pharmacies can use dispensing records to determine which patients may be affected and begin discussions with prescribers before supplies run out.
‘It's a matter of looking at who the prescribers are and reaching out to them and saying, “The TGA has alerted us to this. We know you have a number of patients on this medication. What can we do as a collaborative to support this transition and this shortage period?”’ Ms Anderson said.
As a hospital pharmacist, she was able to pass on information about ADHD medicine shortages to paediatricians – who opened telehealth appointments to turn scripts around quickly.
‘I also communicated with the community pharmacies in my area to ask: Who has stock? Who doesn't? Can we redirect patients?’ Ms Anderson said. ‘Working together as a community, making sure you're embedded in that multidisciplinary team really pays off when there are medication shortages.’
Switching medicines safely
When ADHD medicines are in short supply, patients may need to adjust their medicines; so it’s crucial to be aware of the various formulations, durations of action and release characteristics of different stimulant products.
‘If someone's stabilised on Ritalin, they may go on Ritalin LA or Concerta – but Concerta's duration of action is longer and Ritalin LA's is shorter – so we need to be considering: does this person work through to 6.00 pm? Do we need some immediate release on top of that?’ Ms Anderson said. ‘It's not a straight switch between the medicines.’
The challenges become greater when patients need to move between stimulant classes, such as from lisdexamfetamine to methylphenidate.
‘I've seen people that have quite significant adverse effects after switching to lisdexamfetamine and a couple of days later being unable to function,’ she said. ‘Even though we know someone might be getting a positive effect from stimulants, it doesn't mean it's going to be the same for all stimulants.’
Where stimulant options become unavailable, non-stimulant medicines such as atomoxetine, guanfacine (Intuniv) and clonidine (off-label) can provide alternative management pathways.
‘These non-stimulant options have really good evidence [of efficacy], but they're probably underutilised,' Ms Anderson said. ‘When we do come to another significant shortage, there's only a handful of stimulant medicines – so we need to be able to talk to patients about other options.’
Learn more about supporting patients through ADHD medicine shortages by attending the ADHD care Session at PSA26, held from 31 July to 2 August at the ICC in Sydney.
[post_title] => How to manage ADHD medicine shortages
[post_excerpt] => Australia's ADHD medicine supply has stabilised – for now. Here’s what pharmacists need to know before ADHD medicine shortages strike again.
[post_status] => publish
[comment_status] => open
[ping_status] => open
[post_password] =>
[post_name] => how-to-manage-adhd-medicine-shortages
[to_ping] =>
[pinged] =>
[post_modified] => 2026-06-01 15:15:51
[post_modified_gmt] => 2026-06-01 05:15:51
[post_content_filtered] =>
[post_parent] => 0
[guid] => https://www.australianpharmacist.com.au/?p=32211
[menu_order] => 0
[post_type] => post
[post_mime_type] =>
[comment_count] => 0
[filter] => raw
)
[title_attribute] => How to manage ADHD medicine shortages
[title] => How to manage ADHD medicine shortages
[href] => https://www.australianpharmacist.com.au/how-to-manage-adhd-medicine-shortages/
[module_atts:td_module:private] => Array
(
)
[td_review:protected] => Array
(
)
[is_review:protected] =>
[post_thumb_id:protected] => 32214
[authorType] =>
)
td_module_mega_menu Object
(
[post] => WP_Post Object
(
[ID] => 32207
[post_author] => 12503
[post_date] => 2026-06-01 11:03:42
[post_date_gmt] => 2026-06-01 01:03:42
[post_content] => Two pharmacists were honoured at PSA’s Consultant Pharmacist Conference (CPC26), held from 29–31 May 2026 on the Gold Coast.
Kelly Abbott MPS was awarded 2026 PSA MIMS Consultant Pharmacist of the Year for her outstanding contribution to consultant pharmacy practice and passion for improving patient care throughout her hometown of Gippsland, Victoria.
Tiernan McDonough MPS, based in South Australia, was named 2026 PSA AMH Aged Care Pharmacist of the Year for excellence, leadership, and innovation in aged care pharmacy.
Tireless HMR advocate
Highly regarded across the industry for her work in delivering Home Medicines Reviews, Residential Medication Management Reviews, and providing Quality Use of Medicines (QUM) services, Ms Abbott’s 15 years of service to consultant pharmacy make her a well-deserving winner of this year’s award.
PSA National President, Professor Mark Naunton MPS, said Ms Abbott epitomises many credentialed pharmacists as a passionate and relentless advocate for HMRs, her patients, and the greater credentialed pharmacist workforce.
‘Kelly has continued to be actively involved in grassroots advocacy, notably in recent months during the First Pharmacy Programs Agreement negotiations led by PSA, ensuring the voices of frontline pharmacists and patients were heard,’ he said.
‘Kelly is a loyal, responsive, and reliable contributor to PSA and the Consultant Pharmacist of Australia working groups, while balancing multiple professional roles and caring for her family.
‘Her contributions to PSA and the profession are honourable. We are proud to recognise Kelly as the PSA MIMS Consultant Pharmacist of the Year and celebrate her significant and ongoing impact.’
Aged care champion
Mr McDonough has been recognised for his ongoing dedication and commitment to aged care pharmacy practice through his support for residents and healthcare teams in delivering medication reviews and QUM services.
PSA National President, Professor Mark Naunton MPS, said Mr McDonough embodies the essential role pharmacists play in aged care.
‘Pharmacists working in aged care are critical to ensuring safe and effective medicines use, but Tiernan’s impact extends beyond the aged care sector in which he practices,’ he said.
‘Tiernan has led an important mentoring program that connects pharmacists within the residential aged care profession, to reduce professional isolation and support further workforce development.
‘His work demonstrates his compassion, leadership, and strong drive for improving care for older Australians.’
[post_title] => Consultant pharmacist excellence awarded
[post_excerpt] => Two pharmacists were honoured at PSA’s Consultant Pharmacist Conference (CPC26), held from 29–31 May 2026 on the Gold Coast.
[post_status] => publish
[comment_status] => open
[ping_status] => open
[post_password] =>
[post_name] => top-consultant-pharmacists-honoured-at-cpc26
[to_ping] =>
[pinged] =>
[post_modified] => 2026-06-01 15:18:21
[post_modified_gmt] => 2026-06-01 05:18:21
[post_content_filtered] =>
[post_parent] => 0
[guid] => https://www.australianpharmacist.com.au/?p=32207
[menu_order] => 0
[post_type] => post
[post_mime_type] =>
[comment_count] => 0
[filter] => raw
)
[title_attribute] => Consultant pharmacist excellence awarded
[title] => Consultant pharmacist excellence awarded
[href] => https://www.australianpharmacist.com.au/top-consultant-pharmacists-honoured-at-cpc26/
[module_atts:td_module:private] => Array
(
)
[td_review:protected] => Array
(
)
[is_review:protected] =>
[post_thumb_id:protected] => 32209
[authorType] =>
)
td_module_mega_menu Object
(
[post] => WP_Post Object
(
[ID] => 31968
[post_author] => 8963
[post_date] => 2026-06-01 09:56:44
[post_date_gmt] => 2026-05-31 23:56:44
[post_content] =>
Case scenarioJohn, 62, regularly visits your community pharmacy. Following his referral to a gastroenterologist, he presents to purchase a bowel cleansing kit and asks for advice about his upcoming endoscopy and colonoscopy. John reports a history of difficulty swallowing certain foods and says a family member recently told him that his grandfather had bowel cancer. He is feeling anxious about the procedures and the bowel preparation. |
Endoscopy and colonoscopy are commonly performed gastrointestinal procedures that pharmacists may encounter. Patients may require counselling before and after investigations. Endoscopy refers to procedures that use an endoscope – a thin, flexible tube with a light and camera – to examine internal organs. However, in both clinical and public contexts, particularly in gastroenterology, the term endoscopy is generally used to refer specifically to oesophagoscopy, gastroscopy or duodenoscopy, which involves examining the upper gastrointestinal tract (oesophagus, stomach and duodenum). Colonoscopy is the term used specifically for the examination of the lower gastrointestinal tract (colon/bowel) and includes the entire colon, rectum and terminal ileum. Both procedures are usually performed by gastroenterologists, although colonoscopies may also be performed by colorectal surgeons.1
Learning objectivesAfter reading this article, pharmacists should be able to:
Competency standards (2016) addressed: 1.1, 1.4, 1.5, 2.2, 3.1, 3.5 Accreditation number: PSAAP2606BW Accreditation expiry: 31/05/2028 Accreditation points: 1.5 Group 2 credits |
Both endoscopy and colonoscopy may be used for diagnostic and therapeutic purposes. Standard diagnostic functions include inspection, biopsy, photography and videorecording; diagnostic observations may include focal benign or malignant lesions, mucosal changes, luminal obstruction, motility and strictures. Common therapeutic interventions include polypectomy, dilation of strictures, stent placement, removal of foreign bodies, gastrostomy and treatment of gastrointestinal bleeding.2

EndoscopyOesophagogastroduodenoscopy, more commonly referred to as endoscopy, is a valuable diagnostic procedure in the upper gastrointestinal tract, and is used mainly for detection and surveillance of premalignant conditions and cancers.3 It provides a clear view of mucosal surfaces of the oesophagus, stomach and proximal duodenum.2 It is being increasingly used for gastric cancer and oesophageal adenocarcinoma, the latter having an endoscopically detectable (and potentially curable) precursor lesion, Barrett’s oesophagus.3 Other forms of oesophagitis include eosinophilic oesophagitis, a chronic immune-mediated inflammatory disease of the oesophagus that causes dysphagia, food impaction of the oesophagus, and oesophageal strictures.4 |
Endoscopy with biopsy is mainly used for diagnosing stomach and oesophageal cancers. It is usually performed as day surgery and requires patients to fast for 4–6 hours before the procedure. Before insertion of the endoscope, the throat is sprayed with a local anaesthetic, and a sedative or appropriate anaesthesia is given to the patient. If any suspicious-looking areas are seen, a biopsy may be taken for examination by a pathologist, with results usually available within a few days.1
An endoscopy generally takes about 15 minutes. The patient may have a sore throat afterwards and feel a little bloated. An endoscopic ultrasound is sometimes performed at the same time as a standard endoscopy. This procedure helps determine whether a cancer has spread into the oesophageal wall, nearby tissues or lymph nodes, and is also used to determine whether the patient is a suitable candidate for surgery.1
Endoscopies have a low risk of adverse events. The risk of mortality is rated as 1 in 10,000.5 Possible adverse events include infection, perforation, bleeding, aspiration and cardiopulmonary events.5


ColonoscopyColonoscopy is the main procedure used to investigate or diagnose bowel cancer and is usually performed as day surgery. In 2023–24, there were 691,514 Medicare-subsidised colonoscopies in Australia, of which approximately 20% were repeat procedures performed within 2 years and 10 months of a previous colonoscopy.6 A colonoscope is a tiny camera on a long, flexible tube that is used to examine the entire length of the large bowel. It is inserted into the anus and up into the rectum and colon. Air or carbon dioxide is passed into the colon. Any abnormal tissue can be detected, polyps removed and biopsies taken for further examination. The process usually takes about 20–30 minutes.1 |
Indications for colonoscopy may include:
Australians aged 50–74 years with an average risk of bowel cancer should participate in Australia’s National Bowel Cancer Screening Program. Since 1 July 2024, people aged 45–49 years can also participate on request. A free screening kit is mailed to participants. This is a faecal occult blood test which uses a small stool sample that can be easily collected at home and returned for laboratory testing.1
Before insertion of the colonoscope, an appropriate anaesthesia is given. Patients may feel drowsy or weak prior to and particularly after the colonoscopy, therefore it is recommended that patients do not drive or travel alone. An occasional side effect of a colonoscopy is temporary wind pain and flatulence, especially if air rather than carbon dioxide is passed into the bowel during the procedure.1 More serious but rare complications include perforation (1 in 1,000 risk), infection and risks of sedation and anaesthesia.7
High-quality colonoscopy is required to ensure that cancers and adenomas are not missed, and to avoid adverse patient experiences. The Australian Colonoscopy Clinical Care Standard lists nine key factors, three of which are relevant to pharmacists7:
Informed decision making and patient consent: this is essentially between the patient and gastroenterologist, but patients may seek further advice.
Adequate bowel preparation: bowel cleansing products are generally provided by pharmacies. Reporting and patient follow-up: this is essentially between the patient and gastroenterologist, but patients may seek further advice.
For patients, bowel preparation can be an unpleasant experience.7 However, the importance of adequate bowel preparation and following instructions must be emphasised to patients to ensure that there is a clear view of the mucosa to detect adenomas.8 If the bowel preparation is inadequate, there is not only a lower likelihood of adenoma detection, but it may also result in longer procedures, repeat procedures, more risk to the patient and higher costs.7
Suboptimal bowel preparation has been reported in up to 25% of patients.9 Factors which may affect the bowel preparation include product choice, timing, diet and patient compliance.8
Product selection
Various products are available for bowel cleansing, and there is no consensus on which product is best.7 Over the years, many products have been used, including enemas, mannitol and large-volume solutions. The use of polyethylene glycol 3350 (PEG), also known as macrogol, represented a breakthrough in 1980. This was followed by sodium phosphate preparations in 1990, but these are no longer used because of the risk of renal toxicity.9 Today, many products are based on PEG with the addition of a balanced electrolyte solution and sometimes combined with sodium sulfate, bisacodyl or sodium ascorbate/ascorbic acid.
PEG does not alter the histology of the mucosa and rarely causes electrolyte disturbances.9 The volume to be consumed is an important consideration, with split dosing highly recommended for 4 L solutions (2 L the evening before and 2 L 5–6 hours before the procedure).7,9
The addition of ascorbic acid allows for lower-volume PEG formulations, as its osmotic effect enhances bowel cleansing. These preparations are generally better tolerated due to the reduced volume and improved palatability (typically ~3 L).9
Products based on magnesium citrate combined with sodium picosulfate are also available. These are as effective as PEG-based products and may be better tolerated.9 However, they need to be used with caution in elderly patients, those with renal impairment or heart disease, and those taking certain medications such as diuretics.9 It should be noted that bisacodyl and picosulfate are both converted in the gut to the same active metabolite.9 The decision regarding choice of product is generally made by the gastroenterologist depending on the patient characteristics.
Dietary modification
For a few days before the colonoscopy, patients may be advised to avoid high-fibre foods (such as fruit, vegetables, brown rice, wholegrain pasta, bran, cereals, nuts and seeds). Instead, low-fibre foods (such as white rice, white bread, meat, chicken, fish, yoghurt, cheese, potato and pumpkin) may be recommended.1 Patients may also be advised to drink only fluids, such as water, clear broth, black tea and coffee, and clear fruit juice (without pulp) for 12–24 hours before the procedure to prevent dehydration.1
Special populations and precautions
Patients requiring special care include those with lower gastrointestinal bleeding, chronic kidney disease, inflammatory bowel disease, heart failure, chronic constipation, those who have had previous bowel resection or are pregnant or lactating, as well as hospital inpatients and older people.8 Prevention of dehydration in patients undergoing bowel preparation minimises the risk of severe adverse events.9
Adverse events following bowel preparation are uncommon but may be potentially serious. The product selection decision is an important one because the risk of adverse effects is increased if there is inadequate hydration, inappropriate dosing and inappropriate patient selection.9
Hyponatraemia, because of vomiting or diarrhoea, can develop after taking any colonoscopy preparation. Vomiting may reduce plasma volume and increase thirst. Patients with renal insufficiency, hypothyroidism, mineralocorticoid deficiency, liver cirrhosis, heart failure, or those on diuretics, NSAIDs or ACEIs are at an increased risk of hyponatraemia.9
Improving tolerability
Various methods have been promoted to improve the tolerability and palatability of bowel cleansing preparations. These include chilling the preparation, drinking the preparation through a straw, drinking clear pulp-free juices or approved herbal teas, and chewing gum or various confectioneries concomitantly with the preparation.8

Post colonoscopy syndrome occurs in a third of patients and is more common in females and after lengthy procedures. Symptoms, which include abdominal pain, discomfort and bloating, persist for up to 48 hours following the procedure and may be caused by transient changes in the gut microbiota due to bowel cleansing.8
If a colonoscopy reveals anything of concern, a biopsy is taken for testing by a pathologist. Imaging (such as abdominal scans) may be required. If bowel cancer is diagnosed, scans will determine what stage it is at. Tissue taken at the time of colonoscopy may be tested for genetic changes in the cancer cells, which can guide the choice of treatment.1
Pharmacists are frequently involved in supporting patients undergoing endoscopy and colonoscopy, particularly in the community setting. Patients may seek reassurance about the purpose, safety and expected after-effects of these procedures. Providing clear, evidence-based information can help reduce anxiety. Pharmacists can provide advice regarding colonoscopy preparation by counselling on correct use of bowel cleansing products, split dosing, diet and hydration. They should identify patients at high risk of complications, including older adults, those with renal or cardiac impairment, or those taking diuretics, NSAIDs or ACE inhibitors, and provide tailored advice. Following the procedure, pharmacists can reassure patients about common after effects.
Endoscopy and colonoscopy are widely performed diagnostic and therapeutic procedures that play a central role in the detection and management of gastrointestinal disease, including malignancy. Although generally safe, both procedures require appropriate patient education and awareness of potential adverse effects.
Pharmacists are well positioned to support patients before and after these procedures by providing counselling, identifying individuals at increased risk of complications, and promoting effective bowel preparation for colonoscopy. Through proactive engagement, pharmacists can contribute to improved patient experience and procedural outcomes.
Case scenario continuedYou explain the purpose of both procedures and reassure John that they are commonly performed and generally low risk. You counsel him on how to use the bowel cleansing kit, emphasising the importance of following the instructions carefully. You also discuss the dietary and fluid modifications required in the days leading up to the colonoscopy and provide tips to improve tolerability of the preparation. Following the procedures, John returns to the pharmacy with a prescription for budesonide 1 mg orally dispersible tablets. He explains that he has been diagnosed with eosinophilic oesophagitis and that his oesophagus has been dilated. He also mentions that two polyps were removed from his colon. You reassure John and provide counselling on the correct use of his new medicine. |
[cpd_submit_answer_button]
DR Barry Werth PhD, BPharm (Hons), MBA, MPS is a pharmacist with over 50 years’ experience in the Australian pharmaceutical industry, holding a PhD in epidemiology/pharmacoepidemiology, an honours degree in pharmacy and an MBA. He is a Research Affiliate of the University of Sydney and holds senior advisory roles with MND Australia, alongside ongoing consultancy work for pharmaceutical companies.
Rose-marie Pennisi (she/her), BPharm, MBA, MPS
Cancer Council Australia. Cancer Council. Sydney: Cancer Council Australia; 2025. At: www.cancer.org.au
Early DS, Ben-Menachem T, Decker GA, et al. Appropriate use of GI endoscopy. Gastrointest Endosc. 2012;75(6):1127–31.
Januszewicz W, Kaminski MF. Quality indicators in diagnostic upper gastrointestinal endoscopy. Ther Adv Gastroenterol. 2020;13:1756284820916693.
Muir A, Falk GW. Eosinophilic esophagitis: a review. JAMA. 2021;326(13):1310–8.
Ben-Menachem T, Decker GA, Early DS, et al. Adverse events of upper GI endoscopy. Gastrointest Endosc. 2012;76(4):707–18.
Australian Commission on Safety and Quality in Health Care. Atlas focus report: colonoscopy. Sydney: Australian Commission on Safety and Quality in Health Care; 2025. At: www.safetyandquality.gov.au
Duggan A, Skinner IJ, Bhasale AL. All colonoscopies are not created equal: why Australia now has a clinical care standard for colonoscopy. Med J Aust. 2018;209(10):427–30.
Di Leo M, Iannone A, Arena M, et al. Novel frontiers of agents for bowel cleansing for colonoscopy. World J Gastroenterol. 2021;27(45):7748.
Adamcewicz M, Bearelly D, Porat G, et al. Mechanism of action and toxicities of purgatives used for colonoscopy preparation. Expert Opin Drug Metab Toxicol. 2011;7(1):89–101.
td_module_mega_menu Object
(
[post] => WP_Post Object
(
[ID] => 32230
[post_author] => 3410
[post_date] => 2026-06-03 13:02:08
[post_date_gmt] => 2026-06-03 03:02:08
[post_content] => The combination of an award wage rise and pay equity increase is set to deliver a meaningful lift in pharmacist remuneration.
Pharmacists are in line for a pay boost. AP investigates the drivers set to lift wages from 1 July.
Strong award wage increase
Pharmacists employed by the Pharmacy Industry Award 2020 will experience their largest annual wage rise in years. The Fair Work Commission (FWC) announced the 2026 Annual Wage Review decision yesterday (2 June), increasing modern award minimum wages by 4.75% from 1 July.
The increase is lower than the 6% wage rise supported by unions, but higher than the rate proposed by employer and business groups.
Along with the Australian Industry Group and the Australian Chamber of Commerce, the Pharmacy Guild of Australia advocated for a 3.5% wage increase, advising that this was the highest level businesses could accommodate.
However, the FWC’s decision was based on the challenges the nation is facing, including Reserve Bank interest rate tightening and the conflict in the Middle East.
In its decision, the FWC noted ‘ regrettably, … it is not practicable in the current uncertain circumstances to award a real wage increase for employees reliant on modern award wage rates that would be sufficient to close the real wage gap entirely’ compared to the post-pandemic inflation spike after July 2021.
‘I consistently hear from our members that remuneration is their number one concern, particularly given cost-of-living pressures,’ said PSA National President Professor Mark Naunton MPS. ‘For many community pharmacists, who make up a significant proportion of our membership, the award increase feels long overdue.’
Year 2 of the gender undervaluation correction
In 2025, the FWC’s Expert Panel for pay equity in the care and community sector determined that pharmacists have been subject to a total of 14.1% gender-based undervaluation.
‘Last year's gender discrimination determination was so important in addressing longstanding discrimination in the award rate for pharmacists, which was found to be directly linked to the high proportion of the workforce that is female,’ Prof Naunton said.
The Expert Panel issued a determination that there will be a total increase in the minimum wage rates of 14.1% over 3 years, with the second increase set to take place on 30 June 2026.
As AP pointed out last year, not all pharmacists will see a direct pay rise. The award sets a remuneration level, and Fair Work data indicates around 12.7% of pharmacists are paid at award rates only – though the proportion is likely higher in community pharmacy, where collective agreements are less common than in the hospital sector.
While pharmacy interns will see another pay rise, pharmacy assistants and pharmacy students are not included, as their rates were not found to be undervalued.
‘We welcome the next stage of the gender discrimination increase to the Pharmacy Industry Award, which will take effect from 1 July, in addition to the 4.75% increase from the annual wage review,’ Prof Naunton added.
What will the new award hourly rate be?
The exact updated pay rates will be confirmed when the FWC publishes new pay guides ahead of 1 July.
And while the wage increase is a relief for many pharmacists, Prof Naunton said PSA also recognised that there are other members who are being left further behind financially.
‘We continue to fight to increase remuneration for pharmacists who provide medication review services, who have had no fee increase in 7 years,’ he added.
[post_title] => Pharmacists in line for pay increase
[post_excerpt] => The combination of an award wage rise and pay equity increase is set to deliver a meaningful lift in pharmacist remuneration.
[post_status] => publish
[comment_status] => open
[ping_status] => open
[post_password] =>
[post_name] => pharmacists-in-line-for-pay-increase
[to_ping] =>
[pinged] =>
[post_modified] => 2026-06-03 15:41:29
[post_modified_gmt] => 2026-06-03 05:41:29
[post_content_filtered] =>
[post_parent] => 0
[guid] => https://www.australianpharmacist.com.au/?p=32230
[menu_order] => 0
[post_type] => post
[post_mime_type] =>
[comment_count] => 0
[filter] => raw
)
[title_attribute] => Pharmacists in line for pay increase
[title] => Pharmacists in line for pay increase
[href] => https://www.australianpharmacist.com.au/pharmacists-in-line-for-pay-increase/
[module_atts:td_module:private] => Array
(
)
[td_review:protected] => Array
(
)
[is_review:protected] =>
[post_thumb_id:protected] => 32232
[authorType] =>
)
td_module_mega_menu Object
(
[post] => WP_Post Object
(
[ID] => 32217
[post_author] => 12503
[post_date] => 2026-06-01 12:04:52
[post_date_gmt] => 2026-06-01 02:04:52
[post_content] => Four of Australia’s leading health peak bodies have united in calling for investment in general practice pharmacists to make medicine use safer and strengthen multidisciplinary care.
Speaking at an inter-professional panel session at the 2026 GP Pharmacist Symposium today on the Gold Coast, presidents from PSA, Australian Medical Association, Royal Australian College of General Practitioners and Australian Primary Health Care Nurses Association reinforced the clinical need and value of embedding pharmacists within general practice.
The session provided a real-life demonstration of cohesion, bringing together representatives from general practice, medicine, nursing and pharmacy.
Exploring how collaborative healthcare efforts can improve patient care, reduce pressure on hospitals and strengthen the primary care workforce, the panel was made up of extraordinary leaders, including:
td_module_mega_menu Object
(
[post] => WP_Post Object
(
[ID] => 32211
[post_author] => 3410
[post_date] => 2026-06-01 11:39:57
[post_date_gmt] => 2026-06-01 01:39:57
[post_content] => Australia's ADHD medicine supply has stabilised – for now. Here’s what pharmacists need to know before the next shortage hits.
After several years of disruption, Australia's ADHD medicine supply has largely recovered. But the shortages of lisdexamfetamine (Vyvanse) in 2023–24 and methylphenidate in 2025 exposed global supply chain vulnerabilities. With diagnosis rates rising, prescribing arrangements expanding and global manufacturing concentrated overseas, pharmacists may once again find themselves managing the fallout from supply interruptions.
[caption id="attachment_32071" align="alignright" width="300"] Yvette Anderson MPS[/caption]
Yvette Anderson MPS – founder of Spectrum Pharmacist, which sets out to bridge the gap for neurodivergent families – tells AP how preparation, collaboration and neuroaffirming care will be essential when the next shortage arrives.
Yvette Anderson MPS[/caption]
Yvette Anderson MPS – founder of Spectrum Pharmacist, which sets out to bridge the gap for neurodivergent families – tells AP how preparation, collaboration and neuroaffirming care will be essential when the next shortage arrives.
A fragile recovery
The shortages that dominated ADHD care over the past 3 years have largely eased, Ms Anderson said.
‘Except for one particular strength of long-acting methylphenidate (30 mg), which is a bit hit and miss.’
However, the situation remains precarious.
Australia relies heavily on overseas manufacturing for ADHD medicines, with production historically concentrated in Ireland and Germany. Supply is also influenced by annual manufacturing quotas established by the United States Drug Enforcement Administration (DEA), which shape global production volumes for controlled substances.
When the Vyvanse patent expired in the United States in 2023, demand for generic lisdexamfetamine surged – rapidly straining international supply chains. More recently, scrutiny of DEA production quotas under the Make America Healthy Again policy framework contributed to a significant reduction in manufacturing, creating flow-on effects internationally.
Due to international uproar, production recommenced. ‘But it's still not probably at the rate and quantity needed globally, so we're always going to sit in a bit of a precarious area,’ Ms Anderson said.
Demand is surging
While supply remains vulnerable, Australia's ADHD treatment landscape is also undergoing significant change.
Initiation of ADHD medicines was largely restricted to paediatricians and psychiatrists. Now, most states and territories have introduced pathways that allow appropriately trained GPs to diagnose and prescribe ADHD medicines.
'A number of GPs in different states have either already been trained, or their state or territory has put laws in place to allow them to be trained to diagnose and prescribe medications,' Ms Anderson said.
Diagnosis rates are also increasing due to changes in diagnosis criteria. More accessible and affordable treatment pathways will help to ensure patients can receive timely access to care.
Importantly, diagnosis does not automatically lead to pharmacological treatment.
'Just because there is potentially going to be an increase in the number of Australians getting diagnosed, that number won't directly correlate to the same number of increasing supply of medication,' Ms Anderson said. 'Medication is only one tiny piece of your management plan, and for some people, medicines aren't the way they want to go, or they don't suit them, or find them effective,'
Nevertheless, more diagnoses and more prescribers are likely to increase demand for medicines supplied through already fragile global supply chains.
Is it possible to get ahead of shortages?
Yes, and no.
Ms Anderson encourages pharmacies to establish systems that allow them to identify and respond to potential disruptions early. This includes monitoring Therapeutic Goods Administration (TGA) medicine shortage alerts.
Once a shortage is identified, pharmacies can use dispensing records to determine which patients may be affected and begin discussions with prescribers before supplies run out.
‘It's a matter of looking at who the prescribers are and reaching out to them and saying, “The TGA has alerted us to this. We know you have a number of patients on this medication. What can we do as a collaborative to support this transition and this shortage period?”’ Ms Anderson said.
As a hospital pharmacist, she was able to pass on information about ADHD medicine shortages to paediatricians – who opened telehealth appointments to turn scripts around quickly.
‘I also communicated with the community pharmacies in my area to ask: Who has stock? Who doesn't? Can we redirect patients?’ Ms Anderson said. ‘Working together as a community, making sure you're embedded in that multidisciplinary team really pays off when there are medication shortages.’
Switching medicines safely
When ADHD medicines are in short supply, patients may need to adjust their medicines; so it’s crucial to be aware of the various formulations, durations of action and release characteristics of different stimulant products.
‘If someone's stabilised on Ritalin, they may go on Ritalin LA or Concerta – but Concerta's duration of action is longer and Ritalin LA's is shorter – so we need to be considering: does this person work through to 6.00 pm? Do we need some immediate release on top of that?’ Ms Anderson said. ‘It's not a straight switch between the medicines.’
The challenges become greater when patients need to move between stimulant classes, such as from lisdexamfetamine to methylphenidate.
‘I've seen people that have quite significant adverse effects after switching to lisdexamfetamine and a couple of days later being unable to function,’ she said. ‘Even though we know someone might be getting a positive effect from stimulants, it doesn't mean it's going to be the same for all stimulants.’
Where stimulant options become unavailable, non-stimulant medicines such as atomoxetine, guanfacine (Intuniv) and clonidine (off-label) can provide alternative management pathways.
‘These non-stimulant options have really good evidence [of efficacy], but they're probably underutilised,' Ms Anderson said. ‘When we do come to another significant shortage, there's only a handful of stimulant medicines – so we need to be able to talk to patients about other options.’
Learn more about supporting patients through ADHD medicine shortages by attending the ADHD care Session at PSA26, held from 31 July to 2 August at the ICC in Sydney.
[post_title] => How to manage ADHD medicine shortages
[post_excerpt] => Australia's ADHD medicine supply has stabilised – for now. Here’s what pharmacists need to know before ADHD medicine shortages strike again.
[post_status] => publish
[comment_status] => open
[ping_status] => open
[post_password] =>
[post_name] => how-to-manage-adhd-medicine-shortages
[to_ping] =>
[pinged] =>
[post_modified] => 2026-06-01 15:15:51
[post_modified_gmt] => 2026-06-01 05:15:51
[post_content_filtered] =>
[post_parent] => 0
[guid] => https://www.australianpharmacist.com.au/?p=32211
[menu_order] => 0
[post_type] => post
[post_mime_type] =>
[comment_count] => 0
[filter] => raw
)
[title_attribute] => How to manage ADHD medicine shortages
[title] => How to manage ADHD medicine shortages
[href] => https://www.australianpharmacist.com.au/how-to-manage-adhd-medicine-shortages/
[module_atts:td_module:private] => Array
(
)
[td_review:protected] => Array
(
)
[is_review:protected] =>
[post_thumb_id:protected] => 32214
[authorType] =>
)
td_module_mega_menu Object
(
[post] => WP_Post Object
(
[ID] => 32207
[post_author] => 12503
[post_date] => 2026-06-01 11:03:42
[post_date_gmt] => 2026-06-01 01:03:42
[post_content] => Two pharmacists were honoured at PSA’s Consultant Pharmacist Conference (CPC26), held from 29–31 May 2026 on the Gold Coast.
Kelly Abbott MPS was awarded 2026 PSA MIMS Consultant Pharmacist of the Year for her outstanding contribution to consultant pharmacy practice and passion for improving patient care throughout her hometown of Gippsland, Victoria.
Tiernan McDonough MPS, based in South Australia, was named 2026 PSA AMH Aged Care Pharmacist of the Year for excellence, leadership, and innovation in aged care pharmacy.
Tireless HMR advocate
Highly regarded across the industry for her work in delivering Home Medicines Reviews, Residential Medication Management Reviews, and providing Quality Use of Medicines (QUM) services, Ms Abbott’s 15 years of service to consultant pharmacy make her a well-deserving winner of this year’s award.
PSA National President, Professor Mark Naunton MPS, said Ms Abbott epitomises many credentialed pharmacists as a passionate and relentless advocate for HMRs, her patients, and the greater credentialed pharmacist workforce.
‘Kelly has continued to be actively involved in grassroots advocacy, notably in recent months during the First Pharmacy Programs Agreement negotiations led by PSA, ensuring the voices of frontline pharmacists and patients were heard,’ he said.
‘Kelly is a loyal, responsive, and reliable contributor to PSA and the Consultant Pharmacist of Australia working groups, while balancing multiple professional roles and caring for her family.
‘Her contributions to PSA and the profession are honourable. We are proud to recognise Kelly as the PSA MIMS Consultant Pharmacist of the Year and celebrate her significant and ongoing impact.’
Aged care champion
Mr McDonough has been recognised for his ongoing dedication and commitment to aged care pharmacy practice through his support for residents and healthcare teams in delivering medication reviews and QUM services.
PSA National President, Professor Mark Naunton MPS, said Mr McDonough embodies the essential role pharmacists play in aged care.
‘Pharmacists working in aged care are critical to ensuring safe and effective medicines use, but Tiernan’s impact extends beyond the aged care sector in which he practices,’ he said.
‘Tiernan has led an important mentoring program that connects pharmacists within the residential aged care profession, to reduce professional isolation and support further workforce development.
‘His work demonstrates his compassion, leadership, and strong drive for improving care for older Australians.’
[post_title] => Consultant pharmacist excellence awarded
[post_excerpt] => Two pharmacists were honoured at PSA’s Consultant Pharmacist Conference (CPC26), held from 29–31 May 2026 on the Gold Coast.
[post_status] => publish
[comment_status] => open
[ping_status] => open
[post_password] =>
[post_name] => top-consultant-pharmacists-honoured-at-cpc26
[to_ping] =>
[pinged] =>
[post_modified] => 2026-06-01 15:18:21
[post_modified_gmt] => 2026-06-01 05:18:21
[post_content_filtered] =>
[post_parent] => 0
[guid] => https://www.australianpharmacist.com.au/?p=32207
[menu_order] => 0
[post_type] => post
[post_mime_type] =>
[comment_count] => 0
[filter] => raw
)
[title_attribute] => Consultant pharmacist excellence awarded
[title] => Consultant pharmacist excellence awarded
[href] => https://www.australianpharmacist.com.au/top-consultant-pharmacists-honoured-at-cpc26/
[module_atts:td_module:private] => Array
(
)
[td_review:protected] => Array
(
)
[is_review:protected] =>
[post_thumb_id:protected] => 32209
[authorType] =>
)
td_module_mega_menu Object
(
[post] => WP_Post Object
(
[ID] => 31968
[post_author] => 8963
[post_date] => 2026-06-01 09:56:44
[post_date_gmt] => 2026-05-31 23:56:44
[post_content] =>
Case scenarioJohn, 62, regularly visits your community pharmacy. Following his referral to a gastroenterologist, he presents to purchase a bowel cleansing kit and asks for advice about his upcoming endoscopy and colonoscopy. John reports a history of difficulty swallowing certain foods and says a family member recently told him that his grandfather had bowel cancer. He is feeling anxious about the procedures and the bowel preparation. |
Endoscopy and colonoscopy are commonly performed gastrointestinal procedures that pharmacists may encounter. Patients may require counselling before and after investigations. Endoscopy refers to procedures that use an endoscope – a thin, flexible tube with a light and camera – to examine internal organs. However, in both clinical and public contexts, particularly in gastroenterology, the term endoscopy is generally used to refer specifically to oesophagoscopy, gastroscopy or duodenoscopy, which involves examining the upper gastrointestinal tract (oesophagus, stomach and duodenum). Colonoscopy is the term used specifically for the examination of the lower gastrointestinal tract (colon/bowel) and includes the entire colon, rectum and terminal ileum. Both procedures are usually performed by gastroenterologists, although colonoscopies may also be performed by colorectal surgeons.1
Learning objectivesAfter reading this article, pharmacists should be able to:
Competency standards (2016) addressed: 1.1, 1.4, 1.5, 2.2, 3.1, 3.5 Accreditation number: PSAAP2606BW Accreditation expiry: 31/05/2028 Accreditation points: 1.5 Group 2 credits |
Both endoscopy and colonoscopy may be used for diagnostic and therapeutic purposes. Standard diagnostic functions include inspection, biopsy, photography and videorecording; diagnostic observations may include focal benign or malignant lesions, mucosal changes, luminal obstruction, motility and strictures. Common therapeutic interventions include polypectomy, dilation of strictures, stent placement, removal of foreign bodies, gastrostomy and treatment of gastrointestinal bleeding.2

EndoscopyOesophagogastroduodenoscopy, more commonly referred to as endoscopy, is a valuable diagnostic procedure in the upper gastrointestinal tract, and is used mainly for detection and surveillance of premalignant conditions and cancers.3 It provides a clear view of mucosal surfaces of the oesophagus, stomach and proximal duodenum.2 It is being increasingly used for gastric cancer and oesophageal adenocarcinoma, the latter having an endoscopically detectable (and potentially curable) precursor lesion, Barrett’s oesophagus.3 Other forms of oesophagitis include eosinophilic oesophagitis, a chronic immune-mediated inflammatory disease of the oesophagus that causes dysphagia, food impaction of the oesophagus, and oesophageal strictures.4 |
Endoscopy with biopsy is mainly used for diagnosing stomach and oesophageal cancers. It is usually performed as day surgery and requires patients to fast for 4–6 hours before the procedure. Before insertion of the endoscope, the throat is sprayed with a local anaesthetic, and a sedative or appropriate anaesthesia is given to the patient. If any suspicious-looking areas are seen, a biopsy may be taken for examination by a pathologist, with results usually available within a few days.1
An endoscopy generally takes about 15 minutes. The patient may have a sore throat afterwards and feel a little bloated. An endoscopic ultrasound is sometimes performed at the same time as a standard endoscopy. This procedure helps determine whether a cancer has spread into the oesophageal wall, nearby tissues or lymph nodes, and is also used to determine whether the patient is a suitable candidate for surgery.1
Endoscopies have a low risk of adverse events. The risk of mortality is rated as 1 in 10,000.5 Possible adverse events include infection, perforation, bleeding, aspiration and cardiopulmonary events.5


ColonoscopyColonoscopy is the main procedure used to investigate or diagnose bowel cancer and is usually performed as day surgery. In 2023–24, there were 691,514 Medicare-subsidised colonoscopies in Australia, of which approximately 20% were repeat procedures performed within 2 years and 10 months of a previous colonoscopy.6 A colonoscope is a tiny camera on a long, flexible tube that is used to examine the entire length of the large bowel. It is inserted into the anus and up into the rectum and colon. Air or carbon dioxide is passed into the colon. Any abnormal tissue can be detected, polyps removed and biopsies taken for further examination. The process usually takes about 20–30 minutes.1 |
Indications for colonoscopy may include:
Australians aged 50–74 years with an average risk of bowel cancer should participate in Australia’s National Bowel Cancer Screening Program. Since 1 July 2024, people aged 45–49 years can also participate on request. A free screening kit is mailed to participants. This is a faecal occult blood test which uses a small stool sample that can be easily collected at home and returned for laboratory testing.1
Before insertion of the colonoscope, an appropriate anaesthesia is given. Patients may feel drowsy or weak prior to and particularly after the colonoscopy, therefore it is recommended that patients do not drive or travel alone. An occasional side effect of a colonoscopy is temporary wind pain and flatulence, especially if air rather than carbon dioxide is passed into the bowel during the procedure.1 More serious but rare complications include perforation (1 in 1,000 risk), infection and risks of sedation and anaesthesia.7
High-quality colonoscopy is required to ensure that cancers and adenomas are not missed, and to avoid adverse patient experiences. The Australian Colonoscopy Clinical Care Standard lists nine key factors, three of which are relevant to pharmacists7:
Informed decision making and patient consent: this is essentially between the patient and gastroenterologist, but patients may seek further advice.
Adequate bowel preparation: bowel cleansing products are generally provided by pharmacies. Reporting and patient follow-up: this is essentially between the patient and gastroenterologist, but patients may seek further advice.
For patients, bowel preparation can be an unpleasant experience.7 However, the importance of adequate bowel preparation and following instructions must be emphasised to patients to ensure that there is a clear view of the mucosa to detect adenomas.8 If the bowel preparation is inadequate, there is not only a lower likelihood of adenoma detection, but it may also result in longer procedures, repeat procedures, more risk to the patient and higher costs.7
Suboptimal bowel preparation has been reported in up to 25% of patients.9 Factors which may affect the bowel preparation include product choice, timing, diet and patient compliance.8
Product selection
Various products are available for bowel cleansing, and there is no consensus on which product is best.7 Over the years, many products have been used, including enemas, mannitol and large-volume solutions. The use of polyethylene glycol 3350 (PEG), also known as macrogol, represented a breakthrough in 1980. This was followed by sodium phosphate preparations in 1990, but these are no longer used because of the risk of renal toxicity.9 Today, many products are based on PEG with the addition of a balanced electrolyte solution and sometimes combined with sodium sulfate, bisacodyl or sodium ascorbate/ascorbic acid.
PEG does not alter the histology of the mucosa and rarely causes electrolyte disturbances.9 The volume to be consumed is an important consideration, with split dosing highly recommended for 4 L solutions (2 L the evening before and 2 L 5–6 hours before the procedure).7,9
The addition of ascorbic acid allows for lower-volume PEG formulations, as its osmotic effect enhances bowel cleansing. These preparations are generally better tolerated due to the reduced volume and improved palatability (typically ~3 L).9
Products based on magnesium citrate combined with sodium picosulfate are also available. These are as effective as PEG-based products and may be better tolerated.9 However, they need to be used with caution in elderly patients, those with renal impairment or heart disease, and those taking certain medications such as diuretics.9 It should be noted that bisacodyl and picosulfate are both converted in the gut to the same active metabolite.9 The decision regarding choice of product is generally made by the gastroenterologist depending on the patient characteristics.
Dietary modification
For a few days before the colonoscopy, patients may be advised to avoid high-fibre foods (such as fruit, vegetables, brown rice, wholegrain pasta, bran, cereals, nuts and seeds). Instead, low-fibre foods (such as white rice, white bread, meat, chicken, fish, yoghurt, cheese, potato and pumpkin) may be recommended.1 Patients may also be advised to drink only fluids, such as water, clear broth, black tea and coffee, and clear fruit juice (without pulp) for 12–24 hours before the procedure to prevent dehydration.1
Special populations and precautions
Patients requiring special care include those with lower gastrointestinal bleeding, chronic kidney disease, inflammatory bowel disease, heart failure, chronic constipation, those who have had previous bowel resection or are pregnant or lactating, as well as hospital inpatients and older people.8 Prevention of dehydration in patients undergoing bowel preparation minimises the risk of severe adverse events.9
Adverse events following bowel preparation are uncommon but may be potentially serious. The product selection decision is an important one because the risk of adverse effects is increased if there is inadequate hydration, inappropriate dosing and inappropriate patient selection.9
Hyponatraemia, because of vomiting or diarrhoea, can develop after taking any colonoscopy preparation. Vomiting may reduce plasma volume and increase thirst. Patients with renal insufficiency, hypothyroidism, mineralocorticoid deficiency, liver cirrhosis, heart failure, or those on diuretics, NSAIDs or ACEIs are at an increased risk of hyponatraemia.9
Improving tolerability
Various methods have been promoted to improve the tolerability and palatability of bowel cleansing preparations. These include chilling the preparation, drinking the preparation through a straw, drinking clear pulp-free juices or approved herbal teas, and chewing gum or various confectioneries concomitantly with the preparation.8

Post colonoscopy syndrome occurs in a third of patients and is more common in females and after lengthy procedures. Symptoms, which include abdominal pain, discomfort and bloating, persist for up to 48 hours following the procedure and may be caused by transient changes in the gut microbiota due to bowel cleansing.8
If a colonoscopy reveals anything of concern, a biopsy is taken for testing by a pathologist. Imaging (such as abdominal scans) may be required. If bowel cancer is diagnosed, scans will determine what stage it is at. Tissue taken at the time of colonoscopy may be tested for genetic changes in the cancer cells, which can guide the choice of treatment.1
Pharmacists are frequently involved in supporting patients undergoing endoscopy and colonoscopy, particularly in the community setting. Patients may seek reassurance about the purpose, safety and expected after-effects of these procedures. Providing clear, evidence-based information can help reduce anxiety. Pharmacists can provide advice regarding colonoscopy preparation by counselling on correct use of bowel cleansing products, split dosing, diet and hydration. They should identify patients at high risk of complications, including older adults, those with renal or cardiac impairment, or those taking diuretics, NSAIDs or ACE inhibitors, and provide tailored advice. Following the procedure, pharmacists can reassure patients about common after effects.
Endoscopy and colonoscopy are widely performed diagnostic and therapeutic procedures that play a central role in the detection and management of gastrointestinal disease, including malignancy. Although generally safe, both procedures require appropriate patient education and awareness of potential adverse effects.
Pharmacists are well positioned to support patients before and after these procedures by providing counselling, identifying individuals at increased risk of complications, and promoting effective bowel preparation for colonoscopy. Through proactive engagement, pharmacists can contribute to improved patient experience and procedural outcomes.
Case scenario continuedYou explain the purpose of both procedures and reassure John that they are commonly performed and generally low risk. You counsel him on how to use the bowel cleansing kit, emphasising the importance of following the instructions carefully. You also discuss the dietary and fluid modifications required in the days leading up to the colonoscopy and provide tips to improve tolerability of the preparation. Following the procedures, John returns to the pharmacy with a prescription for budesonide 1 mg orally dispersible tablets. He explains that he has been diagnosed with eosinophilic oesophagitis and that his oesophagus has been dilated. He also mentions that two polyps were removed from his colon. You reassure John and provide counselling on the correct use of his new medicine. |
[cpd_submit_answer_button]
DR Barry Werth PhD, BPharm (Hons), MBA, MPS is a pharmacist with over 50 years’ experience in the Australian pharmaceutical industry, holding a PhD in epidemiology/pharmacoepidemiology, an honours degree in pharmacy and an MBA. He is a Research Affiliate of the University of Sydney and holds senior advisory roles with MND Australia, alongside ongoing consultancy work for pharmaceutical companies.
Rose-marie Pennisi (she/her), BPharm, MBA, MPS
Cancer Council Australia. Cancer Council. Sydney: Cancer Council Australia; 2025. At: www.cancer.org.au
Early DS, Ben-Menachem T, Decker GA, et al. Appropriate use of GI endoscopy. Gastrointest Endosc. 2012;75(6):1127–31.
Januszewicz W, Kaminski MF. Quality indicators in diagnostic upper gastrointestinal endoscopy. Ther Adv Gastroenterol. 2020;13:1756284820916693.
Muir A, Falk GW. Eosinophilic esophagitis: a review. JAMA. 2021;326(13):1310–8.
Ben-Menachem T, Decker GA, Early DS, et al. Adverse events of upper GI endoscopy. Gastrointest Endosc. 2012;76(4):707–18.
Australian Commission on Safety and Quality in Health Care. Atlas focus report: colonoscopy. Sydney: Australian Commission on Safety and Quality in Health Care; 2025. At: www.safetyandquality.gov.au
Duggan A, Skinner IJ, Bhasale AL. All colonoscopies are not created equal: why Australia now has a clinical care standard for colonoscopy. Med J Aust. 2018;209(10):427–30.
Di Leo M, Iannone A, Arena M, et al. Novel frontiers of agents for bowel cleansing for colonoscopy. World J Gastroenterol. 2021;27(45):7748.
Adamcewicz M, Bearelly D, Porat G, et al. Mechanism of action and toxicities of purgatives used for colonoscopy preparation. Expert Opin Drug Metab Toxicol. 2011;7(1):89–101.
td_module_mega_menu Object
(
[post] => WP_Post Object
(
[ID] => 32230
[post_author] => 3410
[post_date] => 2026-06-03 13:02:08
[post_date_gmt] => 2026-06-03 03:02:08
[post_content] => The combination of an award wage rise and pay equity increase is set to deliver a meaningful lift in pharmacist remuneration.
Pharmacists are in line for a pay boost. AP investigates the drivers set to lift wages from 1 July.
Strong award wage increase
Pharmacists employed by the Pharmacy Industry Award 2020 will experience their largest annual wage rise in years. The Fair Work Commission (FWC) announced the 2026 Annual Wage Review decision yesterday (2 June), increasing modern award minimum wages by 4.75% from 1 July.
The increase is lower than the 6% wage rise supported by unions, but higher than the rate proposed by employer and business groups.
Along with the Australian Industry Group and the Australian Chamber of Commerce, the Pharmacy Guild of Australia advocated for a 3.5% wage increase, advising that this was the highest level businesses could accommodate.
However, the FWC’s decision was based on the challenges the nation is facing, including Reserve Bank interest rate tightening and the conflict in the Middle East.
In its decision, the FWC noted ‘ regrettably, … it is not practicable in the current uncertain circumstances to award a real wage increase for employees reliant on modern award wage rates that would be sufficient to close the real wage gap entirely’ compared to the post-pandemic inflation spike after July 2021.
‘I consistently hear from our members that remuneration is their number one concern, particularly given cost-of-living pressures,’ said PSA National President Professor Mark Naunton MPS. ‘For many community pharmacists, who make up a significant proportion of our membership, the award increase feels long overdue.’
Year 2 of the gender undervaluation correction
In 2025, the FWC’s Expert Panel for pay equity in the care and community sector determined that pharmacists have been subject to a total of 14.1% gender-based undervaluation.
‘Last year's gender discrimination determination was so important in addressing longstanding discrimination in the award rate for pharmacists, which was found to be directly linked to the high proportion of the workforce that is female,’ Prof Naunton said.
The Expert Panel issued a determination that there will be a total increase in the minimum wage rates of 14.1% over 3 years, with the second increase set to take place on 30 June 2026.
As AP pointed out last year, not all pharmacists will see a direct pay rise. The award sets a remuneration level, and Fair Work data indicates around 12.7% of pharmacists are paid at award rates only – though the proportion is likely higher in community pharmacy, where collective agreements are less common than in the hospital sector.
While pharmacy interns will see another pay rise, pharmacy assistants and pharmacy students are not included, as their rates were not found to be undervalued.
‘We welcome the next stage of the gender discrimination increase to the Pharmacy Industry Award, which will take effect from 1 July, in addition to the 4.75% increase from the annual wage review,’ Prof Naunton added.
What will the new award hourly rate be?
The exact updated pay rates will be confirmed when the FWC publishes new pay guides ahead of 1 July.
And while the wage increase is a relief for many pharmacists, Prof Naunton said PSA also recognised that there are other members who are being left further behind financially.
‘We continue to fight to increase remuneration for pharmacists who provide medication review services, who have had no fee increase in 7 years,’ he added.
[post_title] => Pharmacists in line for pay increase
[post_excerpt] => The combination of an award wage rise and pay equity increase is set to deliver a meaningful lift in pharmacist remuneration.
[post_status] => publish
[comment_status] => open
[ping_status] => open
[post_password] =>
[post_name] => pharmacists-in-line-for-pay-increase
[to_ping] =>
[pinged] =>
[post_modified] => 2026-06-03 15:41:29
[post_modified_gmt] => 2026-06-03 05:41:29
[post_content_filtered] =>
[post_parent] => 0
[guid] => https://www.australianpharmacist.com.au/?p=32230
[menu_order] => 0
[post_type] => post
[post_mime_type] =>
[comment_count] => 0
[filter] => raw
)
[title_attribute] => Pharmacists in line for pay increase
[title] => Pharmacists in line for pay increase
[href] => https://www.australianpharmacist.com.au/pharmacists-in-line-for-pay-increase/
[module_atts:td_module:private] => Array
(
)
[td_review:protected] => Array
(
)
[is_review:protected] =>
[post_thumb_id:protected] => 32232
[authorType] =>
)
td_module_mega_menu Object
(
[post] => WP_Post Object
(
[ID] => 32217
[post_author] => 12503
[post_date] => 2026-06-01 12:04:52
[post_date_gmt] => 2026-06-01 02:04:52
[post_content] => Four of Australia’s leading health peak bodies have united in calling for investment in general practice pharmacists to make medicine use safer and strengthen multidisciplinary care.
Speaking at an inter-professional panel session at the 2026 GP Pharmacist Symposium today on the Gold Coast, presidents from PSA, Australian Medical Association, Royal Australian College of General Practitioners and Australian Primary Health Care Nurses Association reinforced the clinical need and value of embedding pharmacists within general practice.
The session provided a real-life demonstration of cohesion, bringing together representatives from general practice, medicine, nursing and pharmacy.
Exploring how collaborative healthcare efforts can improve patient care, reduce pressure on hospitals and strengthen the primary care workforce, the panel was made up of extraordinary leaders, including:
td_module_mega_menu Object
(
[post] => WP_Post Object
(
[ID] => 32211
[post_author] => 3410
[post_date] => 2026-06-01 11:39:57
[post_date_gmt] => 2026-06-01 01:39:57
[post_content] => Australia's ADHD medicine supply has stabilised – for now. Here’s what pharmacists need to know before the next shortage hits.
After several years of disruption, Australia's ADHD medicine supply has largely recovered. But the shortages of lisdexamfetamine (Vyvanse) in 2023–24 and methylphenidate in 2025 exposed global supply chain vulnerabilities. With diagnosis rates rising, prescribing arrangements expanding and global manufacturing concentrated overseas, pharmacists may once again find themselves managing the fallout from supply interruptions.
[caption id="attachment_32071" align="alignright" width="300"] Yvette Anderson MPS[/caption]
Yvette Anderson MPS – founder of Spectrum Pharmacist, which sets out to bridge the gap for neurodivergent families – tells AP how preparation, collaboration and neuroaffirming care will be essential when the next shortage arrives.
Yvette Anderson MPS[/caption]
Yvette Anderson MPS – founder of Spectrum Pharmacist, which sets out to bridge the gap for neurodivergent families – tells AP how preparation, collaboration and neuroaffirming care will be essential when the next shortage arrives.
A fragile recovery
The shortages that dominated ADHD care over the past 3 years have largely eased, Ms Anderson said.
‘Except for one particular strength of long-acting methylphenidate (30 mg), which is a bit hit and miss.’
However, the situation remains precarious.
Australia relies heavily on overseas manufacturing for ADHD medicines, with production historically concentrated in Ireland and Germany. Supply is also influenced by annual manufacturing quotas established by the United States Drug Enforcement Administration (DEA), which shape global production volumes for controlled substances.
When the Vyvanse patent expired in the United States in 2023, demand for generic lisdexamfetamine surged – rapidly straining international supply chains. More recently, scrutiny of DEA production quotas under the Make America Healthy Again policy framework contributed to a significant reduction in manufacturing, creating flow-on effects internationally.
Due to international uproar, production recommenced. ‘But it's still not probably at the rate and quantity needed globally, so we're always going to sit in a bit of a precarious area,’ Ms Anderson said.
Demand is surging
While supply remains vulnerable, Australia's ADHD treatment landscape is also undergoing significant change.
Initiation of ADHD medicines was largely restricted to paediatricians and psychiatrists. Now, most states and territories have introduced pathways that allow appropriately trained GPs to diagnose and prescribe ADHD medicines.
'A number of GPs in different states have either already been trained, or their state or territory has put laws in place to allow them to be trained to diagnose and prescribe medications,' Ms Anderson said.
Diagnosis rates are also increasing due to changes in diagnosis criteria. More accessible and affordable treatment pathways will help to ensure patients can receive timely access to care.
Importantly, diagnosis does not automatically lead to pharmacological treatment.
'Just because there is potentially going to be an increase in the number of Australians getting diagnosed, that number won't directly correlate to the same number of increasing supply of medication,' Ms Anderson said. 'Medication is only one tiny piece of your management plan, and for some people, medicines aren't the way they want to go, or they don't suit them, or find them effective,'
Nevertheless, more diagnoses and more prescribers are likely to increase demand for medicines supplied through already fragile global supply chains.
Is it possible to get ahead of shortages?
Yes, and no.
Ms Anderson encourages pharmacies to establish systems that allow them to identify and respond to potential disruptions early. This includes monitoring Therapeutic Goods Administration (TGA) medicine shortage alerts.
Once a shortage is identified, pharmacies can use dispensing records to determine which patients may be affected and begin discussions with prescribers before supplies run out.
‘It's a matter of looking at who the prescribers are and reaching out to them and saying, “The TGA has alerted us to this. We know you have a number of patients on this medication. What can we do as a collaborative to support this transition and this shortage period?”’ Ms Anderson said.
As a hospital pharmacist, she was able to pass on information about ADHD medicine shortages to paediatricians – who opened telehealth appointments to turn scripts around quickly.
‘I also communicated with the community pharmacies in my area to ask: Who has stock? Who doesn't? Can we redirect patients?’ Ms Anderson said. ‘Working together as a community, making sure you're embedded in that multidisciplinary team really pays off when there are medication shortages.’
Switching medicines safely
When ADHD medicines are in short supply, patients may need to adjust their medicines; so it’s crucial to be aware of the various formulations, durations of action and release characteristics of different stimulant products.
‘If someone's stabilised on Ritalin, they may go on Ritalin LA or Concerta – but Concerta's duration of action is longer and Ritalin LA's is shorter – so we need to be considering: does this person work through to 6.00 pm? Do we need some immediate release on top of that?’ Ms Anderson said. ‘It's not a straight switch between the medicines.’
The challenges become greater when patients need to move between stimulant classes, such as from lisdexamfetamine to methylphenidate.
‘I've seen people that have quite significant adverse effects after switching to lisdexamfetamine and a couple of days later being unable to function,’ she said. ‘Even though we know someone might be getting a positive effect from stimulants, it doesn't mean it's going to be the same for all stimulants.’
Where stimulant options become unavailable, non-stimulant medicines such as atomoxetine, guanfacine (Intuniv) and clonidine (off-label) can provide alternative management pathways.
‘These non-stimulant options have really good evidence [of efficacy], but they're probably underutilised,' Ms Anderson said. ‘When we do come to another significant shortage, there's only a handful of stimulant medicines – so we need to be able to talk to patients about other options.’
Learn more about supporting patients through ADHD medicine shortages by attending the ADHD care Session at PSA26, held from 31 July to 2 August at the ICC in Sydney.
[post_title] => How to manage ADHD medicine shortages
[post_excerpt] => Australia's ADHD medicine supply has stabilised – for now. Here’s what pharmacists need to know before ADHD medicine shortages strike again.
[post_status] => publish
[comment_status] => open
[ping_status] => open
[post_password] =>
[post_name] => how-to-manage-adhd-medicine-shortages
[to_ping] =>
[pinged] =>
[post_modified] => 2026-06-01 15:15:51
[post_modified_gmt] => 2026-06-01 05:15:51
[post_content_filtered] =>
[post_parent] => 0
[guid] => https://www.australianpharmacist.com.au/?p=32211
[menu_order] => 0
[post_type] => post
[post_mime_type] =>
[comment_count] => 0
[filter] => raw
)
[title_attribute] => How to manage ADHD medicine shortages
[title] => How to manage ADHD medicine shortages
[href] => https://www.australianpharmacist.com.au/how-to-manage-adhd-medicine-shortages/
[module_atts:td_module:private] => Array
(
)
[td_review:protected] => Array
(
)
[is_review:protected] =>
[post_thumb_id:protected] => 32214
[authorType] =>
)
td_module_mega_menu Object
(
[post] => WP_Post Object
(
[ID] => 32207
[post_author] => 12503
[post_date] => 2026-06-01 11:03:42
[post_date_gmt] => 2026-06-01 01:03:42
[post_content] => Two pharmacists were honoured at PSA’s Consultant Pharmacist Conference (CPC26), held from 29–31 May 2026 on the Gold Coast.
Kelly Abbott MPS was awarded 2026 PSA MIMS Consultant Pharmacist of the Year for her outstanding contribution to consultant pharmacy practice and passion for improving patient care throughout her hometown of Gippsland, Victoria.
Tiernan McDonough MPS, based in South Australia, was named 2026 PSA AMH Aged Care Pharmacist of the Year for excellence, leadership, and innovation in aged care pharmacy.
Tireless HMR advocate
Highly regarded across the industry for her work in delivering Home Medicines Reviews, Residential Medication Management Reviews, and providing Quality Use of Medicines (QUM) services, Ms Abbott’s 15 years of service to consultant pharmacy make her a well-deserving winner of this year’s award.
PSA National President, Professor Mark Naunton MPS, said Ms Abbott epitomises many credentialed pharmacists as a passionate and relentless advocate for HMRs, her patients, and the greater credentialed pharmacist workforce.
‘Kelly has continued to be actively involved in grassroots advocacy, notably in recent months during the First Pharmacy Programs Agreement negotiations led by PSA, ensuring the voices of frontline pharmacists and patients were heard,’ he said.
‘Kelly is a loyal, responsive, and reliable contributor to PSA and the Consultant Pharmacist of Australia working groups, while balancing multiple professional roles and caring for her family.
‘Her contributions to PSA and the profession are honourable. We are proud to recognise Kelly as the PSA MIMS Consultant Pharmacist of the Year and celebrate her significant and ongoing impact.’
Aged care champion
Mr McDonough has been recognised for his ongoing dedication and commitment to aged care pharmacy practice through his support for residents and healthcare teams in delivering medication reviews and QUM services.
PSA National President, Professor Mark Naunton MPS, said Mr McDonough embodies the essential role pharmacists play in aged care.
‘Pharmacists working in aged care are critical to ensuring safe and effective medicines use, but Tiernan’s impact extends beyond the aged care sector in which he practices,’ he said.
‘Tiernan has led an important mentoring program that connects pharmacists within the residential aged care profession, to reduce professional isolation and support further workforce development.
‘His work demonstrates his compassion, leadership, and strong drive for improving care for older Australians.’
[post_title] => Consultant pharmacist excellence awarded
[post_excerpt] => Two pharmacists were honoured at PSA’s Consultant Pharmacist Conference (CPC26), held from 29–31 May 2026 on the Gold Coast.
[post_status] => publish
[comment_status] => open
[ping_status] => open
[post_password] =>
[post_name] => top-consultant-pharmacists-honoured-at-cpc26
[to_ping] =>
[pinged] =>
[post_modified] => 2026-06-01 15:18:21
[post_modified_gmt] => 2026-06-01 05:18:21
[post_content_filtered] =>
[post_parent] => 0
[guid] => https://www.australianpharmacist.com.au/?p=32207
[menu_order] => 0
[post_type] => post
[post_mime_type] =>
[comment_count] => 0
[filter] => raw
)
[title_attribute] => Consultant pharmacist excellence awarded
[title] => Consultant pharmacist excellence awarded
[href] => https://www.australianpharmacist.com.au/top-consultant-pharmacists-honoured-at-cpc26/
[module_atts:td_module:private] => Array
(
)
[td_review:protected] => Array
(
)
[is_review:protected] =>
[post_thumb_id:protected] => 32209
[authorType] =>
)
td_module_mega_menu Object
(
[post] => WP_Post Object
(
[ID] => 31968
[post_author] => 8963
[post_date] => 2026-06-01 09:56:44
[post_date_gmt] => 2026-05-31 23:56:44
[post_content] =>
Case scenarioJohn, 62, regularly visits your community pharmacy. Following his referral to a gastroenterologist, he presents to purchase a bowel cleansing kit and asks for advice about his upcoming endoscopy and colonoscopy. John reports a history of difficulty swallowing certain foods and says a family member recently told him that his grandfather had bowel cancer. He is feeling anxious about the procedures and the bowel preparation. |
Endoscopy and colonoscopy are commonly performed gastrointestinal procedures that pharmacists may encounter. Patients may require counselling before and after investigations. Endoscopy refers to procedures that use an endoscope – a thin, flexible tube with a light and camera – to examine internal organs. However, in both clinical and public contexts, particularly in gastroenterology, the term endoscopy is generally used to refer specifically to oesophagoscopy, gastroscopy or duodenoscopy, which involves examining the upper gastrointestinal tract (oesophagus, stomach and duodenum). Colonoscopy is the term used specifically for the examination of the lower gastrointestinal tract (colon/bowel) and includes the entire colon, rectum and terminal ileum. Both procedures are usually performed by gastroenterologists, although colonoscopies may also be performed by colorectal surgeons.1
Learning objectivesAfter reading this article, pharmacists should be able to:
Competency standards (2016) addressed: 1.1, 1.4, 1.5, 2.2, 3.1, 3.5 Accreditation number: PSAAP2606BW Accreditation expiry: 31/05/2028 Accreditation points: 1.5 Group 2 credits |
Both endoscopy and colonoscopy may be used for diagnostic and therapeutic purposes. Standard diagnostic functions include inspection, biopsy, photography and videorecording; diagnostic observations may include focal benign or malignant lesions, mucosal changes, luminal obstruction, motility and strictures. Common therapeutic interventions include polypectomy, dilation of strictures, stent placement, removal of foreign bodies, gastrostomy and treatment of gastrointestinal bleeding.2

EndoscopyOesophagogastroduodenoscopy, more commonly referred to as endoscopy, is a valuable diagnostic procedure in the upper gastrointestinal tract, and is used mainly for detection and surveillance of premalignant conditions and cancers.3 It provides a clear view of mucosal surfaces of the oesophagus, stomach and proximal duodenum.2 It is being increasingly used for gastric cancer and oesophageal adenocarcinoma, the latter having an endoscopically detectable (and potentially curable) precursor lesion, Barrett’s oesophagus.3 Other forms of oesophagitis include eosinophilic oesophagitis, a chronic immune-mediated inflammatory disease of the oesophagus that causes dysphagia, food impaction of the oesophagus, and oesophageal strictures.4 |
Endoscopy with biopsy is mainly used for diagnosing stomach and oesophageal cancers. It is usually performed as day surgery and requires patients to fast for 4–6 hours before the procedure. Before insertion of the endoscope, the throat is sprayed with a local anaesthetic, and a sedative or appropriate anaesthesia is given to the patient. If any suspicious-looking areas are seen, a biopsy may be taken for examination by a pathologist, with results usually available within a few days.1
An endoscopy generally takes about 15 minutes. The patient may have a sore throat afterwards and feel a little bloated. An endoscopic ultrasound is sometimes performed at the same time as a standard endoscopy. This procedure helps determine whether a cancer has spread into the oesophageal wall, nearby tissues or lymph nodes, and is also used to determine whether the patient is a suitable candidate for surgery.1
Endoscopies have a low risk of adverse events. The risk of mortality is rated as 1 in 10,000.5 Possible adverse events include infection, perforation, bleeding, aspiration and cardiopulmonary events.5


ColonoscopyColonoscopy is the main procedure used to investigate or diagnose bowel cancer and is usually performed as day surgery. In 2023–24, there were 691,514 Medicare-subsidised colonoscopies in Australia, of which approximately 20% were repeat procedures performed within 2 years and 10 months of a previous colonoscopy.6 A colonoscope is a tiny camera on a long, flexible tube that is used to examine the entire length of the large bowel. It is inserted into the anus and up into the rectum and colon. Air or carbon dioxide is passed into the colon. Any abnormal tissue can be detected, polyps removed and biopsies taken for further examination. The process usually takes about 20–30 minutes.1 |
Indications for colonoscopy may include:
Australians aged 50–74 years with an average risk of bowel cancer should participate in Australia’s National Bowel Cancer Screening Program. Since 1 July 2024, people aged 45–49 years can also participate on request. A free screening kit is mailed to participants. This is a faecal occult blood test which uses a small stool sample that can be easily collected at home and returned for laboratory testing.1
Before insertion of the colonoscope, an appropriate anaesthesia is given. Patients may feel drowsy or weak prior to and particularly after the colonoscopy, therefore it is recommended that patients do not drive or travel alone. An occasional side effect of a colonoscopy is temporary wind pain and flatulence, especially if air rather than carbon dioxide is passed into the bowel during the procedure.1 More serious but rare complications include perforation (1 in 1,000 risk), infection and risks of sedation and anaesthesia.7
High-quality colonoscopy is required to ensure that cancers and adenomas are not missed, and to avoid adverse patient experiences. The Australian Colonoscopy Clinical Care Standard lists nine key factors, three of which are relevant to pharmacists7:
Informed decision making and patient consent: this is essentially between the patient and gastroenterologist, but patients may seek further advice.
Adequate bowel preparation: bowel cleansing products are generally provided by pharmacies. Reporting and patient follow-up: this is essentially between the patient and gastroenterologist, but patients may seek further advice.
For patients, bowel preparation can be an unpleasant experience.7 However, the importance of adequate bowel preparation and following instructions must be emphasised to patients to ensure that there is a clear view of the mucosa to detect adenomas.8 If the bowel preparation is inadequate, there is not only a lower likelihood of adenoma detection, but it may also result in longer procedures, repeat procedures, more risk to the patient and higher costs.7
Suboptimal bowel preparation has been reported in up to 25% of patients.9 Factors which may affect the bowel preparation include product choice, timing, diet and patient compliance.8
Product selection
Various products are available for bowel cleansing, and there is no consensus on which product is best.7 Over the years, many products have been used, including enemas, mannitol and large-volume solutions. The use of polyethylene glycol 3350 (PEG), also known as macrogol, represented a breakthrough in 1980. This was followed by sodium phosphate preparations in 1990, but these are no longer used because of the risk of renal toxicity.9 Today, many products are based on PEG with the addition of a balanced electrolyte solution and sometimes combined with sodium sulfate, bisacodyl or sodium ascorbate/ascorbic acid.
PEG does not alter the histology of the mucosa and rarely causes electrolyte disturbances.9 The volume to be consumed is an important consideration, with split dosing highly recommended for 4 L solutions (2 L the evening before and 2 L 5–6 hours before the procedure).7,9
The addition of ascorbic acid allows for lower-volume PEG formulations, as its osmotic effect enhances bowel cleansing. These preparations are generally better tolerated due to the reduced volume and improved palatability (typically ~3 L).9
Products based on magnesium citrate combined with sodium picosulfate are also available. These are as effective as PEG-based products and may be better tolerated.9 However, they need to be used with caution in elderly patients, those with renal impairment or heart disease, and those taking certain medications such as diuretics.9 It should be noted that bisacodyl and picosulfate are both converted in the gut to the same active metabolite.9 The decision regarding choice of product is generally made by the gastroenterologist depending on the patient characteristics.
Dietary modification
For a few days before the colonoscopy, patients may be advised to avoid high-fibre foods (such as fruit, vegetables, brown rice, wholegrain pasta, bran, cereals, nuts and seeds). Instead, low-fibre foods (such as white rice, white bread, meat, chicken, fish, yoghurt, cheese, potato and pumpkin) may be recommended.1 Patients may also be advised to drink only fluids, such as water, clear broth, black tea and coffee, and clear fruit juice (without pulp) for 12–24 hours before the procedure to prevent dehydration.1
Special populations and precautions
Patients requiring special care include those with lower gastrointestinal bleeding, chronic kidney disease, inflammatory bowel disease, heart failure, chronic constipation, those who have had previous bowel resection or are pregnant or lactating, as well as hospital inpatients and older people.8 Prevention of dehydration in patients undergoing bowel preparation minimises the risk of severe adverse events.9
Adverse events following bowel preparation are uncommon but may be potentially serious. The product selection decision is an important one because the risk of adverse effects is increased if there is inadequate hydration, inappropriate dosing and inappropriate patient selection.9
Hyponatraemia, because of vomiting or diarrhoea, can develop after taking any colonoscopy preparation. Vomiting may reduce plasma volume and increase thirst. Patients with renal insufficiency, hypothyroidism, mineralocorticoid deficiency, liver cirrhosis, heart failure, or those on diuretics, NSAIDs or ACEIs are at an increased risk of hyponatraemia.9
Improving tolerability
Various methods have been promoted to improve the tolerability and palatability of bowel cleansing preparations. These include chilling the preparation, drinking the preparation through a straw, drinking clear pulp-free juices or approved herbal teas, and chewing gum or various confectioneries concomitantly with the preparation.8

Post colonoscopy syndrome occurs in a third of patients and is more common in females and after lengthy procedures. Symptoms, which include abdominal pain, discomfort and bloating, persist for up to 48 hours following the procedure and may be caused by transient changes in the gut microbiota due to bowel cleansing.8
If a colonoscopy reveals anything of concern, a biopsy is taken for testing by a pathologist. Imaging (such as abdominal scans) may be required. If bowel cancer is diagnosed, scans will determine what stage it is at. Tissue taken at the time of colonoscopy may be tested for genetic changes in the cancer cells, which can guide the choice of treatment.1
Pharmacists are frequently involved in supporting patients undergoing endoscopy and colonoscopy, particularly in the community setting. Patients may seek reassurance about the purpose, safety and expected after-effects of these procedures. Providing clear, evidence-based information can help reduce anxiety. Pharmacists can provide advice regarding colonoscopy preparation by counselling on correct use of bowel cleansing products, split dosing, diet and hydration. They should identify patients at high risk of complications, including older adults, those with renal or cardiac impairment, or those taking diuretics, NSAIDs or ACE inhibitors, and provide tailored advice. Following the procedure, pharmacists can reassure patients about common after effects.
Endoscopy and colonoscopy are widely performed diagnostic and therapeutic procedures that play a central role in the detection and management of gastrointestinal disease, including malignancy. Although generally safe, both procedures require appropriate patient education and awareness of potential adverse effects.
Pharmacists are well positioned to support patients before and after these procedures by providing counselling, identifying individuals at increased risk of complications, and promoting effective bowel preparation for colonoscopy. Through proactive engagement, pharmacists can contribute to improved patient experience and procedural outcomes.
Case scenario continuedYou explain the purpose of both procedures and reassure John that they are commonly performed and generally low risk. You counsel him on how to use the bowel cleansing kit, emphasising the importance of following the instructions carefully. You also discuss the dietary and fluid modifications required in the days leading up to the colonoscopy and provide tips to improve tolerability of the preparation. Following the procedures, John returns to the pharmacy with a prescription for budesonide 1 mg orally dispersible tablets. He explains that he has been diagnosed with eosinophilic oesophagitis and that his oesophagus has been dilated. He also mentions that two polyps were removed from his colon. You reassure John and provide counselling on the correct use of his new medicine. |
[cpd_submit_answer_button]
DR Barry Werth PhD, BPharm (Hons), MBA, MPS is a pharmacist with over 50 years’ experience in the Australian pharmaceutical industry, holding a PhD in epidemiology/pharmacoepidemiology, an honours degree in pharmacy and an MBA. He is a Research Affiliate of the University of Sydney and holds senior advisory roles with MND Australia, alongside ongoing consultancy work for pharmaceutical companies.
Rose-marie Pennisi (she/her), BPharm, MBA, MPS
Cancer Council Australia. Cancer Council. Sydney: Cancer Council Australia; 2025. At: www.cancer.org.au
Early DS, Ben-Menachem T, Decker GA, et al. Appropriate use of GI endoscopy. Gastrointest Endosc. 2012;75(6):1127–31.
Januszewicz W, Kaminski MF. Quality indicators in diagnostic upper gastrointestinal endoscopy. Ther Adv Gastroenterol. 2020;13:1756284820916693.
Muir A, Falk GW. Eosinophilic esophagitis: a review. JAMA. 2021;326(13):1310–8.
Ben-Menachem T, Decker GA, Early DS, et al. Adverse events of upper GI endoscopy. Gastrointest Endosc. 2012;76(4):707–18.
Australian Commission on Safety and Quality in Health Care. Atlas focus report: colonoscopy. Sydney: Australian Commission on Safety and Quality in Health Care; 2025. At: www.safetyandquality.gov.au
Duggan A, Skinner IJ, Bhasale AL. All colonoscopies are not created equal: why Australia now has a clinical care standard for colonoscopy. Med J Aust. 2018;209(10):427–30.
Di Leo M, Iannone A, Arena M, et al. Novel frontiers of agents for bowel cleansing for colonoscopy. World J Gastroenterol. 2021;27(45):7748.
Adamcewicz M, Bearelly D, Porat G, et al. Mechanism of action and toxicities of purgatives used for colonoscopy preparation. Expert Opin Drug Metab Toxicol. 2011;7(1):89–101.
Get your weekly dose of the news and research you need to help advance your practice.
Protected by Google reCAPTCHA v3.

Australian Pharmacist is the official journal for Pharmaceutical Society of Australia Ltd.